Korean J Physiol Pharmacol.
2015 Nov;19(6):491-497. 10.4196/kjpp.2015.19.6.491.
Epigallocatechin-3-Gallate (EGCG) Attenuates Traumatic Brain Injury by Inhibition of Edema Formation and Oxidative Stress
- Affiliations
-
- 1Intensive Care Unit, Tianjin Huanhu Hospital, Tianjin Key Laboratory of Cerebral Vascular and Neurodegenerative Diseases, Tianjin 300060, PR China.
- 2Intensive Care Unit, Tianjin First Center Hospital, Tianjin Institute of Emergency Medicine, Tianjin 300192, PR China. yongqiangwangdr@163.com
- KMID: 2070788
- DOI: http://doi.org/10.4196/kjpp.2015.19.6.491
Abstract
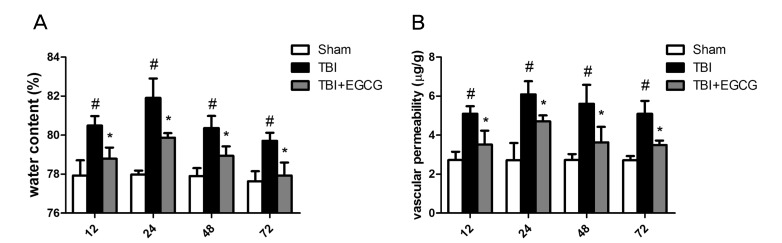
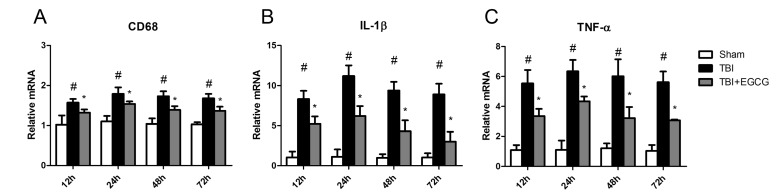
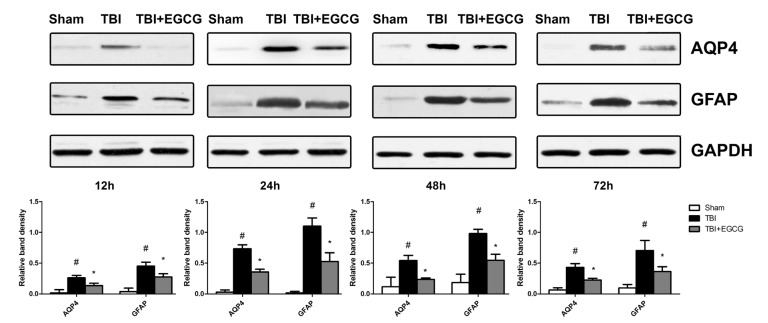
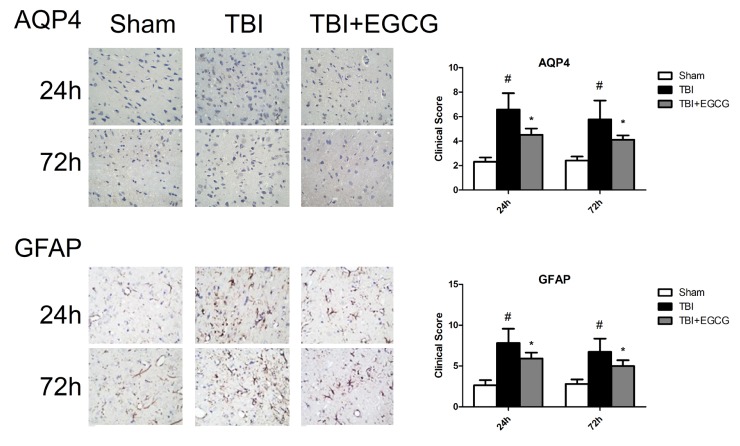
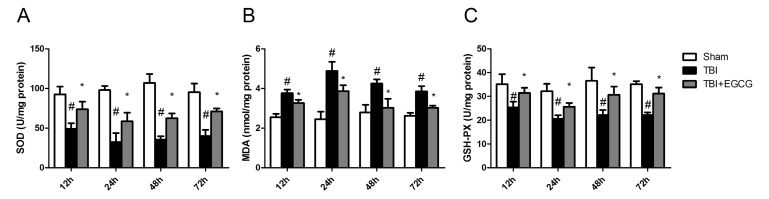
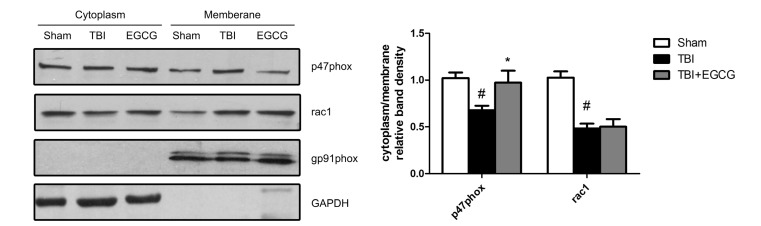
- Traumatic brain injury (TBI) is a major cause of mortality and long-term disability, which can decrease quality of life. In spite of numerous studies suggesting that Epigallocatechin-3-gallate (EGCG) has been used as a therapeutic agent for a broad range of disorders, the effect of EGCG on TBI remains unknown. In this study, a weight drop model was established to evaluate the therapeutic potential of EGCG on TBI. Rats were administered with 100 mg/kg EGCG or PBS intraperitoneally. At different times following trauma, rats were sacrificed for analysis. It was found that EGCG (100 mg/kg, i.p.) treatment significantly reduced brain water content and vascular permeability at 12, 24, 48, 72 hour after TBI. Real-time PCR results revealed that EGCG inhibited TBI-induced IL-1beta and TNF-alpha mRNA expression. Importantly, CD68 mRNA expression decreasing in the brain suggested that EGCG inhibited microglia activation. Western blotting and immunohistochemistry results showed that administering of EGCG significantly inhibited the levels of aquaporin-4 (AQP4) and glial fibrillary acidic protein (GFAP) expression. TBI-induced oxidative stress was remarkably impaired by EGCG treatment, which elevated the activities of SOD and GSH-PX. Conversely, EGCG significantly reduced the contents of MDA after TBI. In addition, EGCG decreased TBI-induced NADPH oxidase activation through inhibition of p47phox translocation from cytoplasm to plasma membrane. These data demonstrate that EGCG treatment may be an effective therapeutic strategy for TBI and the underlying mechanism involves inhibition of oxidative stress.
Keyword
MeSH Terms
-
Animals
Blotting, Western
Brain
Brain Injuries*
Capillary Permeability
Cell Membrane
Cytoplasm
Edema*
Glial Fibrillary Acidic Protein
Immunohistochemistry
Microglia
Mortality
NADP
NADPH Oxidase
Oxidative Stress*
Quality of Life
Rats
Real-Time Polymerase Chain Reaction
RNA, Messenger
Tumor Necrosis Factor-alpha
Water
Glial Fibrillary Acidic Protein
NADP
NADPH Oxidase
RNA, Messenger
Tumor Necrosis Factor-alpha
Water
Figure
Cited by 1 articles
-
Voluntary stand-up physical activity enhances endurance exercise capacity in rats
Dae Yun Seo, Sung Ryul Lee, Hyo-Bum Kwak, Kyo Won Seo, Robin A McGregor, Ji Young Yeo, Tae Hee Ko, Saranhuu Bolorerdene, Nari Kim, Kyung Soo Ko, Byoung Doo Rhee, Jin Han
Korean J Physiol Pharmacol. 2016;20(3):287-295. doi: 10.4196/kjpp.2016.20.3.287.
Reference
-
2. Greve MW, Zink BJ. Pathophysiology of traumatic brain injury. Mt Sinai J Med. 2009; 76:97–104. PMID: 19306379.
Article3. Ziebell JM, Morganti-Kossmann MC. Involvement of pro- and anti-inflammatory cytokines and chemokines in the pathophysiology of traumatic brain injury. Neurotherapeutics. 2010; 7:22–30. PMID: 20129494.
Article4. Sharp DJ, Scott G, Leech R. Network dysfunction after traumatic brain injury. Nat Rev Neurol. 2014; 10:156–166. PMID: 24514870.
Article5. Marklund N, Bakshi A, Castelbuono DJ, Conte V, McIntosh TK. Evaluation of pharmacological treatment strategies in traumatic brain injury. Curr Pharm Des. 2006; 12:1645–1680. PMID: 16729876.
Article6. Tyurin VA, Tyurina YY, Borisenko GG, Sokolova TV, Ritov VB, Quinn PJ, Rose M, Kochanek P, Graham SH, Kagan VE. Oxidative stress following traumatic brain injury in rats: quantitation of biomarkers and detection of free radical intermediates. J Neurochem. 2000; 75:2178–2189. PMID: 11032908.7. Choi BY, Jang BG, Kim JH, Lee BE, Sohn M, Song HK, Suh SW. Prevention of traumatic brain injury-induced neuronal death by inhibition of NADPH oxidase activation. Brain Res. 2012; 1481:49–58. PMID: 22975130.
Article8. Rezai-Zadeh K, Shytle D, Sun N, Mori T, Hou H, Jeanniton D, Ehrhart J, Townsend K, Zeng J, Morgan D, Hardy J, Town T, Tan J. Green tea epigallocatechin-3-gallate (EGCG) modulates amyloid precursor protein cleavage and reduces cerebral amyloidosis in Alzheimer transgenic mice. J Neurosci. 2005; 25:8807–8814. PMID: 16177050.
Article9. Ahn HY, Kim CH, Ha TS. Epigallocatechin-3-gallate regulates Nadph oxidase expression in human umbilical vein endothelial cells. Korean J Physiol Pharmacol. 2010; 14:325–329. PMID: 21165332.
Article10. Surh YJ, Chun KS, Cha HH, Han SS, Keum YS, Park KK, Lee SS. Molecular mechanisms underlying chemopreventive activities of anti-inflammatory phytochemicals: down-regulation of COX-2 and iNOS through suppression of NF-kappa B activation. Mutat Res. 2001; 480-481:243–268. PMID: 11506818.11. Tobi SE, Gilbert M, Paul N, McMillan TJ. The green tea polyphenol, epigallocatechin-3-gallate, protects against the oxidative cellular and genotoxic damage of UVA radiation. Int J Cancer. 2002; 102:439–444. PMID: 12432544.
Article12. Steinmann J, Buer J, Pietschmann T, Steinmann E. Anti-infective properties of epigallocatechin-3-gallate (EGCG), a component of green tea. Br J Pharmacol. 2013; 168:1059–1073. PMID: 23072320.
Article13. Kim KC, Lee C. Reversal of Cisplatin resistance by epigallocatechin gallate is mediated by downregulation of axl and tyro 3 expression in human lung cancer cells. Korean J Physiol Pharmacol. 2014; 18:61–66. PMID: 24634598.
Article14. Fang MZ, Wang Y, Ai N, Hou Z, Sun Y, Lu H, Welsh W, Yang CS. Tea polyphenol (-)-epigallocatechin-3-gallate inhibits DNA methyltransferase and reactivates methylation-silenced genes in cancer cell lines. Cancer Res. 2003; 63:7563–7570. PMID: 14633667.15. Singh BN, Shankar S, Srivastava RK. Green tea catechin, epigallocatechin-3-gallate (EGCG): mechanisms, perspectives and clinical applications. Biochem Pharmacol. 2011; 82:1807–1821. PMID: 21827739.
Article16. Schroeder EK, Kelsey NA, Doyle J, Breed E, Bouchard RJ, Loucks FA, Harbison RA, Linseman DA. Green tea epigallocatechin 3-gallate accumulates in mitochondria and displays a selective antiapoptotic effect against inducers of mitochondrial oxidative stress in neurons. Antioxid Redox Signal. 2009; 11:469–480. PMID: 18754708.
Article17. Levites Y, Amit T, Youdim MB, Mandel S. Involvement of protein kinase C activation and cell survival/cell cycle genes in green tea polyphenol (-)-epigallocatechin 3-gallate neuroprotective action. J Biol Chem. 2002; 277:30574–30580. PMID: 12058035.18. Ge R, Zhu Y, Diao Y, Tao L, Yuan W, Xiong XC. Anti-edema effect of epigallocatechin gallate on spinal cord injury in rats. Brain Res. 2013; 1527:40–46. PMID: 23831998.
Article19. Lee H, Bae JH, Lee SR. Protective effect of green tea polyphenol EGCG against neuronal damage and brain edema after unilateral cerebral ischemia in gerbils. J Neurosci Res. 2004; 77:892–900. PMID: 15334607.
Article20. Feeney DM, Boyeson MG, Linn RT, Murray HM, Dail WG. Responses to cortical injury: I. Methodology and local effects of contusions in the rat. Brain Res. 1981; 211:67–77. PMID: 7225844.
Article21. Shaftel SS, Carlson TJ, Olschowka JA, Kyrkanides S, Matousek SB, O'Banion MK. Chronic interleukin-1beta expression in mouse brain leads to leukocyte infiltration and neutrophil-independent blood brain barrier permeability without overt neurodegeneration. J Neurosci. 2007; 27:9301–9309. PMID: 17728444.22. Li GX, Wang XM, Jiang T, Gong JF, Niu LY, Li N. Berberine prevents intestinal mucosal barrier damage during early phase of sepsis in rat through the toll-like receptors signaling pathway. Korean J Physiol Pharmacol. 2015; 19:1–7. PMID: 25605990.
Article23. Soslow RA, Dannenberg AJ, Rush D, Woerner BM, Khan KN, Masferrer J, Koki AT. COX-2 is expressed in human pulmonary, colonic, and mammary tumors. Cancer. 2000; 89:2637–2645. PMID: 11135226.
Article24. Donkin JJ, Nimmo AJ, Cernak I, Blumbergs PC, Vink R. Substance P is associated with the development of brain edema and functional deficits after traumatic brain injury. J Cereb Blood Flow Metab. 2009; 29:1388–1398. PMID: 19436311.
Article25. Kou Z, VandeVord PJ. Traumatic white matter injury and glial activation: from basic science to clinics. Glia. 2014; 62:1831–1855. PMID: 24807544.
Article26. Huang J, Sun SQ, Lu WT, Xu J, Gan SW, Chen Z, Qiu GP, Huang SQ, Zhuo F, Liu Q, Xu SY. The internalization and lysosomal degradation of brain AQP4 after ischemic injury. Brain Res. 2013; 1539:61–72. PMID: 24070677.
Article27. Metting Z, Wilczak N, Rodiger LA, Schaaf JM, van der Naalt J. GFAP and S100B in the acute phase of mild traumatic brain injury. Neurology. 2012; 78:1428–1433. PMID: 22517109.
Article28. Lambeth JD. NOX enzymes and the biology of reactive oxygen. Nat Rev Immunol. 2004; 4:181–189. PMID: 15039755.
Article29. Adachi N, Tomonaga S, Tachibana T, Denbow DM, Furuse M. (-)-Epigallocatechin gallate attenuates acute stress responses through GABAergic system in the brain. Eur J Pharmacol. 2006; 531:171–175. PMID: 16457806.
Article30. Liu X, Wang Z, Wang P, Yu B, Liu Y, Xue Y. Green tea polyphenols alleviate early BBB damage during experimental focal cerebral ischemia through regulating tight junctions and PKCalpha signaling. BMC Complement Altern Med. 2013; 13:187. PMID: 23870286.
Article31. Suganuma M, Okabe S, Oniyama M, Tada Y, Ito H, Fujiki H. Wide distribution of [3H](-)-epigallocatechin gallate, a cancer preventive tea polyphenol, in mouse tissue. Carcinogenesis. 1998; 19:1771–1776. PMID: 9806157.
Article32. Yu J, Jia Y, Guo Y, Chang G, Duan W, Sun M, Li B, Li C. Epigallocatechin-3-gallate protects motor neurons and regulates glutamate level. FEBS Lett. 2010; 584:2921–2925. PMID: 20488180.
Article33. Lehmann GL, Gradilone SA, Marinelli RA. Aquaporin water channels in central nervous system. Curr Neurovasc Res. 2004; 1:293–303. PMID: 16181079.
Article34. Manley GT, Fujimura M, Ma T, Noshita N, Filiz F, Bollen AW, Chan P, Verkman AS. Aquaporin-4 deletion in mice reduces brain edema after acute water intoxication and ischemic stroke. Nat Med. 2000; 6:159–163. PMID: 10655103.
Article35. Csuka E, Hans VH, Ammann E, Trentz O, Kossmann T, Morganti-Kossmann MC. Cell activation and inflammatory response following traumatic axonal injury in the rat. Neuroreport. 2000; 11:2587–2590. PMID: 10943727.
Article36. Chan PH. Reactive oxygen radicals in signaling and damage in the ischemic brain. J Cereb Blood Flow Metab. 2001; 21:2–14. PMID: 11149664.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Cytoprotective Effect of Epigallocatechin gallate (EGCG) in Oxidative-stressed PC12 Cells Following H2O2 Exposure-Effect of EGCG on Phosphoinositide 3-kinase/Akt and Glycogen Synthase Kinase-3 Pathway
- High-concentration Epigallocatechin Gallate Treatment Causes Endoplasmic Reticulum Stress-mediated Cell Death in HepG2 Cells
- Epigallocatechin-3-gallate Regulates Inducible Nitric Oxide Synthase Expression in Human Umbilical Vein Endothelial Cells
- Epigallocatechin gallate attenuates L-DOPA-induced apoptosis in rat PC12 cells
- The Effect of Delayed Administration of Green Tea Polyphenol, (-)-pigallocatechin-3-gallate, on the Change of Putrescine Level and Hippocampal Neuronal Cell Damage after Transient Global Ischemia in Gerbil