Korean J Physiol Pharmacol.
2020 Jan;24(1):1-10. 10.4196/kjpp.2020.24.1.1.
Autophagy in neutrophils
- Affiliations
-
- 1Department of Physiology, School of Medicine, Kyungpook National University, Daegu 41944, Korea. cwhong@knu.ac.kr
- 2Department of Biochemistry and Cell Biology, School of Medicine, Kyungpook National University, Daegu 41944, Korea. jaemanlee@knu.ac.kr
- KMID: 2466564
- DOI: http://doi.org/10.4196/kjpp.2020.24.1.1
Abstract
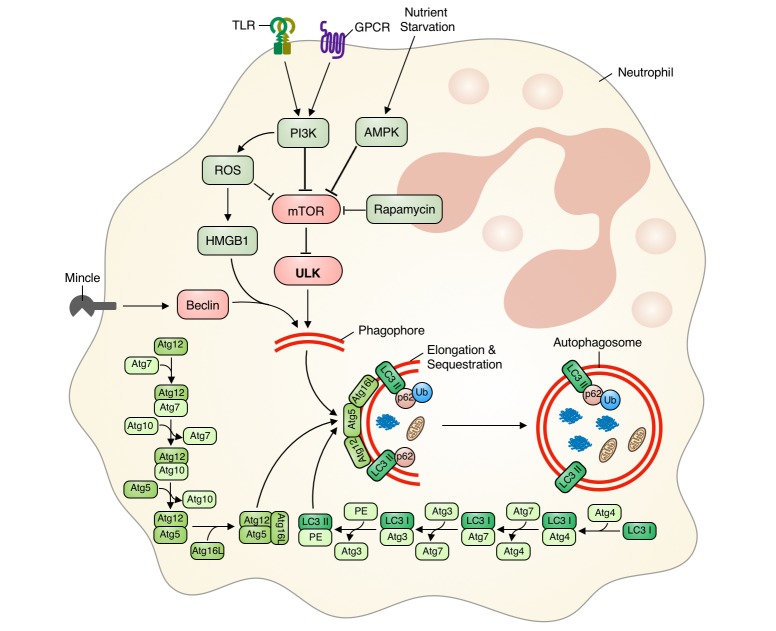
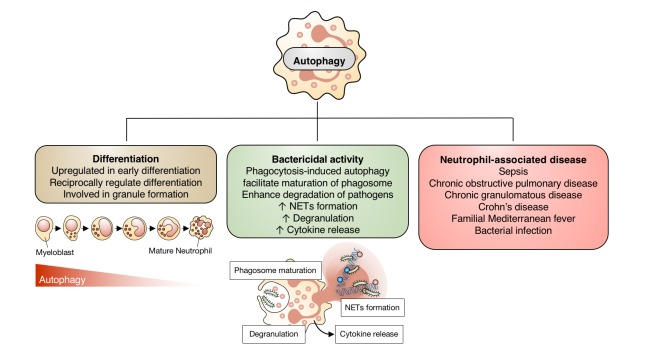
- Autophagy is a highly conserved intracellular degradation and energy-recycling mechanism that contributes to the maintenance of cellular homeostasis. Extensive researches over the past decades have defined the role of autophagy innate immune cells. In this review, we describe the current state of knowledge regarding the role of autophagy in neutrophil biology and a picture of molecular mechanism underlying autophagy in neutrophils. Neutrophils are professional phagocytes that comprise the first line of defense against pathogen. Autophagy machineries are highly conserved in neutrophils. Autophagy is not only involved in generalized function of neutrophils such as differentiation in bone marrow but also plays crucial role effector functions of neutrophils such as granule formation, degranulation, neutrophil extracellular traps release, cytokine production, bactericidal activity and controlling inflammation. This review outlines the current understanding of autophagy in neutrophils and provides insight towards identification of novel therapeutics targeting autophagy in neutrophils.
MeSH Terms
Figure
Reference
-
1. Nathan C. Neutrophils and immunity: challenges and opportunities. Nat Rev Immunol. 2006; 6:173–182. PMID: 16498448.
Article2. Furze RC, Rankin SM. Neutrophil mobilization and clearance in the bone marrow. Immunology. 2008; 125:281–288. PMID: 19128361.
Article3. Hampton MB, Kettle AJ, Winterbourn CC. Inside the neutrophil phagosome: oxidants, myeloperoxidase, and bacterial killing. Blood. 1998; 92:3007–3017. PMID: 9787133.
Article4. Segal AW. How neutrophils kill microbes. Annu Rev Immunol. 2005; 23:197–223. PMID: 15771570.
Article5. Brinkmann V, Reichard U, Goosmann C, Fauler B, Uhlemann Y, Weiss DS, Weinrauch Y, Zychlinsky A. Neutrophil extracellular traps kill bacteria. Science. 2004; 303:1532–1535. PMID: 15001782.
Article6. Naegele M, Tillack K, Reinhardt S, Schippling S, Martin R, Sospedra M. Neutrophils in multiple sclerosis are characterized by a primed phenotype. J Neuroimmunol. 2012; 242:60–71. PMID: 22169406.
Article7. Borregaard N. Neutrophils, from marrow to microbes. Immunity. 2010; 33:657–670. PMID: 21094463.
Article8. Ryter SW, Cloonan SM, Choi AM. Autophagy: a critical regulator of cellular metabolism and homeostasis. Mol Cells. 2013; 36:7–16. PMID: 23708729.
Article9. Nezis IP, Vaccaro MI, Devenish RJ, Juhász G. Autophagy in development, cell differentiation, and homeodynamics: from molecular mechanisms to diseases and pathophysiology. Biomed Res Int. 2014; 2014:349623. PMID: 25247173.
Article10. Mitroulis I, Kourtzelis I, Kambas K, Rafail S, Chrysanthopoulou A, Speletas M, Ritis K. Regulation of the autophagic machinery in human neutrophils. Eur J Immunol. 2010; 40:1461–1472. PMID: 20162553.
Article11. Yang Z, Klionsky DJ. An overview of the molecular mechanism of autophagy. Curr Top Microbiol Immunol. 2009; 335:1–32. PMID: 19802558.
Article12. Das G, Shravage BV, Baehrecke EH. Regulation and function of autophagy during cell survival and cell death. Cold Spring Harb Perspect Biol. 2012; 4:a008813. PMID: 22661635.
Article13. Cuervo AM. Autophagy: many paths to the same end. Mol Cell Biochem. 2004; 263:55–72.
Article14. Wang CW, Klionsky DJ. The molecular mechanism of autophagy. Mol Med. 2003; 9:65–76. PMID: 12865942.
Article15. Mizushima N. Autophagy: process and function. Genes Dev. 2007; 21:2861–2873. PMID: 18006683.
Article16. Itakura A, McCarty OJ. Pivotal role for the mTOR pathway in the formation of neutrophil extracellular traps via regulation of autophagy. Am J Physiol Cell Physiol. 2013; 305:C348–C354. PMID: 23720022.
Article17. Parzych KR, Klionsky DJ. An overview of autophagy: morphology, mechanism, and regulation. Antioxid Redox Signal. 2014; 20:460–473. PMID: 23725295.
Article18. Backer JM. The intricate regulation and complex functions of the Class III phosphoinositide 3-kinase Vps34. Biochem J. 2016; 473:2251–2271. PMID: 27470591.
Article19. Mizushima N, Noda T, Yoshimori T, Tanaka Y, Ishii T, George MD, Klionsky DJ, Ohsumi M, Ohsumi Y. A protein conjugation system essential for autophagy. Nature. 1998; 395:395–398. PMID: 9759731.
Article20. Glick D, Barth S, Macleod KF. Autophagy: cellular and molecular mechanisms. J Pathol. 2010; 221:3–12. PMID: 20225336.
Article21. Ichimura Y, Kirisako T, Takao T, Satomi Y, Shimonishi Y, Ishihara N, Mizushima N, Tanida I, Kominami E, Ohsumi M, Noda T, Ohsumi Y. A ubiquitin-like system mediates protein lipidation. Nature. 2000; 408:488–492. PMID: 11100732.
Article22. Codogno P, Mehrpour M, Proikas-Cezanne T. Canonical and non-canonical autophagy: variations on a common theme of self-eating? Nat Rev Mol Cell Biol. 2011; 13:7–12. PMID: 22166994.
Article23. Chargui A, Cesaro A, Mimouna S, Fareh M, Brest P, Naquet P, Darfeuille-Michaud A, Hébuterne X, Mograbi B, Vouret-Craviari V, Hofman P. Subversion of autophagy in adherent invasive Escherichia coli-infected neutrophils induces inflammation and cell death. PLoS One. 2012; 7:e51727. PMID: 23272151.
Article24. Goldmann O, Medina E. The expanding world of extracellular traps: not only neutrophils but much more. Front Immunol. 2013; 3:420. PMID: 23335924.
Article25. Mihalache CC, Yousefi S, Conus S, Villiger PM, Schneider EM, Simon HU. Inflammation-associated autophagy-related programmed necrotic death of human neutrophils characterized by organelle fusion events. J Immunol. 2011; 186:6532–6542. PMID: 21515790.
Article26. Huang J, Canadien V, Lam GY, Steinberg BE, Dinauer MC, Magalhaes MA, Glogauer M, Grinstein S, Brumell JH. Activation of antibacterial autophagy by NADPH oxidases. Proc Natl Acad Sci U S A. 2009; 106:6226–6231. PMID: 19339495.
Article27. Lv XX, Liu SS, Li K, Cui B, Liu C, Hu ZW. Cigarette smoke promotes COPD by activating platelet-activating factor receptor and inducing neutrophil autophagic death in mice. Oncotarget. 2017; 8:74720–74735. PMID: 29088819.
Article28. Sharma A, Simonson TJ, Jondle CN, Mishra BB, Sharma J. Mincle-mediated neutrophil extracellular trap formation by regulation of autophagy. J Infect Dis. 2017; 215:1040–1048. PMID: 28186242.
Article29. Zhao M, Klionsky DJ. AMPK-dependent phosphorylation of ULK1 induces autophagy. Cell Metab. 2011; 13:119–120. PMID: 21284977.
Article30. Xu F, Zhang C, Zou Z, Fan EKY, Chen L, Li Y, Billiar TR, Wilson MA, Shi X, Fan J. Aging-related Atg5 defect impairs neutrophil extracellular traps formation. Immunology. 2017; 151:417–432. PMID: 28375544.
Article31. Bhattacharya A, Wei Q, Shin JN, Abdel Fattah E, Bonilla DL, Xiang Q, Eissa NT. Autophagy Is required for neutrophil-mediated inflammation. Cell Rep. 2015; 12:1731–1739. PMID: 26344765.
Article32. Park SY, Shrestha S, Youn YJ, Kim JK, Kim SY, Kim HJ, Park SH, Ahn WG, Kim S, Lee MG, Jung KS, Park YB, Mo EK, Ko Y, Lee SY, Koh Y, Park MJ, Song DK, Hong CW. Autophagy primes neutrophils for neutrophil extracellular trap formation during sepsis. Am J Respir Crit Care Med. 2017; 196:577–589. PMID: 28358992.
Article33. Klionsky DJ, Abdelmohsen K, Abe A, Abedin MJ, Abeliovich H, Acevedo Arozena A, Adachi H, Adams CM, Adams PD, Adeli K, Adhihetty PJ, Adler SG, Agam G, Agarwal R, Aghi MK, Agnello M, Agostinis P, Aguilar PV, Aguirre-Ghiso J, Airoldi EM, et al. Guidelines for the use and interpretation of assays for monitoring autophagy (3rd edition). Autophagy. 2016; 12:1–222. PMID: 26799652.34. Rožman S, Yousefi S, Oberson K, Kaufmann T, Benarafa C, Simon HU. The generation of neutrophils in the bone marrow is controlled by autophagy. Cell Death Differ. 2015; 22:445–456. PMID: 25323583.
Article35. Kimmey JM, Huynh JP, Weiss LA, Park S, Kambal A, Debnath J, Virgin HW, Stallings CL. Unique role for ATG5 in neutrophil-mediated immunopathology during M. tuberculosis infection. Nature. 2015; 528:565–569. PMID: 26649827.
Article36. Theilgaard-Mönch K, Jacobsen LC, Borup R, Rasmussen T, Bjerregaard MD, Nielsen FC, Cowland JB, Borregaard N. The transcriptional program of terminal granulocytic differentiation. Blood. 2005; 105:1785–1796. PMID: 15514007.37. Akashi K, He X, Chen J, Iwasaki H, Niu C, Steenhard B, Zhang J, Haug J, Li L. Transcriptional accessibility for genes of multiple tissues and hematopoietic lineages is hierarchically controlled during early hematopoiesis. Blood. 2003; 101:383–389. PMID: 12393558.
Article38. Huang Y, Tan P, Wang X, Yi Y, Hu Y, Wang D, Wang F. Transcriptomic insights into temporal expression pattern of autophagy genes during monocytic and granulocytic differentiation. Autophagy. 2018; 14:558–559. PMID: 29313406.
Article39. Koschade SE, Brandts CH. Selective autophagy in normal and malignant hematopoiesis. J Mol Biol. 2019; DOI: 10.1016/j.jmb.2019.06.025. [Epub ahead of print].
Article40. Orsini M, Chateauvieux S, Rhim J, Gaigneaux A, Cheillan D, Christov C, Dicato M, Morceau F, Diederich M. Sphingolipid-mediated inflammatory signaling leading to autophagy inhibition converts erythropoiesis to myelopoiesis in human hematopoietic stem/progenitor cells. Cell Death Differ. 2019; 26:1796–1812. PMID: 30546074.
Article41. da Silva FM, Massart-Leën AM, Burvenich C. Development and maturation of neutrophils. Vet Q. 1994; 16:220–225. PMID: 7740747.
Article42. Riffelmacher T, Clarke A, Richter FC, Stranks A, Pandey S, Danielli S, Hublitz P, Yu Z, Johnson E, Schwerd T, McCullagh J, Uhlig H, Jacobsen SEW, Simon AK. Autophagy-dependent generation of free fatty acids is critical for normal neutrophil differentiation. Immunity. 2017; 47:466–480.e5. PMID: 28916263.
Article43. Kantari C, Pederzoli-Ribeil M, Witko-Sarsat V. The role of neutrophils and monocytes in innate immunity. Contrib Microbiol. 2008; 15:118–146. PMID: 18511859.
Article44. Wong CO, Gregory S, Hu H, Chao Y, Sepúlveda VE, He Y, Li-Kroeger D, Goldman WE, Bellen HJ, Venkatachalam K. Lysosomal degradation is required for sustained phagocytosis of bacteria by macrophages. Cell Host Microbe. 2017; 21:719–730.e6. PMID: 28579255.
Article45. Lee WL, Harrison RE, Grinstein S. Phagocytosis by neutrophils. Microbes Infect. 2003; 5:1299–1306. PMID: 14613773.
Article46. Chargui A, El May MV. Autophagy mediates neutrophil responses to bacterial infection. APMIS. 2014; 122:1047–1058. PMID: 24735202.
Article47. Gong L, Cullinane M, Treerat P, Ramm G, Prescott M, Adler B, Boyce JD, Devenish RJ. The Burkholderia pseudomallei type III secretion system and BopA are required for evasion of LC3-associated phagocytosis. PLoS One. 2011; 6:e17852. PMID: 21412437.
Article48. Ramachandran G, Gade P, Tsai P, Lu W, Kalvakolanu DV, Rosen GM, Cross AS. Potential role of autophagy in the bactericidal activity of human PMNs for Bacillus anthracis. Pathog Dis. 2015; 73:ftv080. PMID: 26424808.49. Itoh H, Matsuo H, Kitamura N, Yamamoto S, Higuchi T, Takematsu H, Kamikubo Y, Kondo T, Yamashita K, Sasada M, Takaori-Kondo A, Adachi S. Enhancement of neutrophil autophagy by an IVIG preparation against multidrug-resistant bacteria as well as drug-sensitive strains. J Leukoc Biol. 2015; 98:107–117. PMID: 25908735.
Article50. Ullah I, Ritchie ND, Evans TJ. The interrelationship between phagocytosis, autophagy and formation of neutrophil extracellular traps following infection of human neutrophils by Streptococcus pneumoniae. Innate Immun. 2017; 23:413–423. PMID: 28399692.
Article51. Wang C, Mendonsa GR, Symington JW, Zhang Q, Cadwell K, Virgin HW, Mysorekar IU. Atg16L1 deficiency confers protection from uropathogenic Escherichia coli infection in vivo. Proc Natl Acad Sci U S A. 2012; 109:11008–11013. PMID: 22715292.
Article52. Rinchai D, Riyapa D, Buddhisa S, Utispan K, Titball RW, Stevens MP, Stevens JM, Ogawa M, Tanida I, Koike M, Uchiyama Y, Ato M, Lertmemongkolchai G. Macroautophagy is essential for killing of intracellular Burkholderia pseudomallei in human neutrophils. Autophagy. 2015; 11:748–755. PMID: 25996656.53. Remijsen Q, Kuijpers TW, Wirawan E, Lippens S, Vandenabeele P, Vanden Berghe T. Dying for a cause: NETosis, mechanisms behind an antimicrobial cell death modality. Cell Death Differ. 2011; 18:581–588. PMID: 21293492.
Article54. Urban CF, Ermert D, Schmid M, Abu-Abed U, Goosmann C, Nacken W, Brinkmann V, Jungblut PR, Zychlinsky A. Neutrophil extracellular traps contain calprotectin, a cytosolic protein complex involved in host defense against Candida albicans. PLoS Pathog. 2009; 5:e1000639. PMID: 19876394.
Article55. Boe DM, Curtis BJ, Chen MM, Ippolito JA, Kovacs EJ. Extracellular traps and macrophages: new roles for the versatile phagocyte. J Leukoc Biol. 2015; 97:1023–1035. PMID: 25877927.
Article56. Kenny EF, Herzig A, Krüger R, Muth A, Mondal S, Thompson PR, Brinkmann V, Bernuth HV, Zychlinsky A. Diverse stimuli engage different neutrophil extracellular trap pathways. Elife. 2017; 6:e24437. PMID: 28574339.
Article57. Kaplan MJ, Radic M. Neutrophil extracellular traps: double-edged swords of innate immunity. J Immunol. 2012; 189:2689–2695. PMID: 22956760.
Article58. Peng HH, Liu YJ, Ojcius DM, Lee CM, Chen RH, Huang PR, Martel J, Young JD. Mineral particles stimulate innate immunity through neutrophil extracellular traps containing HMGB1. Sci Rep. 2017; 7:16628. PMID: 29192209.
Article59. Neumann A, Papareddy P, Westman J, Hyldegaard O, Snäll J, Norrby-Teglund A, Herwald H. Immunoregulation of neutrophil extracellular trap formation by endothelial-derived p33 (gC1q receptor). J Innate Immun. 2018; 10:30–43. PMID: 29035880.
Article60. van der Linden M, Westerlaken GHA, van der Vlist M, van Montfrans J, Meyaard L. Differential signalling and kinetics of neutrophil extracellular trap release revealed by quantitative live imaging. Sci Rep. 2017; 7:6529. PMID: 28747804.
Article61. Maugeri N, Campana L, Gavina M, Covino C, De Metrio M, Panciroli C, Maiuri L, Maseri A, D'Angelo A, Bianchi ME, Rovere-Querini P, Manfredi AA. Activated platelets present high mobility group box 1 to neutrophils, inducing autophagy and promoting the extrusion of neutrophil extracellular traps. J Thromb Haemost. 2014; 12:2074–2088. PMID: 25163512.
Article62. Chatfield SM, Grebe K, Whitehead LW, Rogers KL, Nebl T, Murphy JM, Wicks IP. Monosodium urate crystals generate nuclease-resistant neutrophil extracellular traps via a distinct molecular pathway. J Immunol. 2018; 200:1802–1816. PMID: 29367211.
Article63. Remijsen Q, Vanden Berghe T, Wirawan E, Asselbergh B, Parthoens E, De Rycke R, Noppen S, Delforge M, Willems J, Vandenabeele P. Neutrophil extracellular trap cell death requires both autophagy and superoxide generation. Cell Res. 2011; 21:290–304. PMID: 21060338.
Article64. Douda DN, Khan MA, Grasemann H, Palaniyar N. SK3 channel and mitochondrial ROS mediate NADPH oxidase-independent NETosis induced by calcium influx. Proc Natl Acad Sci U S A. 2015; 112:2817–2822. PMID: 25730848.
Article65. Pilsczek FH, Salina D, Poon KK, Fahey C, Yipp BG, Sibley CD, Robbins SM, Green FH, Surette MG, Sugai M, Bowden MG, Hussain M, Zhang K, Kubes P. A novel mechanism of rapid nuclear neutrophil extracellular trap formation in response to Staphylococcus aureus. J Immunol. 2010; 185:7413–7425. PMID: 21098229.66. Chow OA, von Köckritz-Blickwede M, Bright AT, Hensler ME, Zinkernagel AS, Cogen AL, Gallo RL, Monestier M, Wang Y, Glass CK, Nizet V. Statins enhance formation of phagocyte extracellular traps. Cell Host Microbe. 2010; 8:445–454. PMID: 21075355.
Article67. Byrd AS, O'Brien XM, Johnson CM, Lavigne LM, Reichner JS. An extracellular matrix-based mechanism of rapid neutrophil extracellular trap formation in response to Candida albicans. J Immunol. 2013; 190:4136–4148. PMID: 23509360.68. Stranks AJ, Hansen AL, Panse I, Mortensen M, Ferguson DJ, Puleston DJ, Shenderov K, Watson AS, Veldhoen M, Phadwal K, Cerundolo V, Simon AK. Autophagy controls acquisition of aging features in macrophages. J Innate Immun. 2015; 7:375–391. PMID: 25764971.
Article69. Ma R, Li T, Cao M, Si Y, Wu X, Zhao L, Yao Z, Zhang Y, Fang S, Deng R, Novakovic VA, Bi Y, Kou J, Yu B, Yang S, Wang J, Zhou J, Shi J. Extracellular DNA traps released by acute promyelocytic leukemia cells through autophagy. Cell Death Dis. 2016; 7:e2283. PMID: 27362801.
Article70. Germic N, Stojkov D, Oberson K, Yousefi S, Simon HU. Neither eosinophils nor neutrophils require ATG5-dependent autophagy for extracellular DNA trap formation. Immunology. 2017; 152:517–525. PMID: 28703297.
Article71. Rørvig S, Østergaard O, Heegaard NH, Borregaard N. Proteome profiling of human neutrophil granule subsets, secretory vesicles, and cell membrane: correlation with transcriptome profiling of neutrophil precursors. J Leukoc Biol. 2013; 94:711–721. PMID: 23650620.
Article72. Masson PL, Heremans JF, Schonne E. Lactoferrin, an iron-binding protein in neutrophilic leukocytes. J Exp Med. 1969; 130:643–658. PMID: 4979954.73. Faurschou M, Borregaard N. Neutrophil granules and secretory vesicles ininflammation. Microbes Infect. 2003; 5:1317–1327. PMID: 14613775.74. Patel JM, Sapey E, Parekh D, Scott A, Dosanjh D, Gao F, Thickett DR. Sepsis Induces a Dysregulated neutrophil phenotype that is associated with increased mortality. Mediators Inflamm. 2018; 2018:4065362. PMID: 29849488.
Article75. Meng W, Paunel-Görgülü A, Flohé S, Hoffmann A, Witte I, MacKenzie C, Baldus SE, Windolf J, Lögters TT. Depletion of neutrophil extracellular traps in vivo results in hypersusceptibility to polymicrobial sepsis in mice. Crit Care. 2012; 16:R137. PMID: 22835277.
Article76. Konstantinidis T, Kambas K, Mitsios A, Panopoulou M, Tsironidou V, Dellaporta E, Kouklakis G, Arampatzioglou A, Angelidou I, Mitroulis I, Skendros P, Ritis K. Immunomodulatory role of clarithromycin in acinetobacter baumannii infection via formation of neutrophil extracellular traps. Antimicrob Agents Chemother. 2015; 60:1040–1048. PMID: 26643338.
Article77. Sharma A, Steichen AL, Jondle CN, Mishra BB, Sharma J. Protective role of Mincle in bacterial pneumonia by regulation of neutrophil mediated phagocytosis and extracellular trap formation. J Infect Dis. 2014; 209:1837–1846. PMID: 24353272.
Article78. Li XF, Chen DP, Ouyang FZ, Chen MM, Wu Y, Kuang DM, Zheng L. Increased autophagy sustains the survival and pro-tumourigenic effects of neutrophils in human hepatocellular carcinoma. J Hepatol. 2015; 62:131–139. PMID: 25152203.
Article79. Park J, Wysocki RW, Amoozgar Z, Maiorino L, Fein MR, Jorns J, Schott AF, Kinugasa-Katayama Y, Lee Y, Won NH, Nakasone ES, Hearn SA, Küttner V, Qiu J, Almeida AS, Perurena N, Kessenbrock K, Goldberg MS, Egeblad M. Cancer cells induce metastasis-supporting neutrophil extracellular DNA traps. Sci Transl Med. 2016; 8:361ra138.
Article80. Tohme S, Yazdani HO, Al-Khafaji AB, Chidi AP, Loughran P, Mowen K, Wang Y, Simmons RL, Huang H, Tsung A. Neutrophil extracellular traps promote the development and progression of liver metastases after surgical stress. Cancer Res. 2016; 76:1367–1380. PMID: 26759232.
Article81. Najmeh S, Cools-Lartigue J, Rayes RF, Gowing S, Vourtzoumis P, Bourdeau F, Giannias B, Berube J, Rousseau S, Ferri LE, Spicer JD. Neutrophil extracellular traps sequester circulating tumor cells via β1-integrin mediated interactions. Int J Cancer. 2017; 140:2321–2330. PMID: 28177522.
Article82. Leach J, Morton JP, Sansom OJ. Neutrophils: Homing in on the myeloid mechanisms of metastasis. Mol Immunol. 2019; 110:69–76. PMID: 29269005.
Article83. Borregaard N, Cowland JB. Granules of the human neutrophilic polymorphonuclear leukocyte. Blood. 1997; 89:3503–3521. PMID: 9160655.
Article84. Cowland JB, Borregaard N. Granulopoiesis and granules of human neutrophils. Immunol Rev. 2016; 273:11–28. PMID: 27558325.
Article85. Lawrence SM, Corriden R, Nizet V. The ontogeny of a neutrophil: mechanisms of granulopoiesis and homeostasis. Microbiol Mol Biol Rev. 2018; 82:e00057–e00017. PMID: 29436479.
Article86. Gullberg U, Andersson E, Garwicz D, Lindmark A, Olsson I. Biosynthesis, processing and sorting of neutrophil proteins: insight into neutrophil granule development. Eur J Haematol. 1997; 58:137–153. PMID: 9150707.
Article87. Lacy P. Mechanisms of degranulation in neutrophils. Allergy Asthma Clin Immunol. 2006; 2:98–108. PMID: 20525154.
Article88. Mantovani A, Cassatella MA, Costantini C, Jaillon S. Neutrophils in the activation and regulation of innate and adaptive immunity. Nat Rev Immunol. 2011; 11:519–531. PMID: 21785456.
Article89. Tecchio C, Cassatella MA. Neutrophil-derived cytokines involved in physiological and pathological angiogenesis. Chem Immunol Allergy. 2014; 99:123–137. PMID: 24217606.
Article90. Tecchio C, Micheletti A, Cassatella MA. Neutrophil-derived cytokines: facts beyond expression. Front Immunol. 2014; 5:508. PMID: 25374568.
Article91. Naegelen I, Beaume N, Plançon S, Schenten V, Tschirhart EJ, Bréchard S. Regulation of neutrophil degranulation and cytokine secretion: a novel model approach based on linear fitting. J Immunol Res. 2015; 2015:817038. PMID: 26579547.
Article92. Angelidou I, Chrysanthopoulou A, Mitsios A, Arelaki S, Arampatzioglou A, Kambas K, Ritis D, Tsironidou V, Moschos I, Dalla V, Stakos D, Kouklakis G, Mitroulis I, Ritis K, Skendros P. REDD1/ autophagy pathway is associated with Neutrophil-Driven IL-1β inflammatory response in active ulcerative colitis. J Immunol. 2018; 200:3950–3961. PMID: 29712770.93. Apostolidou E, Skendros P, Kambas K, Mitroulis I, Konstantinidis T, Chrysanthopoulou A, Nakos K, Tsironidou V, Koffa M, Boumpas DT, Ritis K. Neutrophil extracellular traps regulate IL-1β-mediated inflammation in familial Mediterranean fever. Ann Rheum Dis. 2016; 75:269–277. PMID: 25261578.
Article94. Iula L, Keitelman IA, Sabbione F, Fuentes F, Guzman M, Galletti JG, Gerber PP, Ostrowski M, Geffner JR, Jancic CC, Trevani AS. Autophagy mediates interleukin-1β secretion in human neutrophils. Front Immunol. 2018; 9:269. PMID: 29515581.
Article96. Harris J, Hartman M, Roche C, Zeng SG, O'Shea A, Sharp FA, Lambe EM, Creagh EM, Golenbock DT, Tschopp J, Kornfeld H, Fitzgerald KA, Lavelle EC. Autophagy controls IL-1beta secretion by targeting pro-IL-1beta for degradation. J Biol Chem. 2011; 286:9587–9597. PMID: 21228274.97. Netea-Maier RT, Plantinga TS, van de Veerdonk FL, Smit JW, Netea MG. Modulation of inflammation by autophagy: consequences for human disease. Autophagy. 2016; 12:245–260. PMID: 26222012.
Article98. Levine B, Mizushima N, Virgin HW. Autophagy in immunity and inflammation. Nature. 2011; 469:323–335. PMID: 21248839.
Article99. Zhang F, Liu AL, Gao S, Ma S, Guo SB. Neutrophil dysfunction in sepsis. Chin Med J (Engl). 2016; 129:2741–2744. PMID: 27824008.
Article100. Segal AW, Loewi G. Neutrophil dysfunction in Crohn's disease. Lancet. 1976; 2:219–221. PMID: 59239.
Article101. Davis WC, Douglas SD, Fudenberg HH. A selective neutrophil dysfunction syndrome: impaired killing of staphylococci. Ann Intern Med. 1968; 69:1237–1243. PMID: 5725737.
Article102. Jin L, Batra S, Jeyaseelan S. Deletion of Nlrp3 augments survival during polymicrobial sepsis by decreasing autophagy and enhancing phagocytosis. J Immunol. 2017; 198:1253–1262. PMID: 28031338.103. Haimovici A, Brigger D, Torbett BE, Fey MF, Tschan MP. Induction of the autophagy-associated gene MAP1S via PU.1 supports APL differentiation. Leuk Res. 2014; 38:1041–1047. PMID: 25043887.
Article104. Schläfli AM, Shan-Krauer D, Garattini E, Tschan MP. Regulation and function of autophagy in retinoic acid mediated therapy of myeloid leukemia and breast cancer. Ital J Anat Embryol. 2015; 120:9–10.105. Britschgi A, Yousefi S, Laedrach J, Shan D, Simon H-U, Tobler A, Fey MF, Tschan MP. Blocking the autophagy gene 5 (ATG5) impairs ATRA-induced myeloid differentiation, and ATG5 is downregulated in AML. Blood. 2008; 112:309.
Article106. Abdel Fattah E, Bhattacharya A, Herron A, Safdar Z, Eissa NT. Critical role for IL-18 in spontaneous lung inflammation caused by autophagy deficiency. J Immunol. 2015; 194:5407–5416. PMID: 25888640.
Article107. Skendros P, Chrysanthopoulou A, Rousset F, Kambas K, Arampatzioglou A, Mitsios A, Bocly V, Konstantinidis T, Pellet P, Angelidou I, Apostolidou E, Ritis D, Tsironidou V, Galtsidis S, Papagoras C, Stakos D, Kouklakis G, Dalla V, Koffa M, Mitroulis I, et al. Regulated in development and DNA damage responses 1 (REDD1) links stress with IL-1β-mediated familial Mediterranean fever attack through autophagy-driven neutrophil extracellular traps. J Allergy Clin Immunol. 2017; 140:1378–1387.e13. PMID: 28342915.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Neuronal Autophagy: Characteristic Features and Roles in Neuronal Pathophysiology
- Autophagy in the placenta
- Functional interplay between non-canonical inflammasomes and autophagy in inflammatory responses and diseases
- The role of autophagy in cell proliferation and differentiation during tooth development
- Herpesviral Interaction with Autophagy