Anat Cell Biol.
2011 Jun;44(2):135-142. 10.5115/acb.2011.44.2.135.
Cannabinoid receptor agonist protects cultured dopaminergic neurons from the death by the proteasomal dysfunction
- Affiliations
-
- 1Department of Physical Medicine and Rehabilitation, Dong-Eui Medical Center, Busan, Korea.
- 2Department of Anatomy, College of Medicine, Kosin University, Busan, Korea. drhkim@kosin.ac.kr
- 3Department of Physical Medicine and Rehabilitation Anatomy, College of Medicine, Kosin University, Busan, Korea.
- KMID: 1447423
- DOI: http://doi.org/10.5115/acb.2011.44.2.135
Abstract
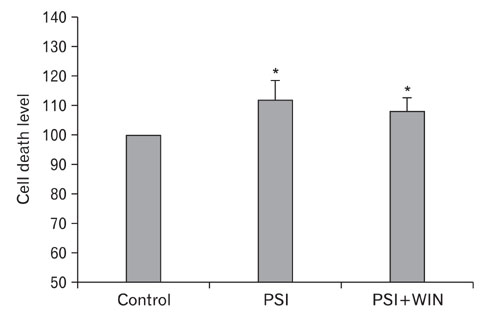
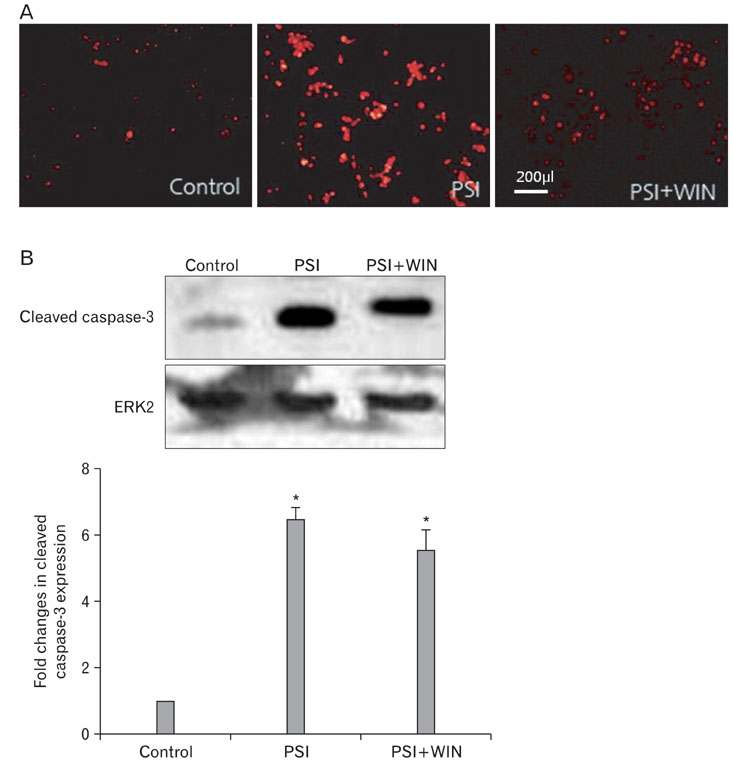
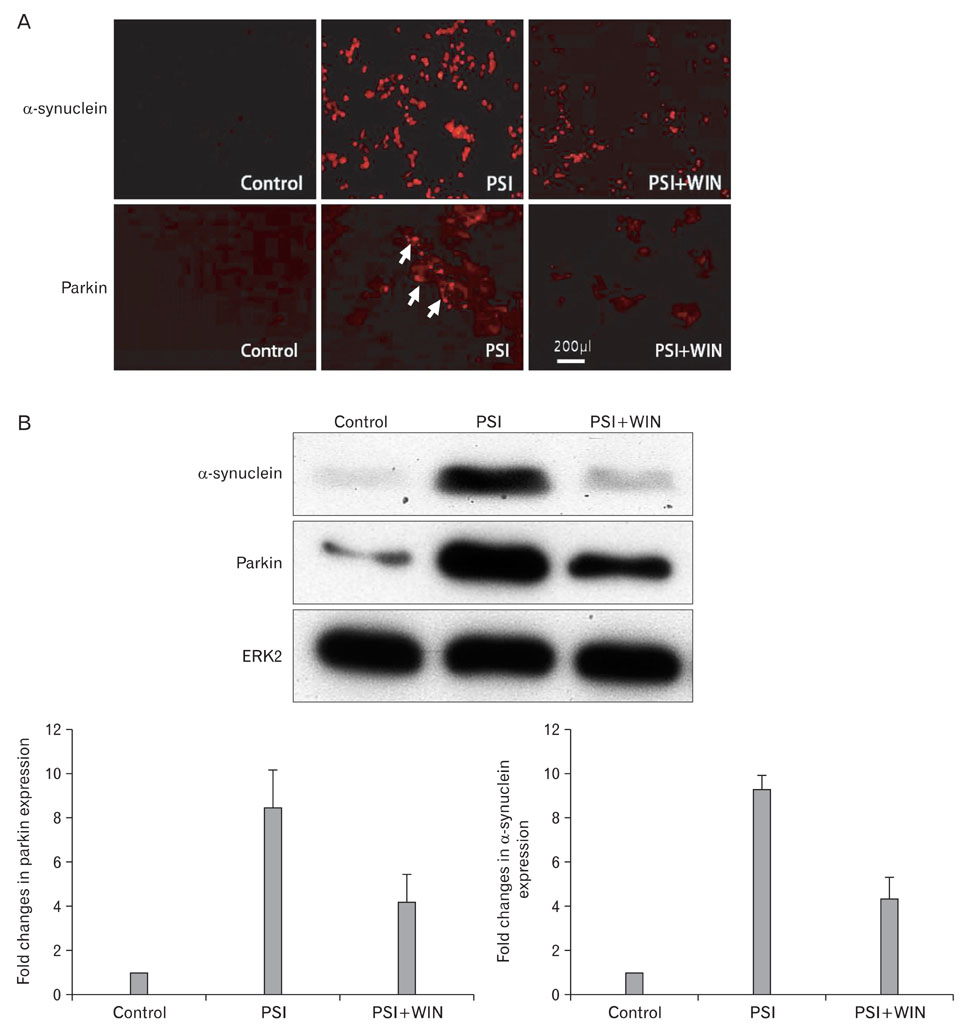
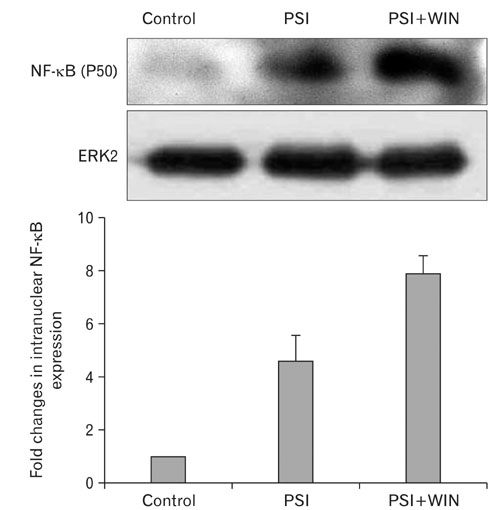
- Cannabinoids have been proposed to possess neuroprotective properties; though their mechanism of action remains contentious, they are posited to prevent neurodegenerative disorders, including Parkinson's disease, the pathogenesis of which has not been established. Recent studies have demonstrated that induction of proteasomal dysfunction in animal models results in a phenotype similar to Parkinson's disease. Here, we investigated the neuroprotective function of a synthetic cannabinoid-receptor agonist (WIN55.212.2) in dopaminergic neuronal death induced by a proteasomal synthase inhibitor (PSI), additionally testing the hypothesis that WIN55.212.2 modulates cytoplasmic accumulation of parkin and alpha-synuclein, a key feature of proteasomal dysfunction in Parkinson's. WIN55.212.2 protects PC12 cells from PSI-induced cytotoxicity, concomitantly inhibiting PSI-induced polyADP ribose polymerase expression and activation of caspase-3. While PSI induces cytoplasmic accumulation of alpha-synuclein and parkin, WIN55.212.2 counters these effects. Interestingly, however, while PSI induces the activation and nuclear translocalization of nuclear factor kappaB, WIN55.212.2 potentiates this effect. These data are suggestive that WIN55.212.2 might confer a neuroprotective benefit in PSI-induced proteasomal dysfunction, and could further protect against neuronal degeneration stemming from cytoplasmic accumulation of alpha-synuclein and parkin. These results indicate that WIN55.212.2 may be a candidate for treatment of neurodegenerative diseases, including Parkinson's disease.
Keyword
MeSH Terms
Figure
Reference
-
1. Ameri A. The effects of cannabinoids on the brain. Prog Neurobiol. 1999. 58:315–348.2. Howlett AC. Efficacy in CB1 receptor-mediated signal transduction. Br J Pharmacol. 2004. 142:1209–1218.3. Mechoulam R, Hanus L, Fride E. Towards cannabinoid drugs: revisited. Prog Med Chem. 1998. 35:199–243.4. Ryberg E, Larsson N, Sjögren S, Hjorth S, Hermansson NO, Leonova J, Elebring T, Nilsson K, Drmota T, Greasley PJ. The orphan receptor GPR55 is a novel cannabinoid receptor. Br J Pharmacol. 2007. 152:1092–1101.5. Baker D, Pryce G, Davies WL, Hiley CR. In silico patent searching reveals a new cannabinoid receptor. Trends Pharmacol Sci. 2006. 27:1–4.6. Gong JP, Onaivi ES, Ishiguro H, Liu QR, Tagliaferro PA, Brusco A, Uhl GR. Cannabinoid CB2 receptors: immunohistochemical localization in rat brain. Brain Res. 2006. 1071:10–23.7. Van Sickle MD, Duncan M, Kingsley PJ, Mouihate A, Urbani P, Mackie K, Stella N, Makriyannis A, Piomelli D, Davison JS, Marnett LJ, Di Marzo V, Pittman QJ, Patel KD, Sharkey KA. Identification and functional characterization of brainstem cannabinoid CB2 receptors. Science. 2005. 310:329–332.8. Howlett AC, Breivogel CS, Childers SR, Deadwyler SA, Hampson RE, Porrino LJ. Cannabinoid physiology and pharmacology: 30 years of progress. Neuropharmacology. 2004. 47:Suppl 1. 345–358.9. Mechoulam R, Spatz M, Shohami E. Endocannabinoids and neuroprotection. Sci STKE. 2002. 2002:re5.10. Nagayama T, Sinor AD, Simon RP, Chen J, Graham SH, Jin K, Greenberg DA. Cannabinoids and neuroprotection in global and focal cerebral ischemia and in neuronal cultures. J Neurosci. 1999. 19:2987–2995.11. Marsicano G, Goodenough S, Monory K, Hermann H, Eder M, Cannich A, Azad SC, Cascio MG, Gutiérrez SO, van der Stelt M, López-Rodriguez ML, Casanova E, Schütz G, Zieglgänsberger W, Di Marzo V, Behl C, Lutz B. CB1 cannabinoid receptors and on-demand defense against excitotoxicity. Science. 2003. 302:84–88.12. Sagredo O, García-Arencibia M, de Lago E, Finetti S, Decio A, Fernández-Ruiz J. Cannabinoids and neuroprotection in basal ganglia disorders. Mol Neurobiol. 2007. 36:82–91.13. Ramírez BG, Blázquez C, Gómez del Pulgar T, Guzmán M, de Ceballos ML. Prevention of Alzheimer's disease pathology by cannabinoids: neuroprotection mediated by blockade of microglial activation. J Neurosci. 2005. 25:1904–1913.14. Pryce G, Giovannoni G, Baker D. Mifepristone or inhibition of 11beta-hydroxylase activity potentiates the sedating effects of the cannabinoid receptor-1 agonist Delta(9)-tetrahydrocannabinol in mice. Neurosci Lett. 2003. 341:164–166.15. Pryce G, Baker D. Control of spasticity in a multiple sclerosis model is mediated by CB1, not CB2, cannabinoid receptors. Br J Pharmacol. 2007. 150:519–525.16. Ehrhart J, Obregon D, Mori T, Hou H, Sun N, Bai Y, Klein T, Fernandez F, Tan J, Shytle RD. Stimulation of cannabinoid receptor 2 (CB2) suppresses microglial activation. J Neuroinflammation. 2005. 2:29.17. McNaught KS, Perl DP, Brownell AL, Olanow CW. Systemic exposure to proteasome inhibitors causes a progressive model of Parkinson's disease. Ann Neurol. 2004. 56:149–162.18. Yang Y, Yu X. Regulation of apoptosis: the ubiquitous way. FASEB J. 2003. 17:790–799.19. Pickart CM. Mechanisms underlying ubiquitination. Annu Rev Biochem. 2001. 70:503–533.20. Colell A, García-Ruiz C, Roman J, Ballesta A, Fernández-Checa JC. Ganglioside GD3 enhances apoptosis by suppressing the nuclear factor-kappa B-dependent survival pathway. FASEB J. 2001. 15:1068–1070.21. McNaught KS, Belizaire R, Isacson O, Jenner P, Olanow CW. Altered proteasomal function in sporadic Parkinson's disease. Exp Neurol. 2003. 179:38–46.22. Li M, Chen D, Shiloh A, Luo J, Nikolaev AY, Qin J, Gu W. Deubiquitination of p53 by HAUSP is an important pathway for p53 stabilization. Nature. 2002. 416:648–653.23. Magnani M, Crinelli R, Bianchi M, Antonelli A. The ubiquitin-dependent proteolytic system and other potential targets for the modulation of nuclear factor-kB (NF-kB). Curr Drug Targets. 2000. 1:387–399.24. Zhang L, Chang M, Li H, Hou S, Zhang Y, Hu Y, Han W, Hu L. Proteomic changes of PC12 cells treated with proteasomal inhibitor PSI. Brain Res. 2007. 1153:196–203.25. Baeuerle PA, Baltimore D. NF-kappa B: ten years after. Cell. 1996. 87:13–20.26. Jüttler E, Potrovita I, Tarabin V, Prinz S, Dong-Si T, Fink G, Schwaninger M. The cannabinoid dexanabinol is an inhibitor of the nuclear factor-kappa B (NF-kappa B). Neuropharmacology. 2004. 47:580–592.27. Braak H, Del Tredici K, Rüb U, de Vos RA, Jansen Steur EN, Braak E. Staging of brain pathology related to sporadic Parkinson's disease. Neurobiol Aging. 2003. 24:197–211.28. Tatton WG, Chalmers-Redman R, Brown D, Tatton N. Apoptosis in Parkinson's disease: signals for neuronal degradation. Ann Neurol. 2003. 53:Suppl 3. S61–S70.29. Marshansky V, Wang X, Bertrand R, Luo H, Duguid W, Chinnadurai G, Kanaan N, Vu MD, Wu J. Proteasomes modulate balance among proapoptotic and antiapoptotic Bcl-2 family members and compromise functioning of the electron transport chain in leukemic cells. J Immunol. 2001. 166:3130–3142.30. Jenner P, Olanow CW. The pathogenesis of cell death in Parkinson's disease. Neurology. 2006. 66:10 Suppl 4. S24–S36.31. Yang SJ, Kim MJ, Jeong HJ, Kim GC, Gil YG, Kim KR, Kim H. Effects of hypoxia on the ubiquitin-proteasome system in primary cortical neuronal cell cultures. Korean J Phys Anthropol. 2008. 21:21–29.32. Chan GC, Hinds TR, Impey S, Storm DR. Hippocampal neurotoxicity of delta9-tetrahydrocannabinol. J Neurosci. 1998. 18:5322–5332.33. Panikashvili D, Simeonidou C, Ben-Shabat S, Hanus L, Breuer A, Mechoulam R, Shohami E. An endogenous cannabinoid (2-AG) is neuroprotective after brain injury. Nature. 2001. 413:527–531.34. van der Stelt M, Veldhuis WB, Bär PR, Veldink GA, Vliegenthart JF, Nicolay K. Neuroprotection by delta9-tetrahydrocannabinol, the main active compound in marijuana, against ouabain-induced in vivo excitotoxicity. J Neurosci. 2001. 21:6475–6479.35. Abood ME, Rizvi G, Sallapudi N, McAllister SD. Activation of the CB1 cannabinoid receptor protects cultured mouse spinal neurons against excitotoxicity. Neurosci Lett. 2001. 309:197–201.36. Jeong HJ, Kim SJ, Moon PD, Kim NH, Kim JS, Park RK, Kim MS, Park BR, Jeong S, Um JY, Kim HM, Hong SH. Antiapoptotic mechanism of cannabinoid receptor 2 agonist on cisplatin-induced apoptosis in the HEI-OC1 auditory cell line. J Neurosci Res. 2007. 85:896–905.37. Downer EJ, Gowran A, Campbell VA. A comparison of the apoptotic effect of delta(9)-tetrahydrocannabinol in the neonatal and adult rat cerebral cortex. Brain Res. 2007. 1175:39–47.38. Parmentier-Batteur S, Jin K, Mao XO, Xie L, Greenberg DA. Increased severity of stroke in CB1 cannabinoid receptor knock-out mice. J Neurosci. 2002. 22:9771–9775.39. Papa SM. The cannabinoid system in Parkinson's disease: multiple targets to motor effects. Exp Neurol. 2008. 211:334–338.40. Geng H, Wittwer T, Dittrich-Breiholz O, Kracht M, Schmitz ML. Phosphorylation of NF-kappaB p65 at Ser468 controls its COMMD1-dependent ubiquitination and target gene-specific proteasomal elimination. EMBO Rep. 2009. 10:381–386.41. Do Y, McKallip RJ, Nagarkatti M, Nagarkatti PS. Activation through cannabinoid receptors 1 and 2 on dendritic cells triggers NF-kappaB-dependent apoptosis: novel role for endogenous and exogenous cannabinoids in immunoregulation. J Immunol. 2004. 173:2373–2382.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- L-PDMP, Ganglioside Synthesis Enhancer Protects Dopaminergic Neurons from Death by Proteasomal Inhibition and the Accumulation of alpha-synuclein
- The Cannabinoid Agonist WIN55,212-2 Suppresses Opioid-induced Pruritus in Mice
- SR144528 as Inverse Agonist of CB2 Cannabinoid Receptor
- Effects of Hypoxia on the Ubiquitin-proteasome System in Primary Cortical Neuronal Cell Cultures
- Asiatic Acid Protects Dopaminergic Neurons from Neuroinflammation by Suppressing Mitochondrial ROS Production