Korean J Physiol Pharmacol.
2009 Jun;13(3):161-168. 10.4196/kjpp.2009.13.3.161.
Extracellular Signal-regulated Kinase Activation Is Required for Serine 727 Phosphorylation of STAT3 in Schwann Cells in vitro and in vivo
- Affiliations
-
- 1Department of Physiology, Medical Science Research Institute, College of Medicine, Dong-A University, Busan 602-714, Korea. phwantae@dau.ac.kr
- 2Department of Microbiology, Medical Science Research Institute, College of Medicine, Dong-A University, Busan 602-714, Korea.
- KMID: 2071666
- DOI: http://doi.org/10.4196/kjpp.2009.13.3.161
Abstract
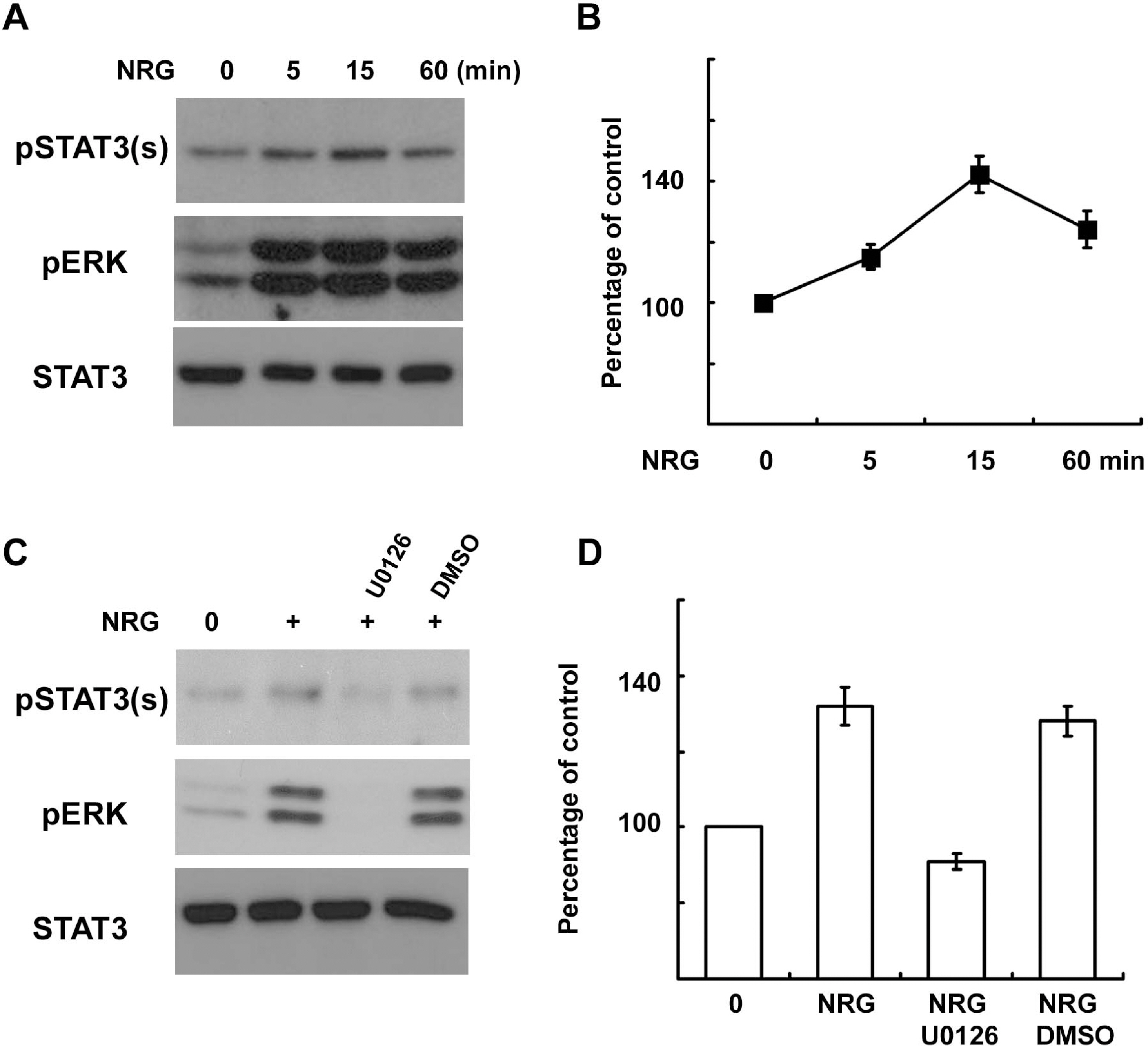
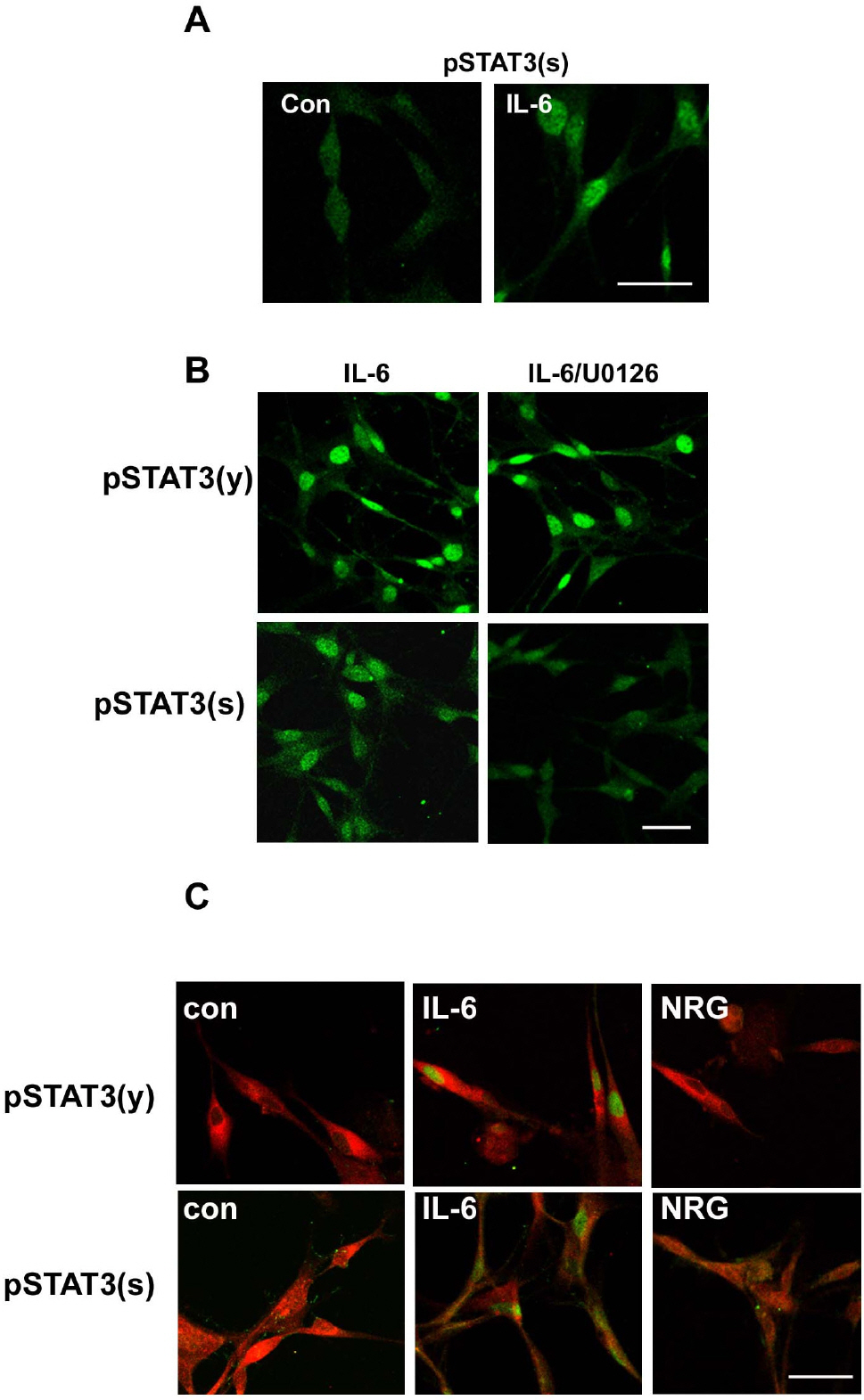
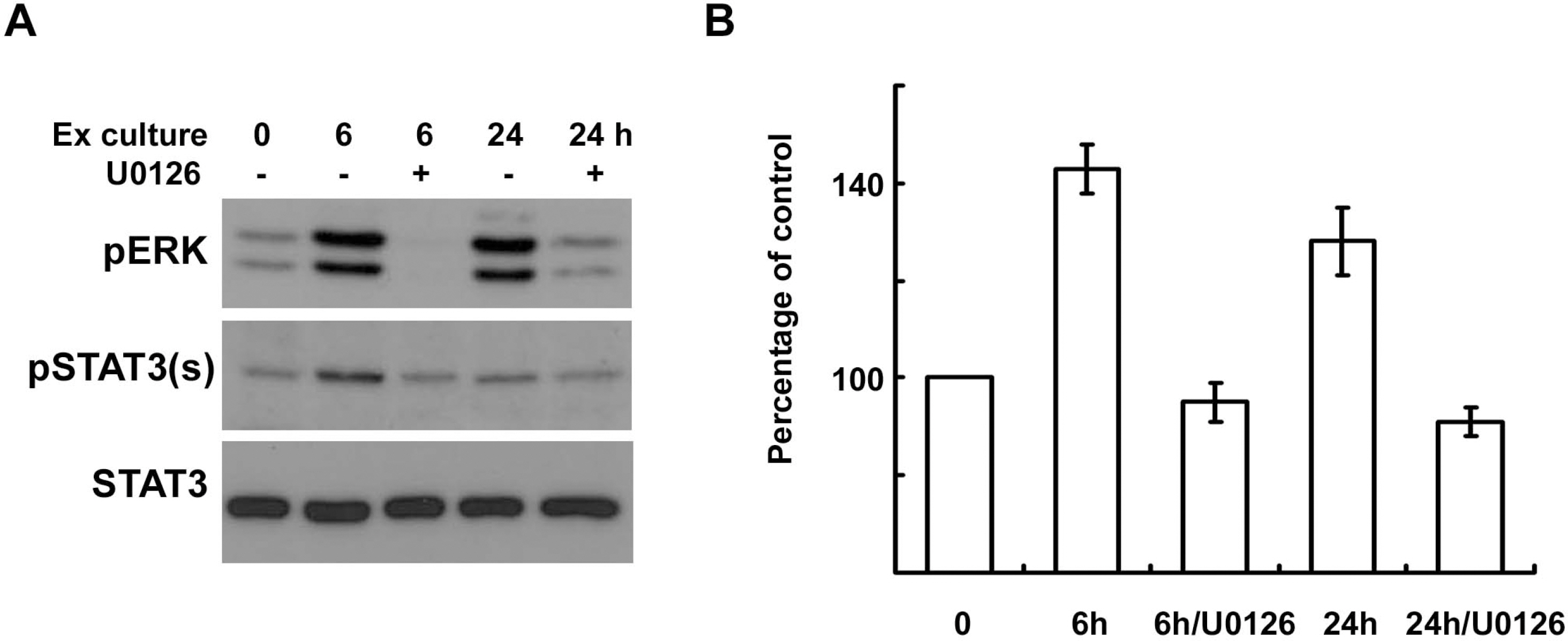
- In the peripheral nerves, injury-induced cytokines and growth factors perform critical functions in the activation of both the MEK/ERK and JAK/STAT3 pathways. In this study, we determined that nerve injury-induced ERK activation was temporally correlated with STAT3 phosphorylation at the serine 727 residue. In cultured Schwann cells, we noted that ERK activation is required for the serine phosphorylation of STAT3 by neuropoietic cytokine interleukin-6 (IL-6). Serine phosphorylated STAT3 by IL-6 was transported into Schwann cell nuclei, thereby indicating that ERK may regulate the transcriptional activity of STAT3 via the induction of serine phosphorylation of STAT3. Neuregulin-1 (NRG) also induced the serine phosphorylation of STAT3 in an ERK-dependent fashion. In contrast with the IL-6 response, serine phosphorylated STAT3 induced by NRG was not detected in the nucleus, thus indicating the non-nuclear function of serine phosphorylated STAT3 in response to NRG. Finally, we determined that the inhibition of ERK prevented injury-induced serine phosphorylation of STAT3 in an ex-vivo explants culture of the sciatic nerves. Collectively, the results of this study show that ERK may be an upstream kinase for the serine phosphorylation of STAT3 induced by multiple stimuli in Schwann cells after peripheral nerve injury.
Keyword
MeSH Terms
-
Cell Nucleus
Cytokines
Intercellular Signaling Peptides and Proteins
Interleukin-6
Neuregulin-1
Peripheral Nerve Injuries
Peripheral Nerves
Phosphorylation
Phosphotransferases
Schwann Cells
Sciatic Nerve
Serine
Cytokines
Intercellular Signaling Peptides and Proteins
Interleukin-6
Neuregulin-1
Phosphotransferases
Serine
Figure
Reference
-
Battle TE., Frank DA. The role of STATs in apoptosis. Curr Mol Med. 2:381–392. 2002.
ArticleBleuel A., Monard D. Regulation of protease nexin-1 and angiotensin II receptor subtype 1 expression: inverse relationship in experimental models of nerve injury. J Neurosci Res. 42:562–570. 1995.
ArticleCafferty WB., Gardiner NJ., Gavazzi I., Powell J., McMahon SB., Heath JK., Munson J., Cohen J., Thompson SW. Leukemia inhibitory factor determines the growth status of injured adult sensory neurons. J Neurosci. 21:7161–7170. 2001.
ArticleChung J., Uchida E., Grammer TC., Blenis J. STAT3 serine phosphorylation by ERK-dependent and -independent pathways negatively modulates its tyrosine phosphorylation. Mol Cell Biol. 17:6508–6516. 1997.
ArticleFu SY., Gordon T. The cellular and molecular basis of peripheral nerve regeneration. Mol Neurobiol. 14:67–116. 1997.
ArticleGartsbein M., Alt A., Hashimoto K., Nakajima K., Kuroki T., Tennenbaum T. The role of protein kinase C delta activation and STAT3 Ser727 phosphorylation in insulin-induced keratinocyte proliferation. J Cell Sci. 119:470–481. 2006.Guertin AD., Zhang DP., Mak KS., Alberta JA., Kim HA. Microanatomy of axon/glial signaling during Wallerian degeneration. J Neurosci. 25:3478–3487. 2005.
ArticleHai M., Muja N., DeVries GH., Quarles RH., Patel PI. Comparative analysis of Schwann cell lines as model systems for myelin gene transcription studies. J Neurosci Res. 69:497–508. 2002.
ArticleHarrisingh MC., Perez-Nadales E., Parkinson DB., Malcolm DS., Mudge AW., Lloyd AC. The Ras/Raf/ERK signalling pathway drives Schwann cell dedifferentiation. EMBO J. 23:3061–3071. 2004.
ArticleJain N., Zhang T., Fong SL., Lim CP., Cao X. Repression of Stat3 activity by activation of mitogen-activated protein kinase (MAPK). Oncogene. 17:3157–3167. 1998.
ArticleJessen KR., Mirsky R. Negative regulation of myelination: relevance for development, injury, and demyelinating disease. Glia. 56:1552–1565. 2008.
ArticleJessen KR., Mirsky R. The origin and development of glial cells in peripheral nerves. Nat Rev Neurosci. 6:671–682. 2005.
ArticleJirsova K., Sodaar P., Mandys V., Bar PR. Cold jet: a method to obtain pure Schwann cell cultures without the need for cytotoxic, apoptosis-inducing drug treatment. J Neurosci Methods. 78:133–137. 1997.Kamimura D., Ishihara K., Hirano T. IL-6 signal transduction and its physiological roles: the signal orchestration model. Rev Physiol Biochem Pharmacol. 149:1–38. 2003.
ArticleLee HK., Seo IA., Park HK., Park YM., Ahn KJ., Yoo YH., Park HT. Nidogen is a prosurvival and promigratory factor for adult Schwann cells. J Neurochem. 102:686–698. 2007.
ArticleLee HK., Seo IA., Suh DK., Hong JI., Yoo YH., Park HT. Interleukin-6 is required for the early induction of glial fibrillary acidic protein in Schwann cells during Wallerian degeneration. J Neurochem. 108:776–786. 2009.
ArticleLee N., Neitzel KL., Devlin BK., MacLennan AJ. STAT3 phosphorylation in injured axons before sensory and motor neuron nuclei: potential role for STAT3 as a retrograde signaling transcription factor. J Comp Neurol. 474:535–545. 2004.
ArticleMonje PV., Bartlett Bunge M., Wood PM. Cyclic AMP synergistically enhances neuregulin-dependent ERK and Akt activation and cell cycle progression in Schwann cells. Glia. 53:649–659. 2006.
ArticleNg DC., Lin BH., Lim CP., Huang G., Zhang T., Poli V., Cao X. Stat3 regulates microtubules by antagonizing the depolymerization activity of stathmin. J Cell Biol. 172:245–257. 2006a.
ArticleNg YP., Cheung ZH., Ip NY. STAT3 as a downstream mediator of Trk signaling and functions. J Biol Chem. 281:15636–15644. 2006b.
ArticlePlaza-Menacho I., van der Sluis T., Hollema H., Gimm O., Buys CH., Magee AI., Isacke CM., Hofstra RM., Eggen BJ. Ras/ERK1/2-mediated STAT3 Ser727 phosphorylation by familial medullary thyroid carcinoma-associated RET mutants induces full activation of STAT3 and is required for c-fos promoter activation, cell mitogenicity, and transformation. J Biol Chem. 282:6415–6424. 2007.
ArticleQiu J., Cafferty WB., McMahon SB., Thompson SW. Conditioning injury-induced spinal axon regeneration requires signal transducer and activator of transcription 3 activation. J Neurosci. 25:1645–1653. 2005.
ArticleReich NC., Liu L. Tracking STAT nuclear traffic. Nat Rev Immunol. 6:602–612. 2006.
ArticleSheu JY., Kulhanek DJ., Eckenstein FP. Differential patterns of ERK and STAT3 phosphorylation after sciatic nerve transection in the rat. Exp Neurol. 166:392–402. 2000.
ArticleShi X., Zhang H., Paddon H., Lee G., Cao X., Pelech S. Phosphorylation of STAT3 serine-727 by cyclin-dependent kinase 1 is critical for nocodazole-induced mitotic arrest. Biochemistry. 45:5857–5867. 2006.
ArticleThomson CE., Griffiths IR., McCulloch MC., Kyriakides E., Barrie JA., Montague P. In vitro studies of axonally-regulated Schwann cell genes during Wallerian degeneration. J Neurocytol. 22:590–602. 1993.
ArticleWen Z., Zhong Z., Darnell JE Jr. Maximal activation of transcription by Stat1 and Stat3 requires both tyrosine and serine phosphorylation. Cell. 82:241–250. 1995.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Cysteine-179 of I kappa B kinase beta plays a critical role in enzyme activation by promoting phosphorylation of activation loop serines
- Peroxisome Proliferator-activated Receptor-gamma Inhibits the Activation of STAT3 in Cerulein-stimulated Pancreatic Acinar Cells
- Sustained Intracellular Acidosis Triggers the Naâº/H⺠Exchager-1 Activation in Glutamate Excitotoxicity
- STAT3 and ERK Signaling Pathways Are Implicated in the Invasion Activity by Oncostatin M through Induction of Matrix Metalloproteinases 2 and 9
- Phosphorylation of chicken protein tyrosine phosphatase 1 by casein kinase II in vitro