Korean J Physiol Pharmacol.
2009 Aug;13(4):281-285. 10.4196/kjpp.2009.13.4.281.
Naringin Protects against Rotenone-induced Apoptosis in Human Neuroblastoma SH-SY5Y Cells
- Affiliations
-
- 1Department of Pharmacology and Kohwang Medical Research Institute, School of Medicine, Kyung Hee University, Seoul 130-701, Korea. jhchung@khu.ac.kr
- 2East-West Neo Medical Center, School of Medicine, Kyung Hee University, Seoul 130-701, Korea.
- 3Department of Physical Medicine and Rehabilitation, School of Medicine, Kyung Hee University, Seoul 130-701, Korea.
- KMID: 2071682
- DOI: http://doi.org/10.4196/kjpp.2009.13.4.281
Abstract
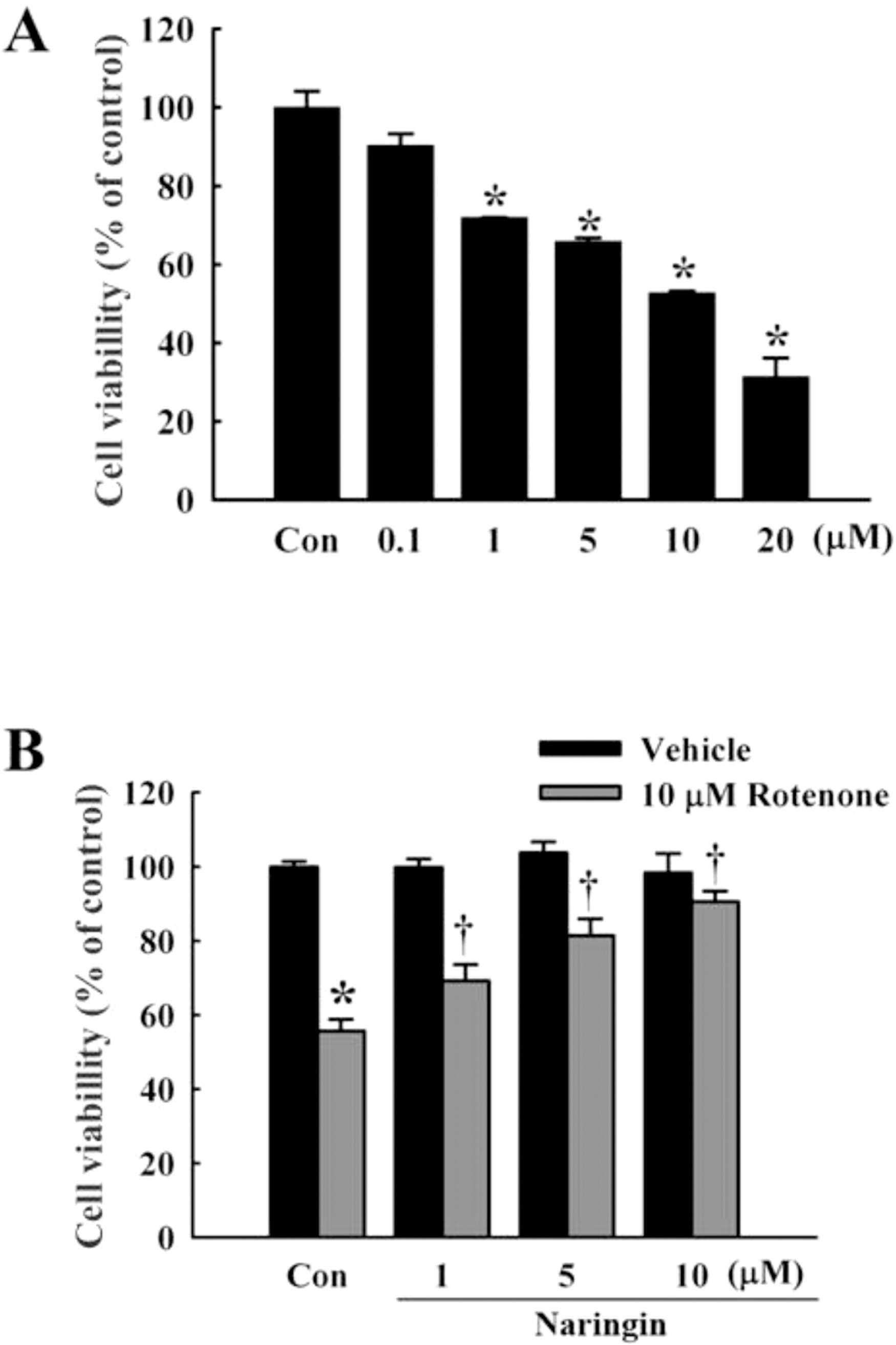
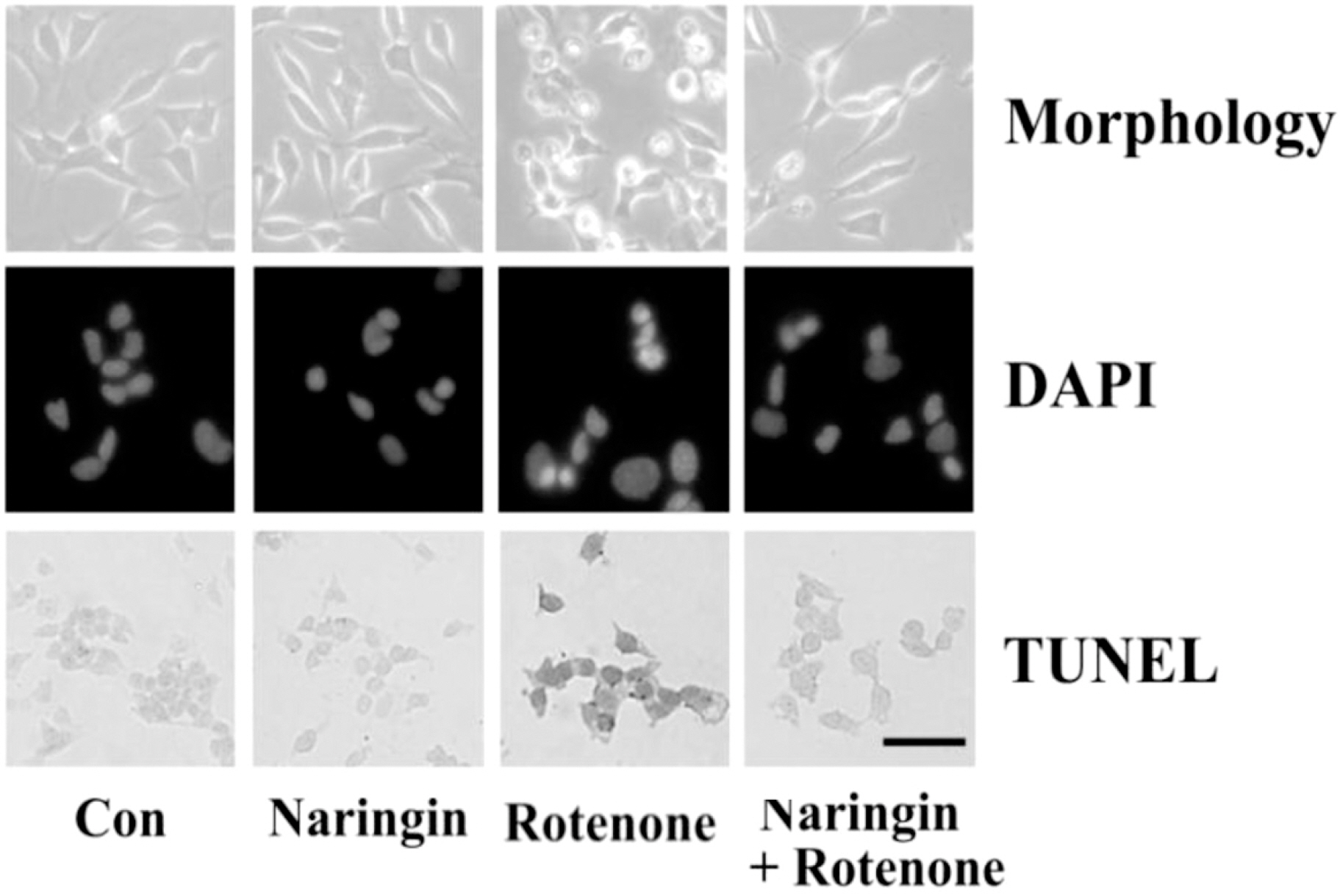
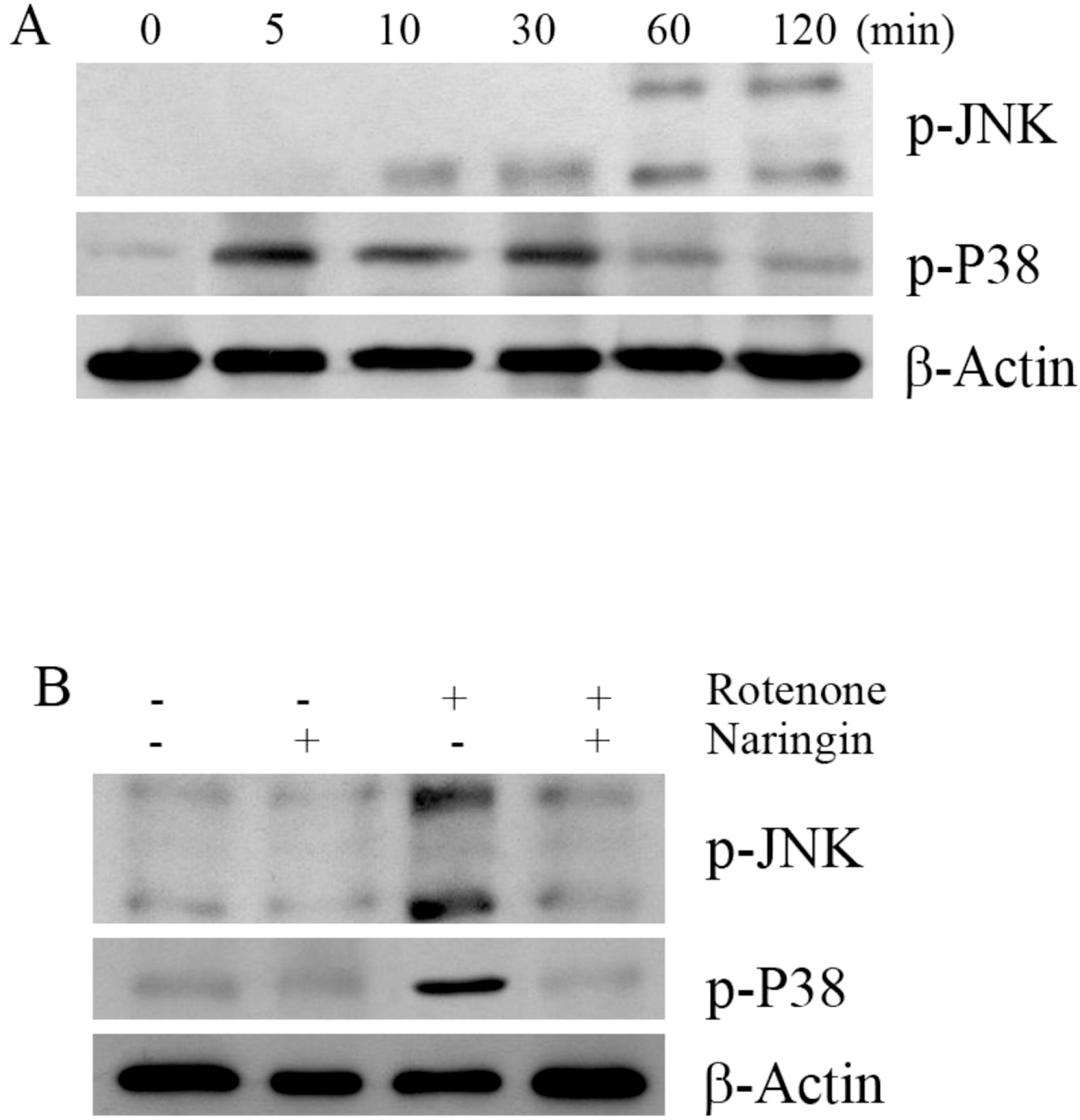
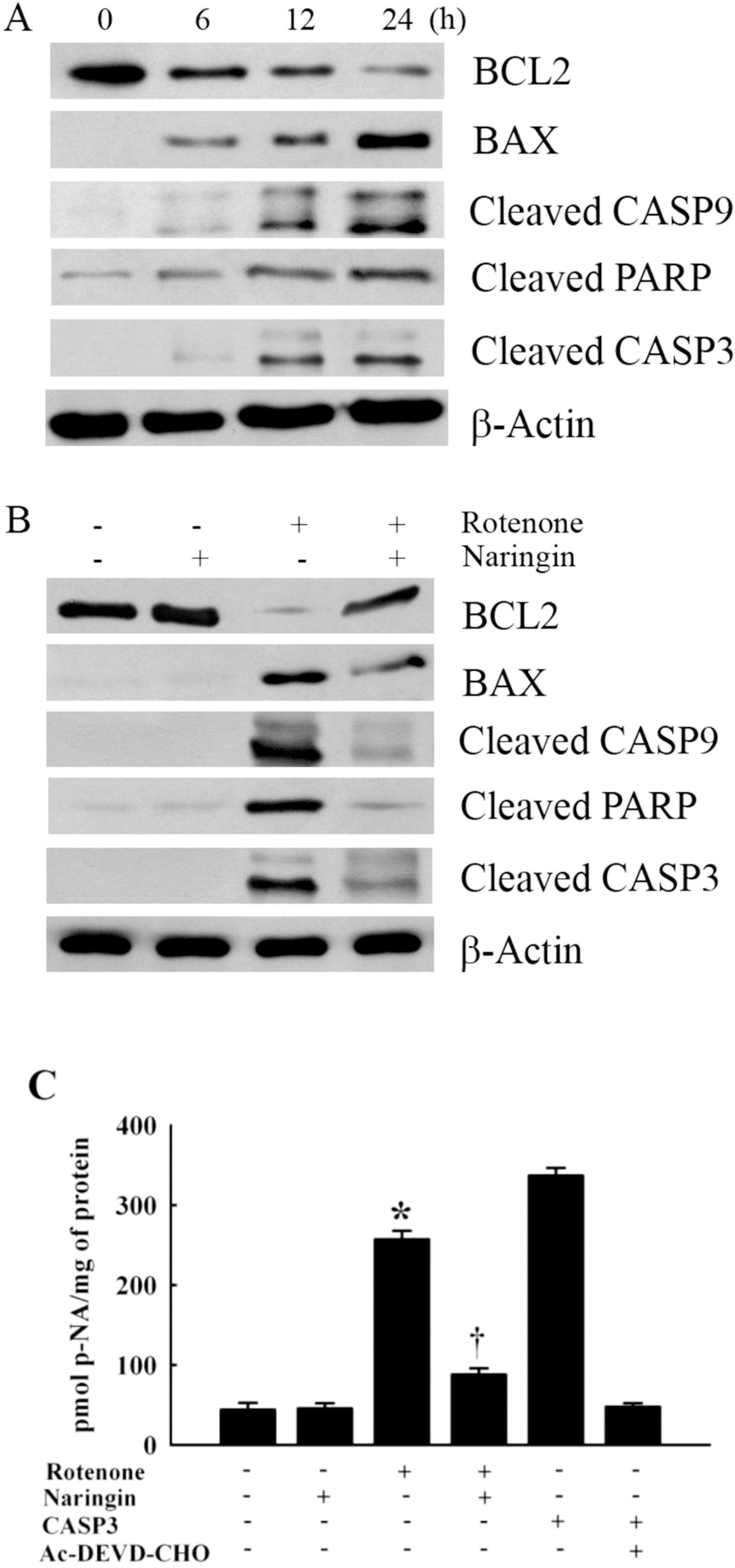
- Rotenone, a mitochondrial complex I inhibitor, can induce the pathological features of Parkinson's disease (PD). In the present study, naringin, a grapefruit flavonoid, inhibited rotenone-induced cell death in human neuroblastoma SH-SY5Y cells. We assessed cell death and apoptosis by measuring mitogen-activated protein kinase (MAPKs) and caspase (CASPs) activities and by performing 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, 4,6-diamidino-2-phenylindole (DAPI) staining, and terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) staining. Naringin also blocked rotenone-induced phosphorylation of Jun NH2-terminal protein kinase (JNK) and P38, and prevented changes in B-cell CLL/lymphoma 2 (BCL2) and BCL2-associated X protein (BAX) expression levels. In addition, naringin reduced the enzyme activity of caspase 3 and cleavages of caspase 9, poly (ADP-ribose) polymerase (PARP), and caspase 3. These results suggest that naringin has a neuroprotective effect on rotenone-induced cell death in human neuroblastoma SH-SY5Y cells.
Keyword
MeSH Terms
-
Apoptosis
B-Lymphocytes
bcl-2-Associated X Protein
Caspase 3
Caspase 9
Cell Death
Citrus paradisi
Flavanones
Humans
Indoles
Neuroblastoma
Neuroprotective Agents
Parkinson Disease
Phosphorylation
Protein Kinases
Rotenone
Tetrazolium Salts
Thiazoles
Caspase 3
Caspase 9
Flavanones
Indoles
Neuroprotective Agents
Protein Kinases
Rotenone
Tetrazolium Salts
Thiazoles
bcl-2-Associated X Protein
Figure
Cited by 2 articles
-
Cytotoxicities and Quantitative Structure Activity Relationships of B13 Sulfonamides in HT-29 and A549 Cells
Seul Ki Chan Lee, Sang Min Park, Chaeuk Im
Korean J Physiol Pharmacol. 2011;15(6):423-429. doi: 10.4196/kjpp.2011.15.6.423.Cytotoxic Activity and Quantitative Structure Activity Relationships of Arylpropyl Sulfonamides
Yu Jin Hwang, Sang Min Park, Chul Bu Yim, Chaeuk Im
Korean J Physiol Pharmacol. 2013;17(3):237-243. doi: 10.4196/kjpp.2013.17.3.237.
Reference
-
Bradshaw J., Saling M., Hopwood M., Anderson V., Brodtmann A. Fluctuating cognition in dementia with Lewy bodies and Alzheimer's disease is qualitatively distinct. J Neurol Neurosurg Psychiatry. 75:382–387. 2004.
ArticleChang L., Karin M. Mammalian MAP kinase signaling cascades. Nature. 410:37–40. 2000.Davis RJ. Signal transduction by the JNK group of MAP kinases. Cell. 103:239–252. 2000.
ArticleDudley DT., Pang L., Decker SJ., Bridged AJ., Saltiel AR. A synthetic inhibitor of the mitogen-activated protein kinase cascade. Proc Natl Acad Sci USA. 92:7686–7689. 1995.
ArticleGreenamyre JT., Sherer TB., Betarbet R., Panov AV. Complex I and Parkinson's disease. IUBMB Life. 52:135–141. 2001.
ArticleHaenen GR., Paquay J., Korthouwer R., Bast A. Peroxynitrite scavenging by flavonoids. Biochem Biophys Res Commun. 236:591–593. 1997.
ArticleJunn E., Mouradian MM. Apoptotic signaling in dopamine induced cell death: the role of oxidative stress, P38 mitogen-activated protein kinase, cytochrome c and caspases. J Neurochem. 78:374–383. 2001.Kanno S., Shouji A., Asou K., Ishikawa M. Effects of naringin on hydrogen peroxide-induced cytotoxicity and apoptosis in P388 cells. J Pharmacol Sci. 92:166–170. 2003.
ArticleKaul TN., Middlenton E Jr., Ogra PL. Antiviral effect of flavonoids on human viruses. J Med Virol. 15:71–79. 1985.
ArticleKlintworth H., Newhouse K., Li T., Choi WS., Faigle R., Xia Z. Activation of c-Jun N-terminal protein kinase is a common mechanism underlying paraquat- and rotenone-induced dopaminergic cell apoptosis. Toxicol Sci. 97:149–162. 2007.
ArticleLi P., Nijhawan D., Budihardjo I., Srinivasula SM., Ahmad M., Alnemri ES., Wang X. Cytochrome c and dATP dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell. 91:479–489. 2004.Maridonneau-Parini I., Braquet P., Garay RP. Heterogeneous effect of flavonoids on K+-loss and lipid peroxidation induced by oxygen free radicals in human red cells. Pharmacol Res Commun. 18:61–72. 1986.Newhouse K., Hsuan SL., Chang SH., Cai B., Wang Y., Xia Z. Rotenone-induced apoptosis is mediated by p38 and JNK MAP kinases in human dopaminergic SH-SY5Y cells. Toxicol Sci. 79:137–146. 2004.
ArticleNg TB., Liu F., Wang ZT. Antioxidative activity of natural products from plants. Life Sci. 66:709–723. 2000.
ArticleOlanow CW., Perl DP., DeMartino GN., McNaught KS. Lewy-body formation is an aggresome-related process: a hypothesis. Lancet Neurol. 3:496–503. 2004.
ArticlePark HJ., Shin DH., Chung WJ., Leem K., Yoon SH., Hong MS., Chung JH., Bae JH., Hwang JS. Epigallocatechin gallate reduces hypoxia-induced apoptosis in human hepatoma cells. Life Sci. 78:2826–2832. 2006.
ArticlePei W., Liou AK., Chen J. Two caspase-mediated apoptotic pathways induced by rotenone toxicity in cortical neuronal cells. FASEB J. 17:520–522. 2003.
ArticleRamsey CP., Giasson BI. Role of mitochondrial dysfunction in Parkinson's disease-implications for treatment. Drugs Aging. 24:95–105. 2007.Rundén E., Seglen PO., Haug FM., Ottersen OP., Wieloch T., Shamloo M., Laake JH. Regional selective neuronal degeneration after protein phosphatase inhibition in hippocampal slice cultures: evidence for a MAP kinase-dependent mechanism. J Neurosci. 18:7296–7305. 1998.
ArticleSaporito MS., Brown EM., Miller M S., Carswell S. CEP-1347/KT-7515, an inhibitor of c-jun N-terminal kinase activation, attenuates the 1-methyl-4-phenyl tetrahydropyridine-mediated loss of nigrostriatal dopaminergic neurons in vivo. J Pharmacol Exp Ther. 288:421–427. 1999.Saporito MS., Thomas BA., Scott RW. MPTP activates c-Jun NH(2)-terminal kinase (JNK) and its upstream regulatory kinase MKK4 in nigrostriatal neurons in vivo. J Neurochem. 75:1200–1208. 2000.
ArticleShamoto-Nagai M., Maruyama W., Kato Y., Isobe K., Tanaka M., Naoi M., Osawa T. An inhibitor of mitochondrial complex I, rotenone, inactivates proteasome by oxidative modification and induces aggregation of oxidized proteins in SH-SY5Y cells. J Neurosci Res. 74:589–597. 2003.
ArticleShastry BS. Parkinson disease: etiology, pathogenesis and future of gene therapy. Neurosci Res. 41:5–12. 2001.
ArticleTansey MG., McCoy MK., Frank-Cannon TC. Neuroinflammatory mechanisms in Parkinson's disease: potential environmental triggers, pathways, and targets for early therapeutic intervention. Exp Neurol. 208:1–25. 2007.
ArticleTsai SH., Lin-Shiau SY., Lin JK. Suppression of nitric oxide synthase and the down-regulation of the activation of NfkappaB in macropahages by resveratrol. Br J Pharmacol. 126:673–680. 1999.Wang X., Qin ZH., Leng Y., Wang Y., Jin X., Chase TN., Bennett MC. Prostaglandin A1 inhibits rotenone-induced apoptosis in SH-SY5Y cells. J Neurochem. 83:1094–1102. 2002.
ArticleYuan H., Zheng JC., Liu P., Zhang SF., Xu JY., Bai LM. Pathogenesis of Parkinson's disease: oxidative stress, environmental impact factors and inflammatory processes. Neurosci Bull. 23:125–130. 2007.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Morphine on Free Radical-induced Apoptosis of Human Neuroblastoma SH-SY5Y Cells
- Overexpression of SIRT3 Suppresses Oxidative Stress-induced Neurotoxicity and Mitochondrial Dysfunction in Dopaminergic Neuronal Cells
- The increased expression of CD99 in a differentiated neuroblastoma cell line
- Antitumor Effects of Arsenic Trioxide on Neuroblastoma
- Protective effects of mealworm (Tenebrio molitor) extract on N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)–induced cellular toxicity in SH-SY5Y neuroblastoma cells