Korean J Ophthalmol.
2014 Jun;28(3):246-256. 10.3341/kjo.2014.28.3.246.
Bone Marrow-derived Mesenchymal Stem Cells Affect Immunologic Profiling of Interleukin-17-secreting Cells in a Chemical Burn Mouse Model
- Affiliations
-
- 1Department of Ophthalmology, Seoul National University College of Medicine, Seoul, Korea. kmk9@snu.ac.kr
- 2Laboratory of Ocular Regenerative Medicine and Immunology, Seoul Artificial Eye Center, Seoul National University Hospital Biomedical Research Institute, Seoul, Korea.
- KMID: 2133324
- DOI: http://doi.org/10.3341/kjo.2014.28.3.246
Abstract
- PURPOSE
This study investigated interleukin (IL)-17-secreting cell involvement in sterile inflammation, and evaluated the effect of mesenchymal stem cells (MSCs) on IL-17-secreting cell immunologic profiling.
METHODS
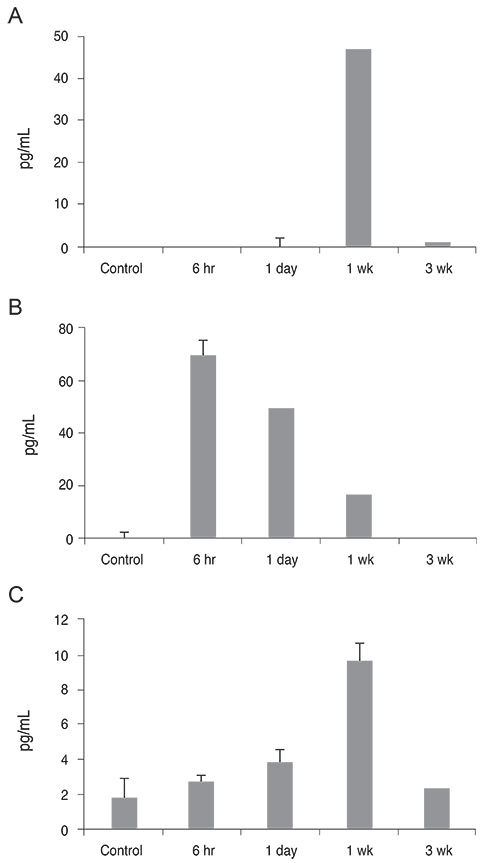
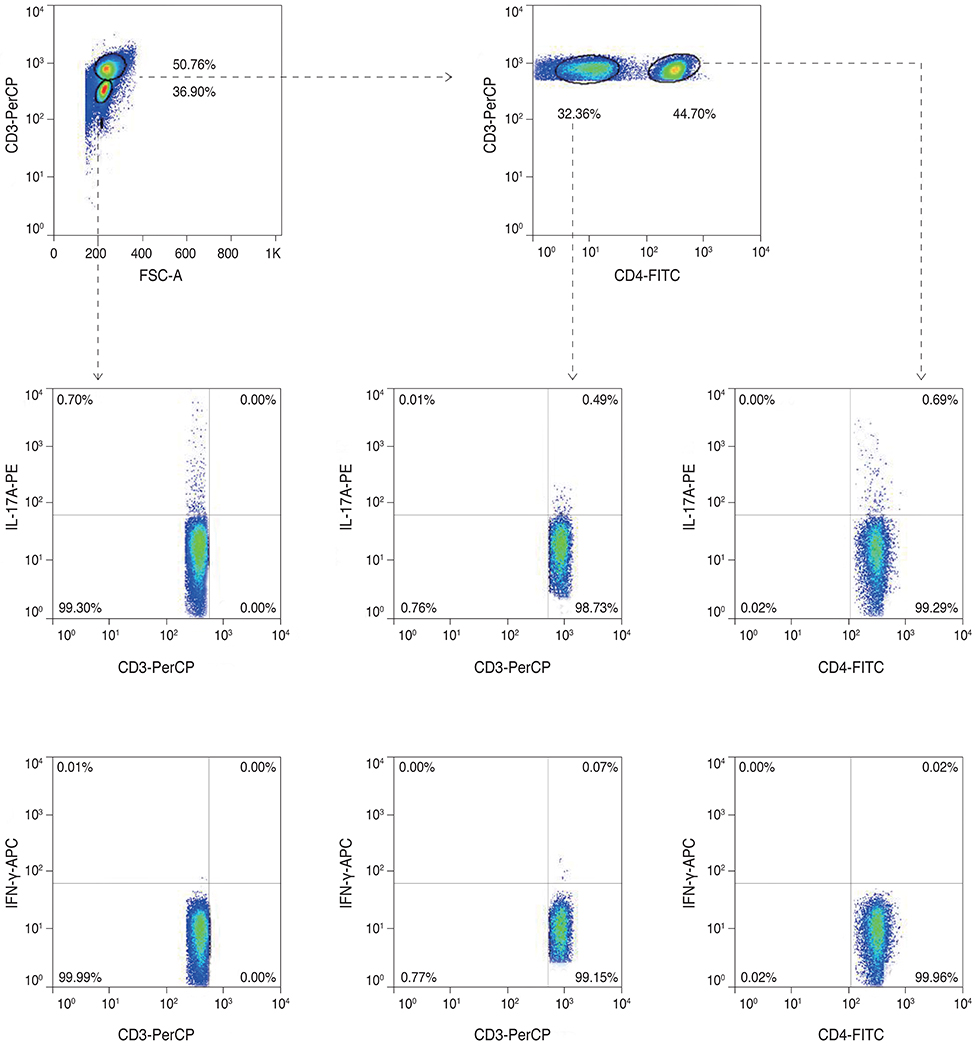
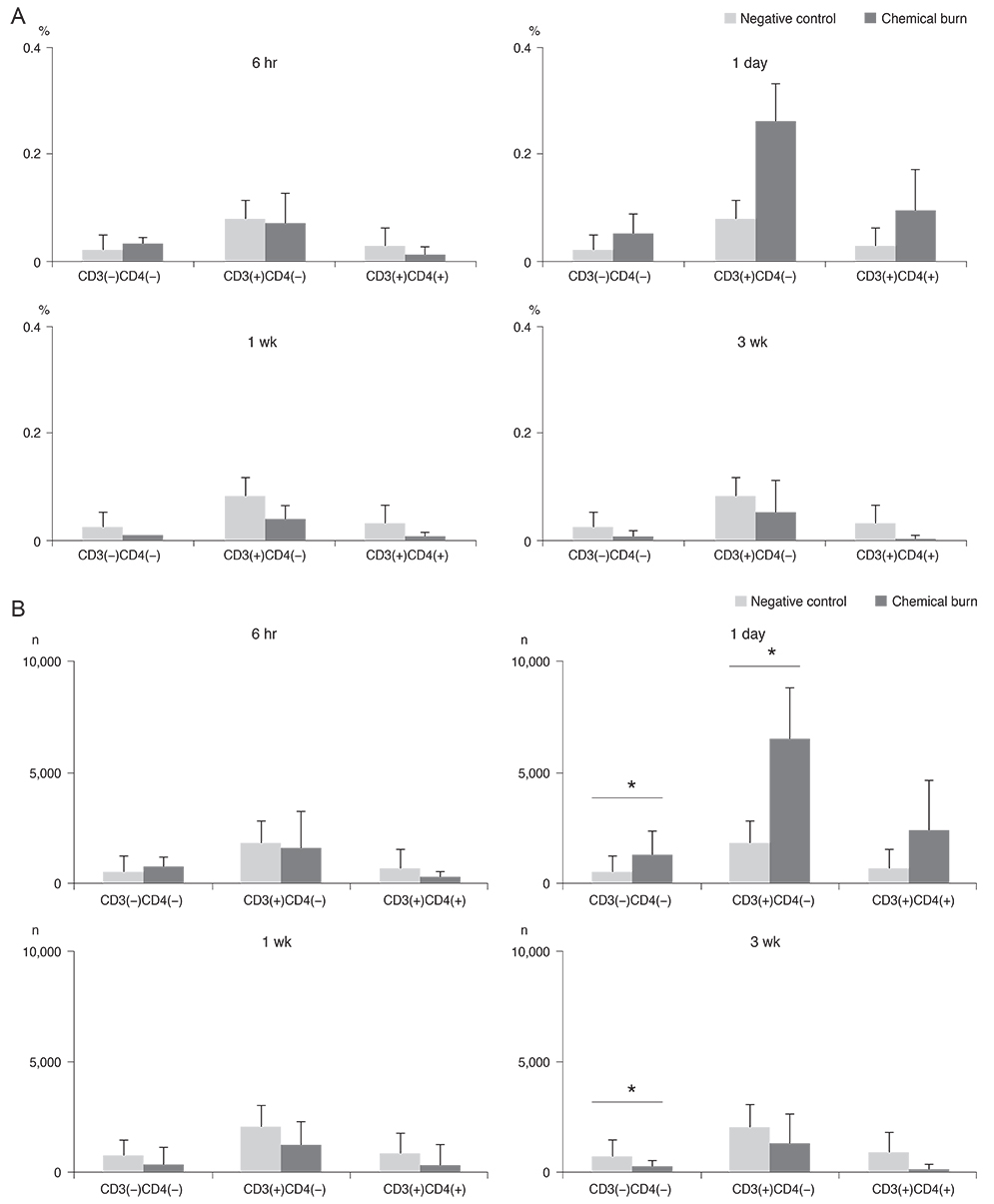
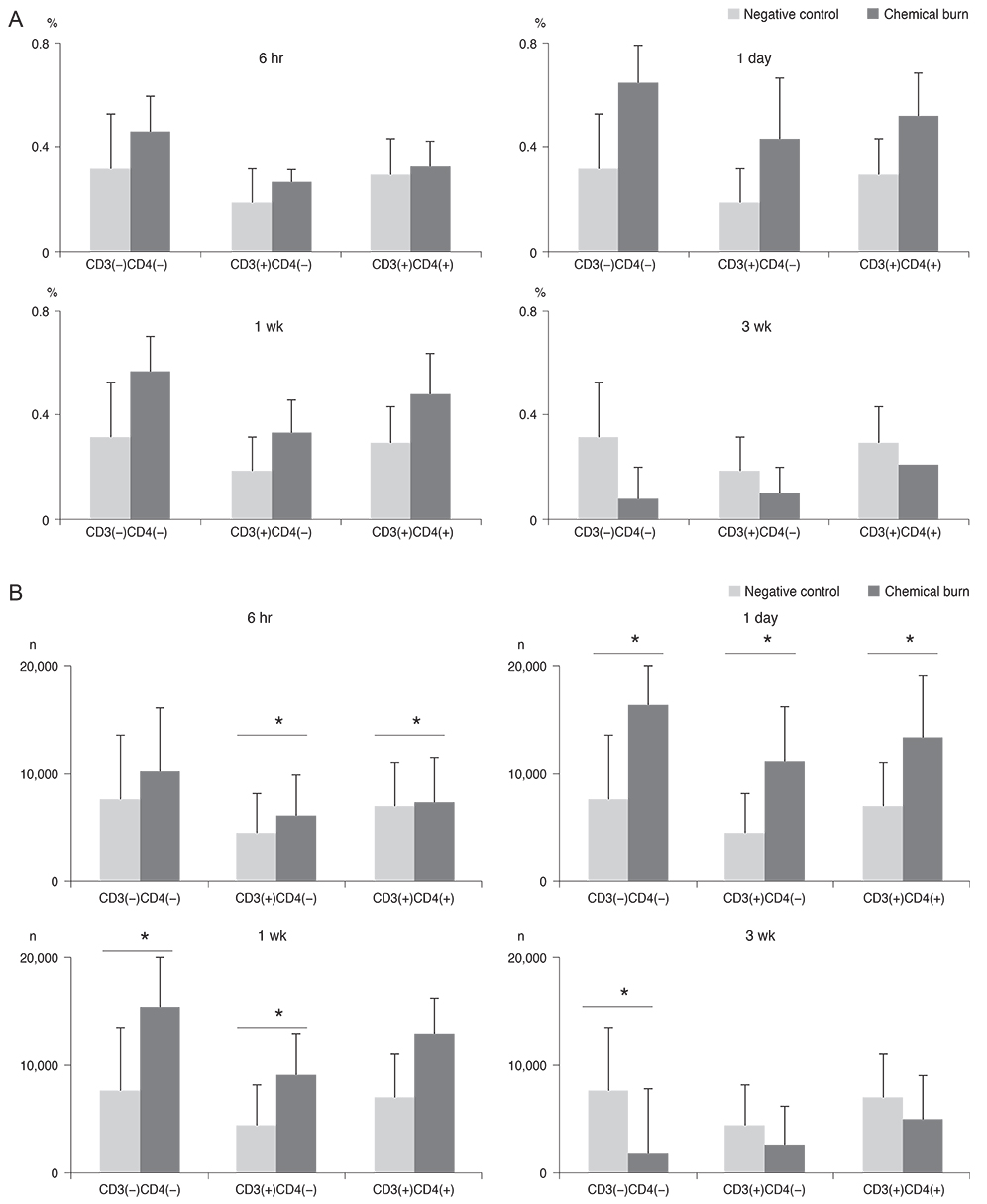
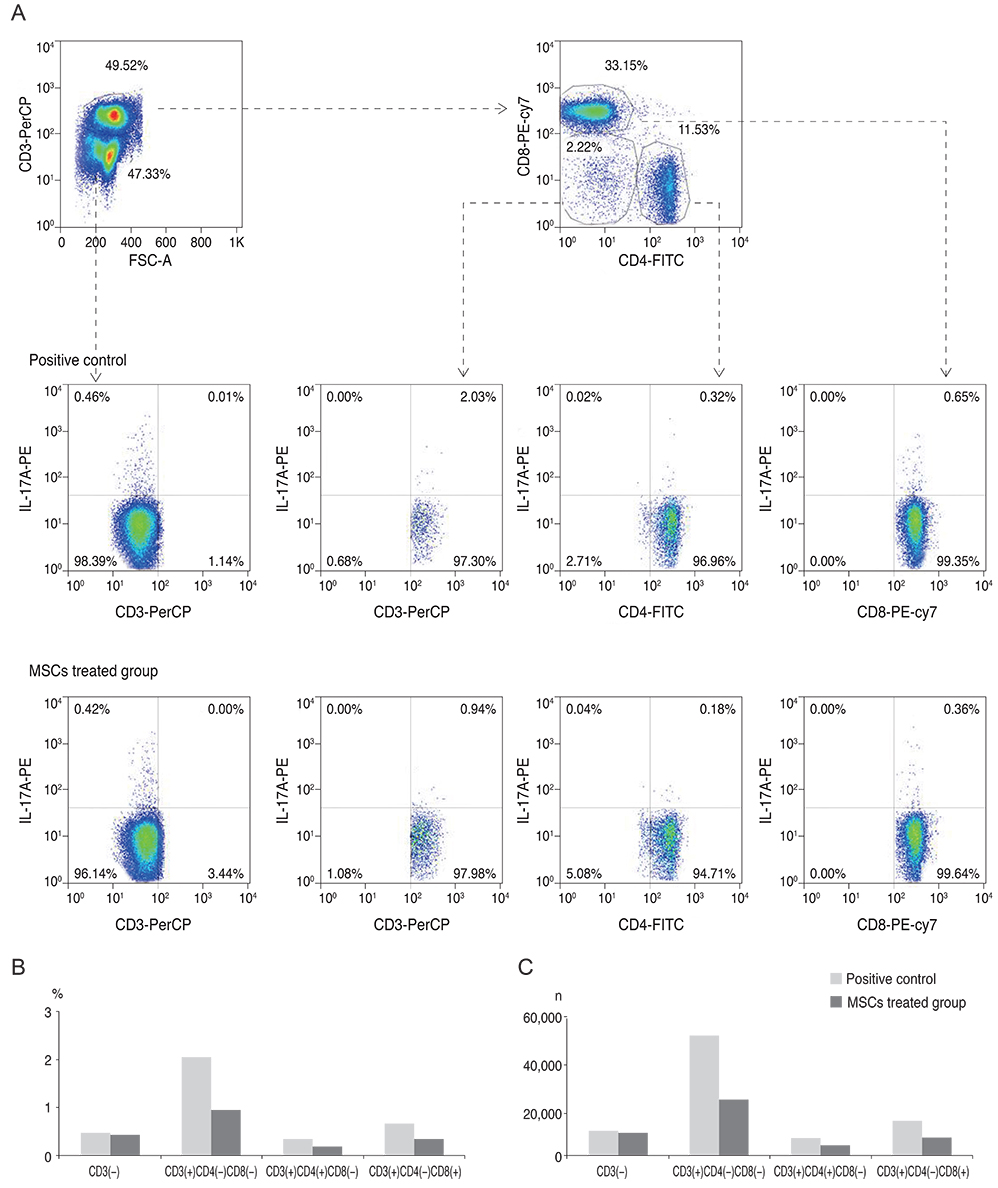
Twenty mice were sacrificed at time points of 6 hours, 1 day, 1 week, and 3 weeks (each group, n = 5) after the cornea was chemically injured with 0.5N NaOH; IL-17 changes in the cornea were evaluated using enzyme-linked immunosorbent assay. Further, IL-17 secreting cells were assessed in the cervical lymph nodes by a flow cytometer. Rat MSCs were applied intraperitoneally in a burn model (n = 10), IL-17-secreting T helper 17 (Th17) cell and non-Th17 cell changes were checked using a flow cytometer in both cornea and cervical lymph nodes at 1week, and compared with those in the positive control (n = 10).
RESULTS
IL-17 was highest in the cornea at 1 week, while, in the cervical lymph nodes, IL-17-secreting cells showed early increase at 6 hours, and maintained the increase through 1 day to 1 week, and levels returned to the basal level at 3 weeks. Specifically, the non-Th17 cells secreted IL-17 earlier than the Th17 cells. When the MSCs were applied, IL-17 secretion was reduced in CD3(+)CD4(-)CD8(-), CD3(+)CD4(+)CD8(-), and CD3(+) CD4(-)CD8(+) cells of the cervical lymph nodes by 53.7%, 43.8%, and 50.8%, respectively. However, in the cornea, IL-17 secretion of CD3(+)CD4(-)CD8(-) cells was completely blocked.
CONCLUSIONS
The results indicated that both IL-17-secreting non-Th17 and Th17 cells were involved in the chemical burn model, and MSCs appeared to mainly modulate non-Th17 cells and also partially suppress the Th17 cells.
MeSH Terms
-
Animals
Burns, Chemical/*immunology/metabolism/pathology
Cells, Cultured
Disease Models, Animal
Enzyme-Linked Immunosorbent Assay
Eye Burns/*immunology/metabolism/pathology
Flow Cytometry
*Immunity, Cellular
Interleukin-17/*secretion
Male
Mesenchymal Stromal Cells/immunology/pathology/*secretion
Mice
Mice, Inbred C57BL
Interleukin-17
Figure
Reference
-
1. Bassi EJ, Aita CA, Camara NO. Immune regulatory properties of multipotent mesenchymal stromal cells: where do we stand? World J Stem Cells. 2011; 3:1–8.2. Bassi EJ, de Almeida DC, Moraes-Vieira PM, Camara NO. Exploring the role of soluble factors associated with immune regulatory properties of mesenchymal stem cells. Stem Cell Rev. 2012; 8:329–342.3. Prockop DJ, Oh JY. Mesenchymal stem/stromal cells (MSCs): role as guardians of inflammation. Mol Ther. 2012; 20:14–20.4. Shi Y, Hu G, Su J, et al. Mesenchymal stem cells: a new strategy for immunosuppression and tissue repair. Cell Res. 2010; 20:510–518.5. Chen SJ, Wang YL, Fan HC, et al. Current status of the immunomodulation and immunomediated therapeutic strategies for multiple sclerosis. Clin Dev Immunol. 2012; 2012:970789.6. Raychaudhuri SP. Role of IL-17 in psoriasis and psoriatic arthritis. Clin Rev Allergy Immunol. 2013; 44:183–193.7. Marwaha AK, Leung NJ, McMurchy AN, Levings MK. TH17 cells in autoimmunity and immunodeficiency: protective or pathogenic? Front Immunol. 2012; 3:129.8. Maddur MS, Miossec P, Kaveri SV, Bayry J. Th17 cells: biology, pathogenesis of autoimmune and inflammatory diseases, and therapeutic strategies. Am J Pathol. 2012; 181:8–18.9. Hirota K, Ahlfors H, Duarte JH, Stockinger B. Regulation and function of innate and adaptive interleukin-17-producing cells. EMBO Rep. 2012; 13:113–120.10. Fossiez F, Banchereau J, Murray R, et al. Interleukin-17. Int Rev Immunol. 1998; 16:541–551.11. Duffy MM, Pindjakova J, Hanley SA, et al. Mesenchymal stem cell inhibition of T-helper 17 cell- differentiation is triggered by cell-cell contact and mediated by prostaglandin E2 via the EP4 receptor. Eur J Immunol. 2011; 41:2840–2851.12. Duffy MM, Ritter T, Ceredig R, Griffin MD. Mesenchymal stem cell effects on T-cell effector pathways. Stem Cell Res Ther. 2011; 2:34.13. Ghannam S, Pene J, Moquet-Torcy G, et al. Mesenchymal stem cells inhibit human Th17 cell differentiation and function and induce a T regulatory cell phenotype. J Immunol. 2010; 185:302–312.14. Ohshima M, Yamahara K, Ishikane S, et al. Systemic transplantation of allogenic fetal membrane-derived mesenchymal stem cells suppresses Th1 and Th17 T cell responses in experimental autoimmune myocarditis. J Mol Cell Cardiol. 2012; 53:420–428.15. Qu X, Liu X, Cheng K, et al. Mesenchymal stem cells inhibit Th17 cell differentiation by IL-10 secretion. Exp Hematol. 2012; 40:761–770.16. Prigione I, Benvenuto F, Bocca P, et al. Reciprocal interactions between human mesenchymal stem cells and gammadelta T cells or invariant natural killer T cells. Stem Cells. 2009; 27:693–702.17. Sasaki JR, Zhang Q, Schwacha MG. Burn induces a Th-17 inflammatory response at the injury site. Burns. 2011; 37:646–651.18. Ye J, Yao K, Kim JC. Mesenchymal stem cell transplantation in a rabbit corneal alkali burn model: engraftment and involvement in wound healing. Eye (Lond). 2006; 20:482–490.19. Ye J, Lee SY, Kook KH, Yao K. Bone marrow-derived progenitor cells promote corneal wound healing following alkali injury. Graefes Arch Clin Exp Ophthalmol. 2008; 246:217–222.20. Yao L, Li ZR, Su WR, et al. Role of mesenchymal stem cells on cornea wound healing induced by acute alkali burn. PLoS One. 2012; 7:e30842.21. Oh JY, Kim MK, Shin MS, et al. The anti-inflammatory and anti-angiogenic role of mesenchymal stem cells in corneal wound healing following chemical injury. Stem Cells. 2008; 26:1047–1055.22. Ma Y, Xu Y, Xiao Z, et al. Reconstruction of chemically burned rat corneal surface by bone marrow-derived human mesenchymal stem cells. Stem Cells. 2006; 24:315–321.23. Church D, Elsayed S, Reid O, et al. Burn wound infections. Clin Microbiol Rev. 2006; 19:403–434.24. Oh JY, Choi H, Lee RH, et al. Identification of the HSPB4/TLR2/NF-κB axis in macrophage as a therapeutic target for sterile inflammation of the cornea. EMBO Mol Med. 2012; 4:435–448.25. Tazetdinova NR, Iurovskaia NN. Changes in the T-lymphocyte subpopulations in patients with severe eye burns. Oftalmol Zh. 1990; (6):335–338.26. Zhao M, Chen J, Yang P. Immunologic experimental studies on the alkali burn of cornea in rats. Zhonghua Yan Ke Za Zhi. 2000; 36:40–42. 4427. Darlington PJ, Boivin MN, Renoux C, et al. Reciprocal Th1 and Th17 regulation by mesenchymal stem cells: Implication for multiple sclerosis. Ann Neurol. 2010; 68:540–545.28. Rubio D, Garcia-Castro J, Martin MC, et al. Spontaneous human adult stem cell transformation. Cancer Res. 2005; 65:3035–3039.29. Wang Y, Huso DL, Harrington J, et al. Outgrowth of a transformed cell population derived from normal human BM mesenchymal stem cell culture. Cytotherapy. 2005; 7:509–519.30. Miura M, Miura Y, Padilla-Nash HM, et al. Accumulated chromosomal instability in murine bone marrow mesenchymal stem cells leads to malignant transformation. Stem Cells. 2006; 24:1095–1103.31. Zhou YF, Bosch-Marce M, Okuyama H, et al. Spontaneous transformation of cultured mouse bone marrow-derived stromal cells. Cancer Res. 2006; 66:10849–10854.32. Aguilar S, Nye E, Chan J, et al. Murine but not human mesenchymal stem cells generate osteosarcoma-like lesions in the lung. Stem Cells. 2007; 25:1586–1594.33. Tolar J, Nauta AJ, Osborn MJ, et al. Sarcoma derived from cultured mesenchymal stem cells. Stem Cells. 2007; 25:371–379.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Concise Review: Differentiation of Human Adult Stem Cells Into Hepatocyte-like Cells In vitro
- Clinical Safety and Efficacy of Autologous Bone Marrow-Derived Mesenchymal Stem Cell Transplantation in Sensorineural Hearing Loss Patients
- Bone marrow-derived stem cells contribute to regeneration of the endometrium
- Clinical Use of Mesenchymal Stem Cells in Bone Regeneration
- Bone Marrow-Derived Mesenchymal Stem Cells for Regenerative Medicine