Int J Stem Cells.
2019 Nov;12(3):474-483. 10.15283/ijsc19075.
Direct Reprogramming to Human Induced Neuronal Progenitors from Fibroblasts of Familial and Sporadic Parkinson’s Disease Patients
- Affiliations
-
- 1Stem Cell Convergence Research Center, Korea Research Institute of Bioscience and Biotechnology (KRIBB), Daejeon, Korea. janghwan.kim@kribb.re.kr, myson@kribb.re.kr
- 2Department of Functional Genomics, KRIBB, School of Bioscience, University of Science and Technology, Daejeon, Korea.
- KMID: 2465888
- DOI: http://doi.org/10.15283/ijsc19075
Abstract
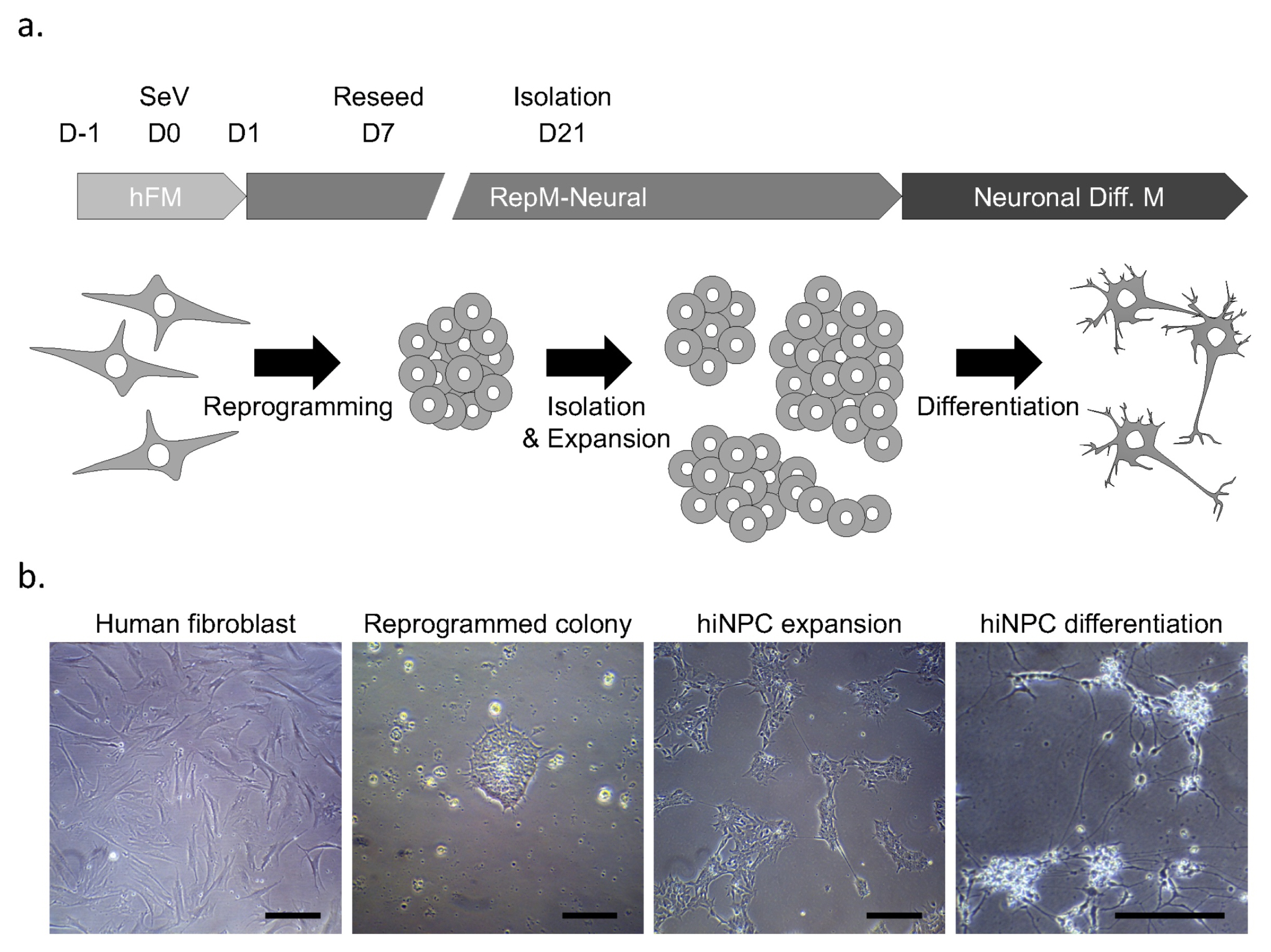
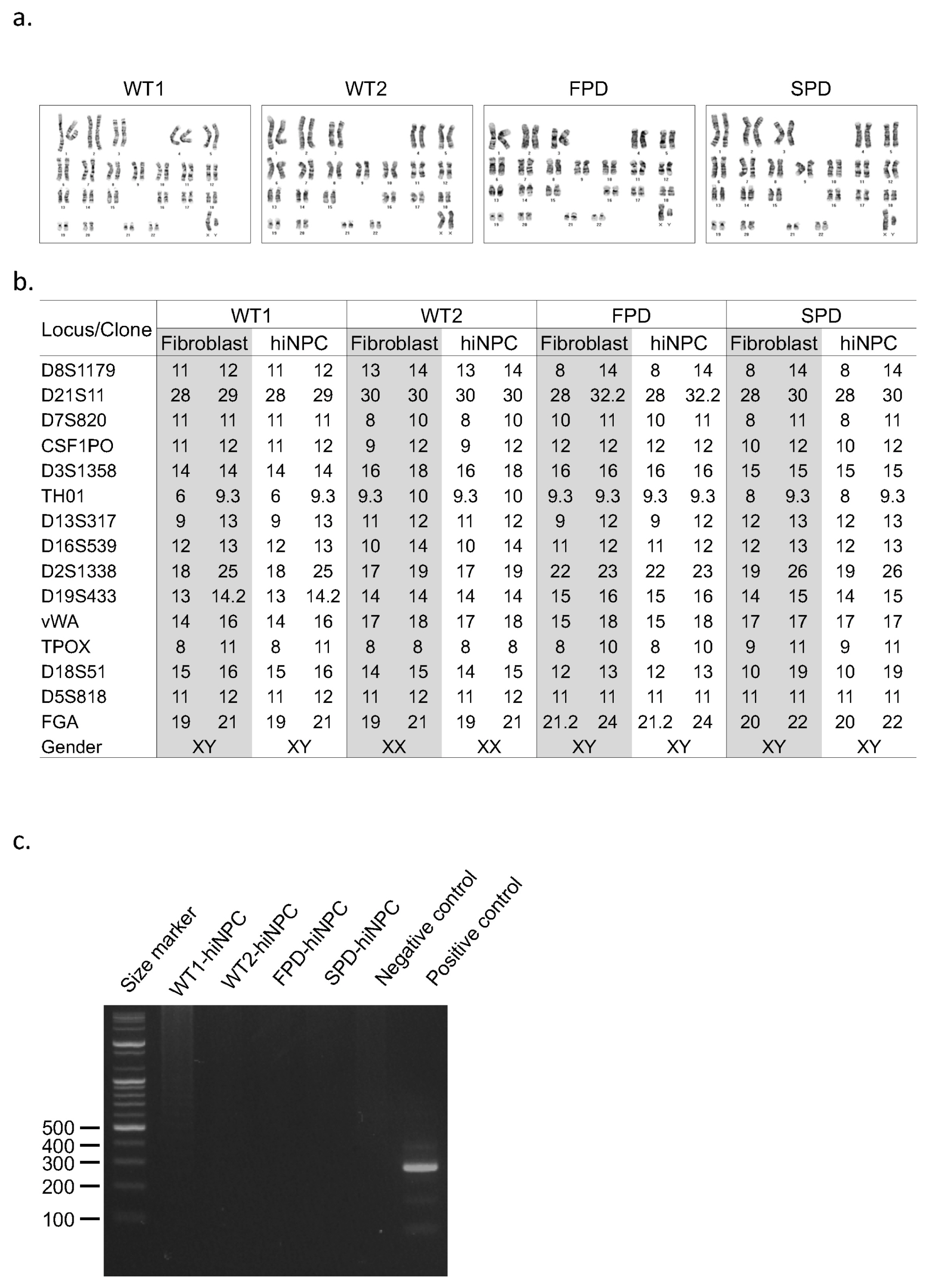
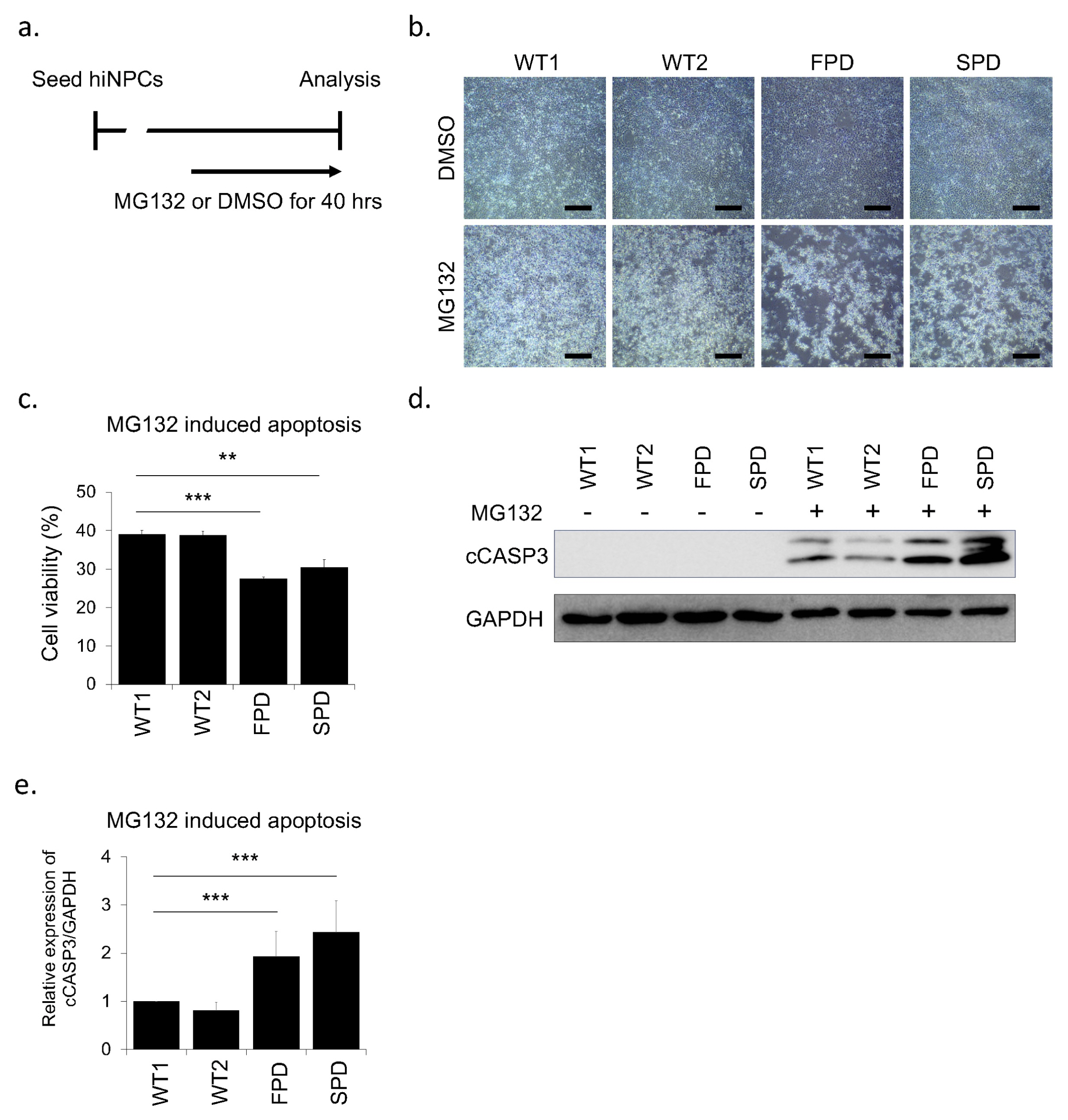
- In Parkinson's disease (PD) research, human neuroblastoma and immortalized neural cell lines have been widely used as in vitro models. The advancement in the field of reprogramming technology has provided tools for generating patient-specific induced pluripotent stem cells (hiPSCs) as well as human induced neuronal progenitor cells (hiNPCs). These cells have revolutionized the field of disease modeling, especially in neural diseases. Although the direct reprogramming to hiNPCs has several advantages over differentiation after hiPSC reprogramming, such as the time required and the simple procedure, relatively few studies have utilized hiNPCs. Here, we optimized the protocol for hiNPC reprogramming using pluripotency factors and Sendai virus. In addition, we generated hiNPCs of two healthy donors, a sporadic PD patient, and a familial patient with the LRRK2 G2019S mutation (L2GS). The four hiNPC cell lines are highly proliferative, expressed NPC markers, maintained the normal karyotype, and have the differentiation potential of dopaminergic neurons. Importantly, the patient hiNPCs show different apoptotic marker expression. Thus, these hiNPCs, in addition to hiPSCs, are a favorable option to study PD pathology.
Keyword
MeSH Terms
Figure
Reference
-
References
1. Falkenburger BH, Saridaki T, Dinter E. Cellular models for Parkinson’s disease. J Neurochem. 2016; 139(Suppl 1):121–130. DOI: 10.1111/jnc.13618. PMID: 27091001.
Article2. Lotharius J, Falsig J, van Beek J, Payne S, Dringen R, Brundin P, Leist M. Progressive degeneration of human mesencephalic neuron-derived cells triggered by dopamine-dependent oxidative stress is dependent on the mixed-lineage kinase pathway. J Neurosci. 2005; 25:6329–6342. DOI: 10.1523/JNEUROSCI.1746-05.2005. PMID: 16000623. PMCID: PMC6725277.
Article3. Jang W, Kim HJ, Li H, Jo KD, Lee MK, Yang HO. The neuroprotective effect of erythropoietin on rotenone-induced neurotoxicity in SH-SY5Y cells through the induction of autophagy. Mol Neurobiol. 2016; 53:3812–3821. DOI: 10.1007/s12035-015-9316-x. PMID: 26156288.
Article4. Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007; 131:861–872. DOI: 10.1016/j.cell.2007.11.019. PMID: 18035408.
Article5. Torrent R, De Angelis Rigotti F, Dell’Era P, Memo M, Raya A, Consiglio A. Using iPS cells toward the understanding of Parkinson’s disease. J Clin Med. 2015; 4:548–566. DOI: 10.3390/jcm4040548. PMID: 26239346. PMCID: PMC4470155.
Article6. Vierbuchen T, Ostermeier A, Pang ZP, Kokubu Y, Südhof TC, Wernig M. Direct conversion of fibroblasts to functional neurons by defined factors. Nature. 2010; 463:1035–1041. DOI: 10.1038/nature08797. PMID: 20107439. PMCID: PMC2829121.
Article7. Yoo J, Lee E, Kim HY, Youn DH, Jung J, Kim H, Chang Y, Lee W, Shin J, Baek S, Jang W, Jun W, Kim S, Hong J, Park HJ, Lengner CJ, Moh SH, Kwon Y, Kim J. Electromagnetized gold nanoparticles mediate direct lineage reprogramming into induced dopamine neurons in vivo for Parkinson’s disease therapy. Nat Nanotechnol. 2017; 12:1006–1014. DOI: 10.1038/nnano.2017.133. PMID: 28737745.
Article8. Pfisterer U, Kirkeby A, Torper O, Wood J, Nelander J, Dufour A, Björklund A, Lindvall O, Jakobsson J, Parmar M. Direct conversion of human fibroblasts to dopaminergic neurons. Proc Natl Acad Sci U S A. 2011; 108:10343–10348. DOI: 10.1073/pnas.1105135108. PMID: 21646515. PMCID: PMC3121829.
Article9. Caiazzo M, Dell’Anno MT, Dvoretskova E, Lazarevic D, Taverna S, Leo D, Sotnikova TD, Menegon A, Roncaglia P, Colciago G, Russo G, Carninci P, Pezzoli G, Gainetdinov RR, Gustincich S, Dityatev A, Broccoli V. Direct generation of functional dopaminergic neurons from mouse and human fibroblasts. Nature. 2011; 476:224–227. DOI: 10.1038/nature10284. PMID: 21725324.
Article10. Addis RC, Hsu FC, Wright RL, Dichter MA, Coulter DA, Gearhart JD. Efficient conversion of astrocytes to functional midbrain dopaminergic neurons using a single polycistronic vector. PLoS One. 2011; 6:e28719. DOI: 10.1371/journal.pone.0028719. PMID: 22174877. PMCID: PMC3235158.
Article11. Liu X, Li F, Stubblefield EA, Blanchard B, Richards TL, Larson GA, He Y, Huang Q, Tan AC, Zhang D, Benke TA, Sladek JR, Zahniser NR, Li CY. Direct reprogramming of human fibroblasts into dopaminergic neuron-like cells. Cell Res. 2012; 22:321–332. DOI: 10.1038/cr.2011.181. PMID: 22105488. PMCID: PMC3271588.
Article12. Kim J, Su SC, Wang H, Cheng AW, Cassady JP, Lodato MA, Lengner CJ, Chung CY, Dawlaty MM, Tsai LH, Jaenisch R. Functional integration of dopaminergic neurons directly converted from mouse fibroblasts. Cell Stem Cell. 2011; 9:413–419. DOI: 10.1016/j.stem.2011.09.011. PMID: 22019014. PMCID: PMC3210333.
Article13. Torper O, Pfisterer U, Wolf DA, Pereira M, Lau S, Jakobsson J, Björklund A, Grealish S, Parmar M. Generation of induced neurons via direct conversion in vivo. Proc Natl Acad Sci U S A. 2013; 110:7038–7043. DOI: 10.1073/pnas.1303829110. PMID: 23530235. PMCID: PMC3637783.
Article14. Jiang H, Xu Z, Zhong P, Ren Y, Liang G, Schilling HA, Hu Z, Zhang Y, Wang X, Chen S, Yan Z, Feng J. Cell cycle and p53 gate the direct conversion of human fibroblasts to dopaminergic neurons. Nat Commun. 2015; 6:10100. DOI: 10.1038/ncomms10100. PMID: 26639555. PMCID: PMC4672381.
Article15. Park H, Kim H, Yoo J, Lee J, Choi H, Baek S, Lee CJ, Kim J, Lengner CJ, Sung JS, Kim J. Homogeneous generation of iDA neurons with high similarity to bona fide DA neurons using a drug inducible system. Biomaterials. 2015; 72:152–162. DOI: 10.1016/j.biomaterials.2015.09.002. PMID: 26370928.
Article16. Rivetti di Val Cervo P, Romanov RA, Spigolon G, Masini D, Martín-Montañez E, Toledo EM, La Manno G, Feyder M, Pifl C, Ng YH, Sánchez SP, Linnarsson S, Wernig M, Harkany T, Fisone G, Arenas E. Induction of functional dopamine neurons from human astrocytes in vitro and mouse astrocytes in a Parkinson’s disease model. Nat Biotechnol. 2017; 35:444–452. DOI: 10.1038/nbt.3835. PMID: 28398344.
Article17. Kim J, Efe JA, Zhu S, Talantova M, Yuan X, Wang S, Lipton SA, Zhang K, Ding S. Direct reprogramming of mouse fibroblasts to neural progenitors. Proc Natl Acad Sci U S A. 2011; 108:7838–7843. DOI: 10.1073/pnas.1103113108. PMID: 21521790. PMCID: PMC3093517.
Article18. Efe JA, Hilcove S, Kim J, Zhou H, Ouyang K, Wang G, Chen J, Ding S. Conversion of mouse fibroblasts into cardiomyocytes using a direct reprogramming strategy. Nat Cell Biol. 2011; 13:215–222. DOI: 10.1038/ncb2164. PMID: 21278734.
Article19. Thier M, Wörsdörfer P, Lakes YB, Gorris R, Herms S, Opitz T, Seiferling D, Quandel T, Hoffmann P, Nöthen MM, Brüstle O, Edenhofer F. Direct conversion of fibroblasts into stably expandable neural stem cells. Cell Stem Cell. 2012; 10:473–479. DOI: 10.1016/j.stem.2012.03.003. PMID: 22445518.
Article20. Kim J, Ambasudhan R, Ding S. Direct lineage reprogramming to neural cells. Curr Opin Neurobiol. 2012; 22:778–784. DOI: 10.1016/j.conb.2012.05.001. PMID: 22652035. PMCID: PMC4945246.
Article21. Zhu S, Ambasudhan R, Sun W, Kim HJ, Talantova M, Wang X, Zhang M, Zhang Y, Laurent T, Parker J, Kim HS, Zaremba JD, Saleem S, Sanz-Blasco S, Masliah E, McKercher SR, Cho YS, Lipton SA, Kim J, Ding S. Small molecules enable OCT4-mediated direct reprogramming into expandable human neural stem cells. Cell Res. 2014; 24:126–129. DOI: 10.1038/cr.2013.156. PMID: 24296783. PMCID: PMC3879704.
Article22. Zhu S, Russ HA, Wang X, Zhang M, Ma T, Xu T, Tang S, Hebrok M, Ding S. Human pancreatic beta-like cells converted from fibroblasts. Nat Commun. 2016; 7:10080. DOI: 10.1038/ncomms10080. PMID: 26733021. PMCID: PMC4729817.
Article23. Wang H, Cao N, Spencer CI, Nie B, Ma T, Xu T, Zhang Y, Wang X, Srivastava D, Ding S. Small molecules enable cardiac reprogramming of mouse fibroblasts with a single factor, Oct4. Cell Rep. 2014; 6:951–960. DOI: 10.1016/j.celrep.2014.01.038. PMID: 24561253. PMCID: PMC4004339.
Article24. Lee JH, Mitchell RR, McNicol JD, Shapovalova Z, Laronde S, Tanasijevic B, Milsom C, Casado F, Fiebig-Comyn A, Collins TJ, Singh KK, Bhatia M. Single transcription factor conversion of human blood fate to NPCs with CNS and PNS developmental capacity. Cell Rep. 2015; 11:1367–1376. DOI: 10.1016/j.celrep.2015.04.056. PMID: 26004181.
Article25. Kurian L, Sancho-Martinez I, Nivet E, Aguirre A, Moon K, Pendaries C, Volle-Challier C, Bono F, Herbert JM, Pulecio J, Xia Y, Li M, Montserrat N, Ruiz S, Dubova I, Rodriguez C, Denli AM, Boscolo FS, Thiagarajan RD, Gage FH, Loring JF, Laurent LC, Izpisua Belmonte JC. Conversion of human fibroblasts to angioblast-like progenitor cells. Nat Methods. 2013; 10:77–83. DOI: 10.1038/nmeth.2255. PMID: 23202434. PMCID: PMC3531579.
Article26. Zhu S, Rezvani M, Harbell J, Mattis AN, Wolfe AR, Benet LZ, Willenbring H, Ding S. Mouse liver repopulation with hepatocytes generated from human fibroblasts. Nature. 2014; 508:93–97. DOI: 10.1038/nature13020. PMID: 24572354. PMCID: PMC4161230.
Article27. Li K, Zhu S, Russ HA, Xu S, Xu T, Zhang Y, Ma T, Hebrok M, Ding S. Small molecules facilitate the reprogramming of mouse fibroblasts into pancreatic lineages. Cell Stem Cell. 2014; 14:228–236. DOI: 10.1016/j.stem.2014.01.006. PMID: 24506886. PMCID: PMC4747235.
Article28. Zhang Y, Cao N, Huang Y, Spencer CI, Fu JD, Yu C, Liu K, Nie B, Xu T, Li K, Xu S, Bruneau BG, Srivastava D, Ding S. Expandable cardiovascular progenitor cells reprogrammed from fibroblasts. Cell Stem Cell. 2016; 18:368–381. DOI: 10.1016/j.stem.2016.02.001. PMID: 26942852. PMCID: PMC5826660.
Article29. Lu J, Liu H, Huang CT, Chen H, Du Z, Liu Y, Sherafat MA, Zhang SC. Generation of integration-free and region-specific neural progenitors from primate fibroblasts. Cell Rep. 2013; 3:1580–1591. DOI: 10.1016/j.celrep.2013.04.004. PMID: 23643533. PMCID: PMC3786191.
Article30. Han DW, Tapia N, Hermann A, Hemmer K, Höing S, Araúzo-Bravo MJ, Zaehres H, Wu G, Frank S, Moritz S, Greber B, Yang JH, Lee HT, Schwamborn JC, Storch A, Schöler HR. Direct reprogramming of fibroblasts into neural stem cells by defined factors. Cell Stem Cell. 2012; 10:465–472. DOI: 10.1016/j.stem.2012.02.021. PMID: 22445517.
Article31. Sheng C, Jungverdorben J, Wiethoff H, Lin Q, Flitsch LJ, Eckert D, Hebisch M, Fischer J, Kesavan J, Weykopf B, Schneider L, Holtkamp D, Beck H, Till A, Wüllner U, Ziller MJ, Wagner W, Peitz M, Brüstle O. A stably self-renewing adult blood-derived induced neural stem cell exhibiting patternability and epigenetic rejuvenation. Nat Commun. 2018; 9:4047. DOI: 10.1038/s41467-018-06398-5. PMID: 30279449. PMCID: PMC6168501.
Article32. Cairns DM, Chwalek K, Moore YE, Kelley MR, Abbott RD, Moss S, Kaplan DL. Expandable and rapidly differentiating human induced neural stem cell lines for multiple tissue engineering applications. Stem Cell Reports. 2016; 7:557–570. DOI: 10.1016/j.stemcr.2016.07.017. PMID: 27569063. PMCID: PMC5032182.
Article33. Jung KB, Lee H, Son YS, Lee JH, Cho HS, Lee MO, Oh JH, Lee J, Kim S, Jung CR, Kim J, Son MY. In vitro and in vivo imaging and tracking of intestinal organoids from human induced pluripotent stem cells. FASEB J. 2018; 32:111–122. DOI: 10.1096/fj.201700504R. PMID: 28855280.
Article34. Tang J, Hu M, Lee S, Roblin R. A polymerase chain reaction based method for detecting Mycoplasma/Acholeplasma contaminants in cell culture. J Microbiol Methods. 2000; 39:121–126. DOI: 10.1016/S0167-7012(99)00107-4. PMID: 10576701.
Article35. Weissbein U, Ben-David U, Benvenisty N. Virtual karyotyping reveals greater chromosomal stability in neural cells derived by transdifferentiation than those from stem cells. Cell Stem Cell. 2014; 15:687–691. DOI: 10.1016/j.stem.2014.10.018. PMID: 25479746.
Article36. Traynelis SF, Wollmuth LP, McBain CJ, Menniti FS, Vance KM, Ogden KK, Hansen KB, Yuan H, Myers SJ, Dingledine R. Glutamate receptor ion channels: structure, regulation, and function. Pharmacol Rev. 2010; 62:405–496. DOI: 10.1124/pr.109.002451. PMID: 20716669. PMCID: PMC2964903.
Article37. Potapova TA, Zhu J, Li R. Aneuploidy and chromosomal instability: a vicious cycle driving cellular evolution and cancer genome chaos. Cancer Metastasis Rev. 2013; 32:377–389. DOI: 10.1007/s10555-013-9436-6. PMID: 23709119. PMCID: PMC3825812.
Article38. Nguyen HN, Byers B, Cord B, Shcheglovitov A, Byrne J, Gujar P, Kee K, Schüle B, Dolmetsch RE, Langston W, Palmer TD, Pera RR. LRRK2 mutant iPSC-derived DA neurons demonstrate increased susceptibility to oxidative stress. Cell Stem Cell. 2011; 8:267–280. DOI: 10.1016/j.stem.2011.01.013. PMID: 21362567. PMCID: PMC3578553.
Article39. Cooper O, Seo H, Andrabi S, Guardia-Laguarta C, Graziotto J, Sundberg M, McLean JR, Carrillo-Reid L, Xie Z, Osborn T, Hargus G, Deleidi M, Lawson T, Bogetofte H, Perez-Torres E, Clark L, Moskowitz C, Mazzulli J, Chen L, Volpicelli-Daley L, Romero N, Jiang H, Uitti RJ, Huang Z, Opala G, Scarffe LA, Dawson VL, Klein C, Feng J, Ross OA, Trojanowski JQ, Lee VM, Marder K, Surmeier DJ, Wszolek ZK, Przedborski S, Krainc D, Dawson TM, Isacson O. Pharmacological rescue of mitochondrial deficits in iPSC-derived neural cells from patients with familial Parkinson’s disease. Sci Transl Med. 2012; 4:141ra90. DOI: 10.1126/scitranslmed.3003985. PMID: 22764206. PMCID: PMC3462009.
Article40. Liu GH, Qu J, Suzuki K, Nivet E, Li M, Montserrat N, Yi F, Xu X, Ruiz S, Zhang W, Wagner U, Kim A, Ren B, Li Y, Goebl A, Kim J, Soligalla RD, Dubova I, Thompson J, Yates J 3rd, Esteban CR, Sancho-Martinez I, Izpisua Belmonte JC. Progressive degeneration of human neural stem cells caused by pathogenic LRRK2. Nature. 2012; 491:603–607. DOI: 10.1038/nature11557. PMID: 23075850. PMCID: PMC3504651.
Article41. Caldwell KA, Tucci ML, Armagost J, Hodges TW, Chen J, Memon SB, Blalock JE, DeLeon SM, Findlay RH, Ruan Q, Webber PJ, Standaert DG, Olson JB, Caldwell GA. Investigating bacterial sources of toxicity as an environmental contributor to dopaminergic neurodegeneration. PLoS One. 2009; 4:e7227. DOI: 10.1371/journal.pone.0007227. PMID: 19806188. PMCID: PMC2751819.
Article42. Bentea E, Verbruggen L, Massie A. The proteasome inhibition model of Parkinson’s disease. J Parkinsons Dis. 2017; 7:31–63. DOI: 10.3233/JPD-160921. PMID: 27802243. PMCID: PMC5302045.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Strategic Application of Epigenetic Regulators for Efficient Neuronal Reprogramming of Human Fibroblasts
- Mbd3 Promotes Reprogramming of Primary Human Fibroblasts
- Comparison of Reprogramming Methods for Generation of Induced-Oligodendrocyte Precursor Cells
- Reprogramming of Cancer Cells into Induced Pluripotent Stem Cells Questioned
- Trends and clinical application of induced pluripotent stem cells