Ann Dermatol.
2019 Aug;31(4):403-413. 10.5021/ad.2019.31.4.403.
Polydeoxyribonucleotides Improve Diabetic Wound Healing in Mouse Animal Model for Experimental Validation
- Affiliations
-
- 1Department of Dermatology, Chung-Ang University College of Medicine, Seoul, Korea. beomjoon@unitel.co.kr
- 2Department of Medicine, Graduate School, Chung-Ang University, Seoul, Korea.
- KMID: 2451477
- DOI: http://doi.org/10.5021/ad.2019.31.4.403
Abstract
- BACKGROUND
Wound healing mechanisms is believed to have effects similar to wound healing disorders in diabetic patients, including abnormal inflammatory cells, angiogenesis disorders, and reduced collagen synthesis. Therefore, reestablishment of structural and promoted angiogenesis could be beneficial to promote wound healing process.
OBJECTIVE
Therefore, we investigated whether the polydeoxyribonucleotide (PDRN) that was self-production in Korea, could be useful as an intradermal injection for promoting wound healing. Also, we validate for wound healing effect of PDRN using healing-impaired (db/db) mice.
METHODS
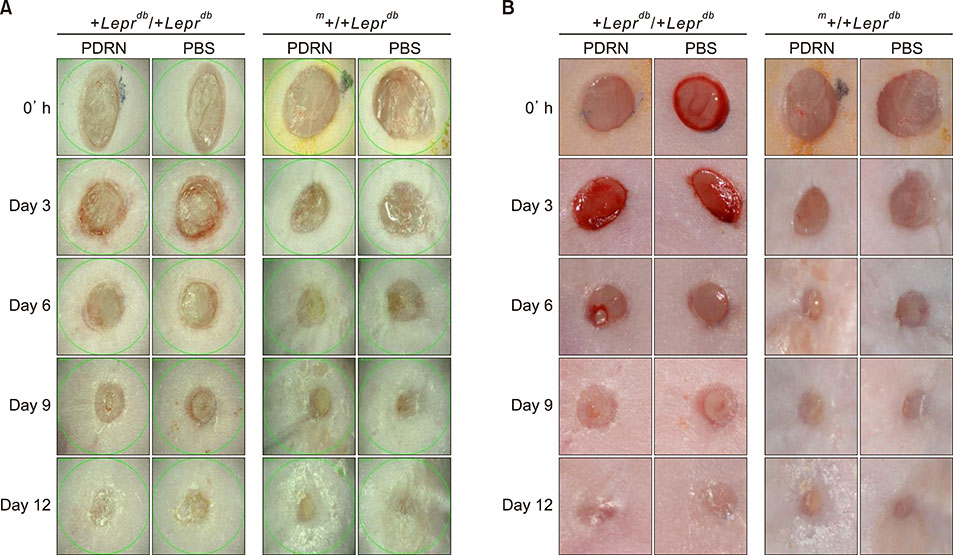
In this study, we confirmed the effects of PDRN by creating wound models in in vitro and in vivo model. Using an in vitro wound healing assay, we observed that PDRN stimulated closure of wounded monolayers of human fibroblast cells. PDRN (8.25 mg/ml) or phosphate-buffered saline (0.9% NaCl) was injected once daily into the dermis adjacent to the wound for 12 days after skin injury.
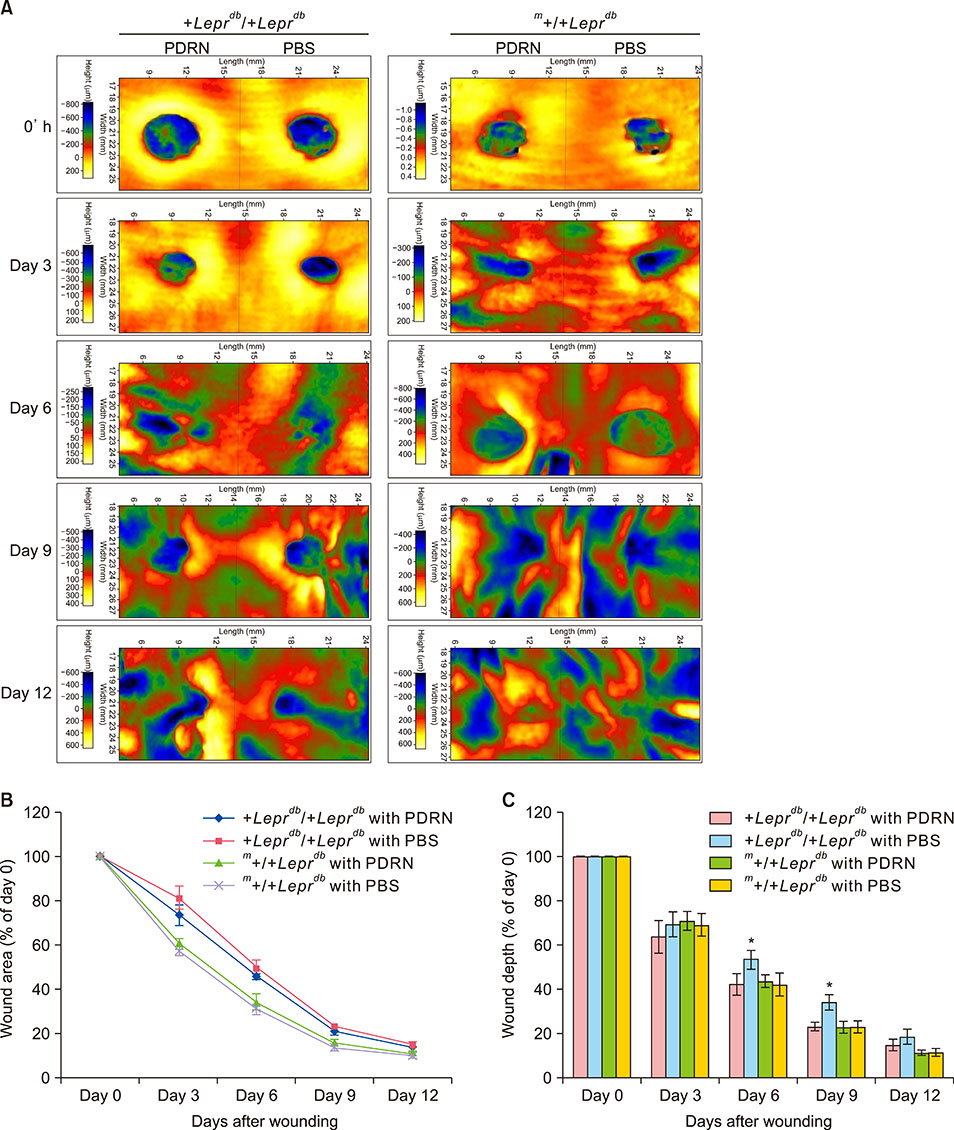
RESULTS
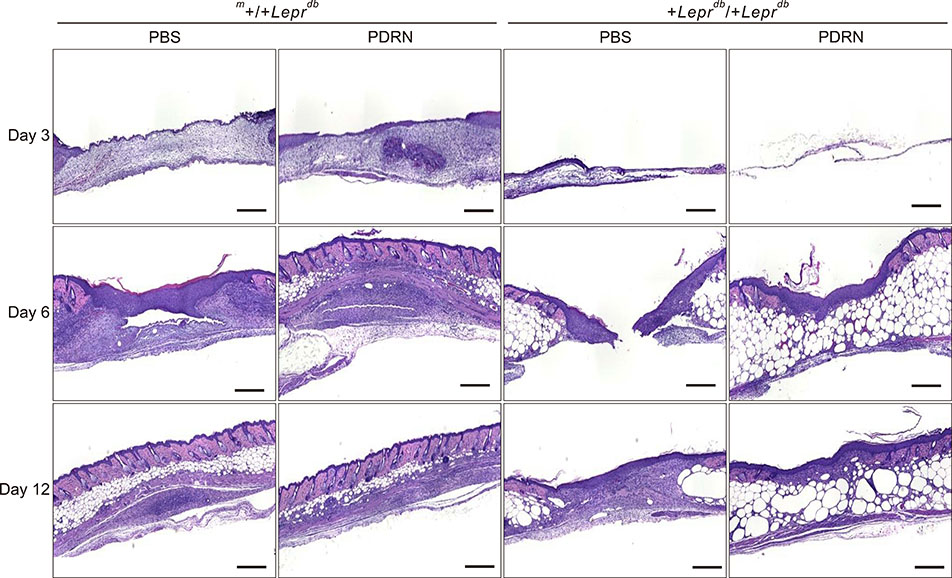
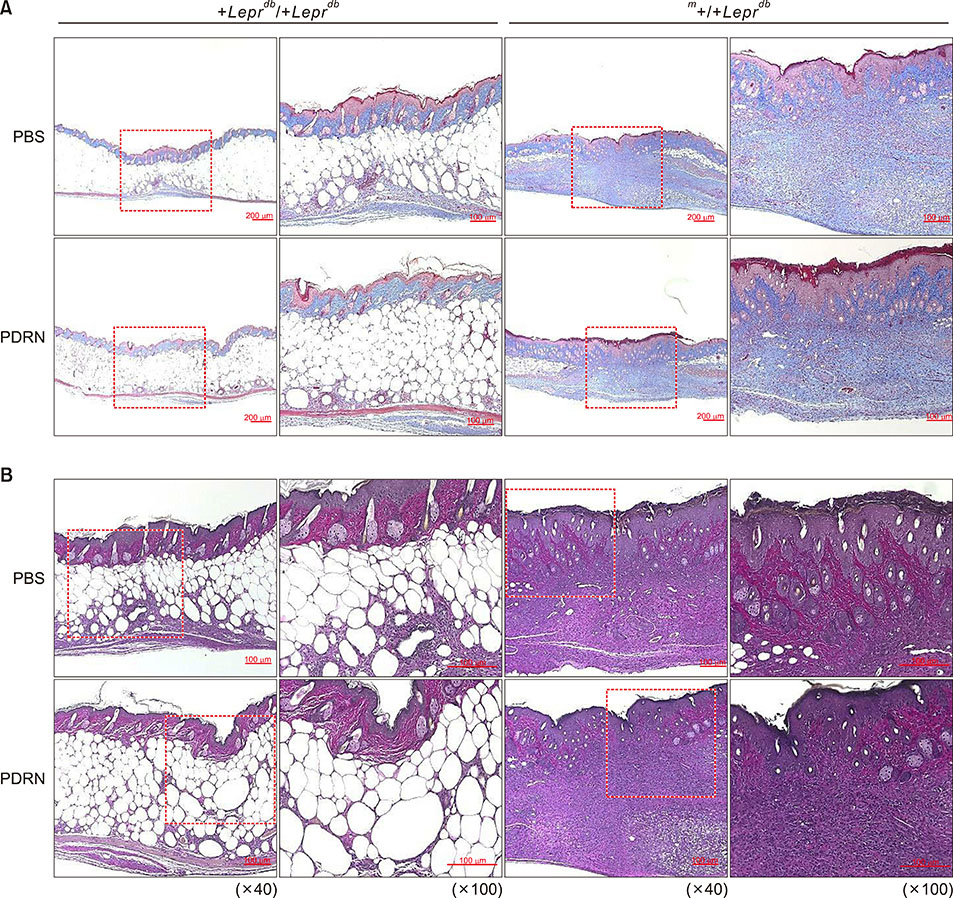
Time course observations revealed that mice treated with PDRN showed accelerated wound closure and epidermal and dermal regeneration, enhanced angiogenesis. The wound area and depth decreased at 3, 6, 9, and 12 days after skin injury. Histological evaluation showed an increase of vascular endothelial growth factor, CD31, and collagen fibers in the PDRN group compared with the control group, indicating that PDRN was effective in the treatment of delayed wound healing caused by diabetes.
CONCLUSION
This study suggests that our PDRN has a wound healing effect in transgenic animal models with cells and diabetes through angiogenesis.
Keyword
MeSH Terms
-
Animals
Animals*
Animals, Genetically Modified
Collagen
Dermis
Fibroblasts
Humans
In Vitro Techniques
Injections, Intradermal
Korea
Mice*
Models, Animal*
Polydeoxyribonucleotides*
Regeneration
Skin
Vascular Endothelial Growth Factor A
Wound Healing*
Wounds and Injuries*
Collagen
Polydeoxyribonucleotides
Vascular Endothelial Growth Factor A
Figure
Reference
-
1. Oryan A, Alemzadeh E. Effects of insulin on wound healing: a review of animal and human evidences. Life Sci. 2017; 174:59–67.
Article2. Galeano M, Bitto A, Altavilla D, Minutoli L, Polito F, Calò M, et al. Polydeoxyribonucleotide stimulates angiogenesis and wound healing in the genetically diabetic mouse. Wound Repair Regen. 2008; 16:208–217.
Article3. Jeong W, Yang CE, Roh TS, Kim JH, Lee JH, Lee WJ. Scar prevention and enhanced wound healing induced by polydeoxyribonucleotide in a rat incisional wound-healing model. Int J Mol Sci. 2017; 18:E1698.
Article4. Barrientos S, Stojadinovic O, Golinko MS, Brem H, Tomic-Canic M. Growth factors and cytokines in wound healing. Wound Repair Regen. 2008; 16:585–601.5. Ebrahimian TG, Pouzoulet F, Squiban C, Buard V, André M, Cousin B, et al. Cell therapy based on adipose tissue-derived stromal cells promotes physiological and pathological wound healing. Arterioscler Thromb Vasc Biol. 2009; 29:503–510.
Article6. Smiell JM, Wieman TJ, Steed DL, Perry BH, Sampson AR, Schwab BH. Efficacy and safety of becaplermin (recombinant human platelet-derived growth factor-BB) in patients with nonhealing, lower extremity diabetic ulcers: a combined analysis of four randomized studies. Wound Repair Regen. 1999; 7:335–346.
Article7. Han G, Ceilley R. Chronic wound healing: a review of current management and treatments. Adv Ther. 2017; 34:599–610.
Article8. Cianfarani F, Zambruno G, Brogelli L, Sera F, Lacal PM, Pesce M, et al. Placenta growth factor in diabetic wound healing: altered expression and therapeutic potential. Am J Pathol. 2006; 169:1167–1182.9. Powell RJ, Goodney P, Mendelsohn FO, Moen EK, Annex BH. HGF-0205 Trial Investigators. Safety and efficacy of patient specific intramuscular injection of HGF plasmid gene therapy on limb perfusion and wound healing in patients with ischemic lower extremity ulceration: results of the HGF-0205 trial. J Vasc Surg. 2010; 52:1525–1530.
Article10. Altavilla D, Bitto A, Polito F, Marini H, Minutoli L, Di Stefano V, et al. Polydeoxyribonucleotide (PDRN): a safe approach to induce therapeutic angiogenesis in peripheral artery occlusive disease and in diabetic foot ulcers. Cardiovasc Hematol Agents Med Chem. 2009; 7:313–321.
Article11. Kim SK, Huh CK, Lee JH, Kim KW, Kim MY. H Histologic study of bone-forming capacity on polydeoxyribonucleotide combined with demineralized dentin matrix. Maxillofac Plast Reconstr Surg. 2016; 38:7.
Article12. Veronesi F, Dallari D, Sabbioni G, Carubbi C, Martini L, Fini M. Polydeoxyribonucleotides (PDRNs) from skin to musculoskeletal tissue regeneration via adenosine A2A receptor involvement. J Cell Physiol. 2017; 232:2299–2307.
Article13. Bitto A, Galeano M, Squadrito F, Minutoli L, Polito F, Dye JF, et al. Polydeoxyribonucleotide improves angiogenesis and wound healing in experimental thermal injury. Crit Care Med. 2008; 36:1594–1602.
Article14. Squadrito F, Bitto A, Irrera N, Pizzino G, Pallio G, Minutoli L, et al. Pharmacological activity and clinical use of PDRN. Front Pharmacol. 2017; 8:224.
Article15. Wu CH, Chang GY, Chang WC, Hsu CT, Chen RS. Wound healing effects of porcine placental extracts on rats with thermal injury. Br J Dermatol. 2003; 148:236–245.
Article16. Altavilla D, Squadrito F, Polito F, Irrera N, Calò M, Lo Cascio P, et al. Activation of adenosine A2A receptors restores the altered cell-cycle machinery during impaired wound healing in genetically diabetic mice. Surgery. 2011; 149:253–261.
Article17. Lee SJ, Lee HM, Ji ST, Lee SR, Mar W, Gho YS. 1,2,3,4,6-Penta-O-galloyl-beta-D-glucose blocks endothelial cell growth and tube formation through inhibition of VEGF binding to VEGF receptor. Cancer Lett. 2004; 208:89–94.
Article18. Montesinos MC, Desai A, Chen JF, Yee H, Schwarzschild MA, Fink JS, et al. Adenosine promotes wound healing and mediates angiogenesis in response to tissue injury via occupancy of A(2A) receptors. Am J Pathol. 2002; 160:2009–2018.
Article19. Brem H, Stojadinovic O, Diegelmann RF, Entero H, Lee B, Pastar I, et al. Molecular markers in patients with chronic wounds to guide surgical debridement. Mol Med. 2007; 13:30–39.
Article20. Brem H, Tomic-Canic M. Cellular and molecular basis of wound healing in diabetes. J Clin Invest. 2007; 117:1219–1222.
Article21. Reiber GE, Vileikyte L, Boyko EJ, del Aguila M, Smith DG, Lavery LA, et al. Causal pathways for incident lower-extremity ulcers in patients with diabetes from two settings. Diabetes Care. 1999; 22:157–162.
Article22. Christman AL, Selvin E, Margolis DJ, Lazarus GS, Garza LA. Hemoglobin A1c predicts healing rate in diabetic wounds. J Invest Dermatol. 2011; 131:2121–2127.
Article23. Emmerson E, Campbell L, Davies FC, Ross NL, Ashcroft GS, Krust A, et al. Insulin-like growth factor-1 promotes wound healing in estrogen-deprived mice: new insights into cutaneous IGF-1R/ERα cross talk. J Invest Dermatol. 2012; 132:2838–2848.
Article24. Lima MH, Caricilli AM, de Abreu LL, Araújo EP, Pelegrinelli FF, Thirone AC, et al. Topical insulin accelerates wound healing in diabetes by enhancing the AKT and ERK pathways: a double-blind placebo-controlled clinical trial. PLoS One. 2012; 7:e36974.
Article25. Polito F, Bitto A, Galeano M, Irrera N, Marini H, Calò M, et al. Polydeoxyribonucleotide restores blood flow in an experimental model of ischemic skin flaps. J Vasc Surg. 2012; 55:479–488.
Article26. Yan Y, Zhu L, Hong L, Deng J, Song Y, Chen X. The impact of ranibizumab on the level of intercellular adhesion molecule type 1 in the vitreous of eyes with proliferative diabetic retinopathy. Acta Ophthalmol. 2016; 94:358–364.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effect of Polydeoxyribonucleotide on Chronic Non-healing Wound of an Amputee: A Case Report
- Effects of Polydeoxyribonucleotide in the Treatment of Pressure Ulcers
- Relationship between the Diurnal Temperature Range and Wound Healing of Diabetic Foot: Animal Study
- Comparison between Ketogenic and Diabetic Conventional Diet on Wound Closure in Diabetic Rat Model
- Effect of beta-glucan from Aureobasidium on dermal wound healing in diabetic C57BL/KsJ-db/db mouse model