J Korean Med Sci.
2014 Nov;29(Suppl 3):S222-S227. 10.3346/jkms.2014.29.S3.S222.
Effects of Polydeoxyribonucleotide in the Treatment of Pressure Ulcers
- Affiliations
-
- 1Department of Plastic and Reconstructive Surgery, Seoul National University College of Medicine, Seoul, Korea. lionheo@snu.ac.kr
- KMID: 2151417
- DOI: http://doi.org/10.3346/jkms.2014.29.S3.S222
Abstract
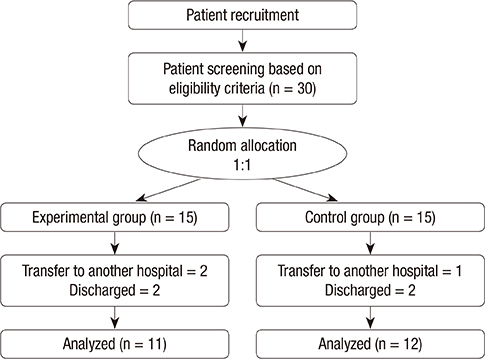
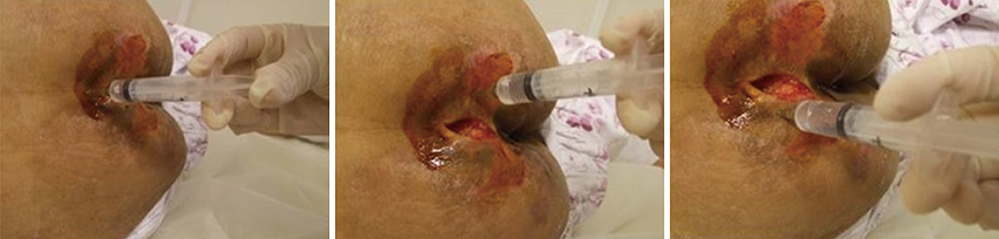
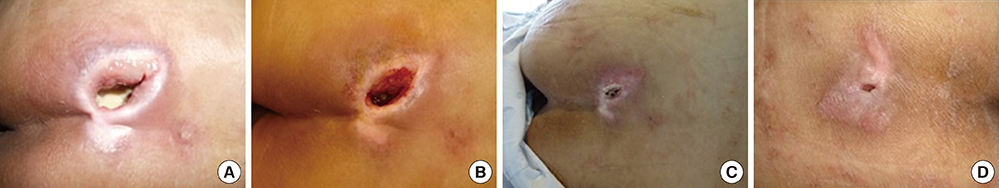
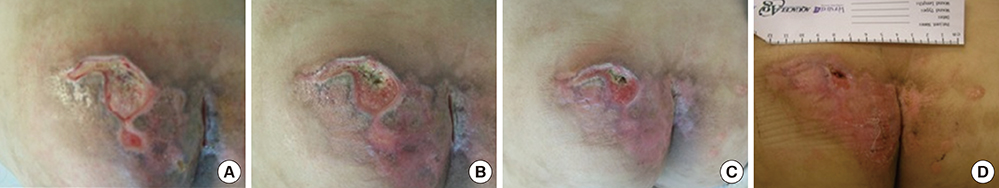
- This study aimed to examine the positive effects of polydeoxyribonucleotide (PDRN) on the wound-healing process in pressure ulcers. In this randomized controlled trial, the effects of PDRN were compared over time between an experimental group (n=11) and a control group (n=12). The former was administered the same dose of PDRN intramuscularly (1 ampule, 3 mL, 5.625 mg, for 5 days) for 2 weeks and perilesionally (1 ampule, 3 mL, 5.625 mg, twice a week) for 4 weeks. The primary endpoint for determining efficacy was wound healing in the pressure ulcers, which was reflected by the wound surface area determined using VISITRAK Digital (Smith & Nephew, Largo, FL). The secondary endpoint was the pressure ulcer scale for healing score, determined using pressure ulcer scale for healing (PUSH Tool 3.0 developed by the National Pressure Ulcer Advisory Panel). After the 4-week treatment period, PDRN therapy was found to significantly reduce the wound size and PUSH score, without adverse effect during the treatment. The findings indicate that PDRN can positively modify the wound healing process in pressure ulcers, and its use could improve the clinical outcomes of patients and lower the need for additional therapies or hospital stay.
MeSH Terms
Figure
Cited by 2 articles
-
How Effective Is the Injection Therapy in Foot and Ankle Disorder?
Ha Heon Song
J Korean Foot Ankle Soc. 2021;25(1):10-16. doi: 10.14193/jkfas.2021.25.1.10.The Effect of Polydeoxyribonucleotide on Chronic Non-healing Wound of an Amputee: A Case Report
Junho Shin, Gahee Park, Junhee Lee, Hasuk Bae
Ann Rehabil Med. 2018;42(4):630-633. doi: 10.5535/arm.2018.42.4.630.
Reference
-
1. Defloor T. The risk of pressure sores: a conceptual scheme. J Clin Nurs. 1999; 8:206–216.2. Baumgarten M, Margolis DJ, Localio AR, Kagan SH, Lowe RA, Kinosian B, Holmes JH, Abbuhl SB, Kavesh W, Ruffin A. Pressure ulcers among elderly patients early in the hospital stay. J Gerontol A Biol Sci Med Sci. 2006; 61:749–754.3. Pressure ulcers in America: prevalence, incidence, and implications for the future. An executive summary of the National Pressure Ulcer Advisory Panel monograph. Adv Skin Wound Care. 2001; 14:208–215.4. Stausberg J, Kröger K, Maier I, Schneider H, Niebel W. Pressure ulcers in secondary care: incidence, prevalence, and relevance. Adv Skin Wound Care. 2005; 18:140–145.5. Lee MJ. Risk factors of pressure ulcers among elderly in the Geriatric Hospital. Seoul: Ewha Woman University;2010. Dissertation.6. Hopkins A, Dealey C, Bale S, Defloor T, Worboys F. Patient stories of living with a pressure ulcer. J Adv Nurs. 2006; 56:345–353.7. Bergstorm N. Patients at risk for pressure ulcers and evidence-based care for pressure ulcer prevention. In : Bader D, Bouten C, Colin D, Oomens C, editors. Pressure ulcer research: current and future perspectives. Berlin: Springer;2005. p. 35–50.8. Parish LC, Dryjski M, Cadden S. Prospective clinical study of a new adhesive gelling foam dressing in pressure ulcers. Int Wound J. 2008; 5:60–67.9. Guizzardi S, Galli C, Govoni P, Boratto R, Cattarini G, Martini D, Belletti S, Scandroglio R. Polydeoxyribonucleotide (PDRN) promotes human osteoblast proliferation: a new proposal for bone tissue repair. Life Sci. 2003; 73:1973–1983.10. Lazzarotto M, Tomasello EM, Caporossi A. Clinical evaluation of corneal epithelialization after photorefractive keratectomy in patients treated with polydeoxyribonucleotide (PDRN) eye drops: a randomized, double-blind, placebo-controlled trial. Eur J Ophthalmol. 2004; 14:284–289.11. De Aloe G, Rubegni P, Biagioli M, Taddeucci P, Fimiani M. Skin graft donor site and use of polydeoxyribonucleotide as a treatment for skin regeneration: a randomized, controlled, double-blind, clinical trial. Wounds. 2004; 16:258–263.12. Rubegni P, De Aloe G, Mazzatenta C, Cattarini L, Fimiani M. Clinical evaluation of the trophic effect of polydeoxyribonucleotide (PDRN) in patients undergoing skin explants: a pilot study. Curr Med Res Opin. 2001; 17:128–131.13. Squadrito F, Bitto A, Altavilla D, Arcoraci V, De Caridi G, De Feo ME, Corrao S, Pallio G, Sterrantino C, Minutoli L, et al. The effect of PDRN, an adenosine receptor A2A agonist, on the healing of chronic diabetic foot ulcers: results of a clinical trial. J Clin Endocrinol Metab. 2014; 99:E746–E753.14. Ferreira MC, Tuma P Jr, Carvalho VF, Kamamoto F. Complex wounds. Clinics (Sao Paulo). 2006; 61:571–578.15. Jaul E. Non-healing wounds: the geriatric approach. Arch Gerontol Geriatr. 2009; 49:224–226.16. Fonder MA, Lazarus GS, Cowan DA, Aronson-Cook B, Kohli AR, Mamelak AJ. Treating the chronic wound: a practical approach to the care of nonhealing wounds and wound care dressings. J Am Acad Dermatol. 2008; 58:185–206.17. Shin HS, Oh HY. The effect of platelet-rich plasma on wounds of OLETF rats using expression of matrix metalloproteinase-2 and -9 mRNA. Arch Plast Surg. 2012; 39:106–112.18. Choi J, Minn KW, Chang H. The efficacy and safety of platelet-rich plasma and adipose-derived stem cells: an update. Arch Plast Surg. 2012; 39:585–592.19. Yang JD, Choi DS, Cho YK, Kim TK, Lee JW, Choi KY, Chung HY, Cho BC, Byun JS. Effect of amniotic fluid stem cells and amniotic fluid cells on the wound healing process in a white rat model. Arch Plast Surg. 2013; 40:496–504.20. Kang BS, Na YC, Jin YW. Comparison of the wound healing effect of cellulose and gelatin: an in vivo study. Arch Plast Surg. 2012; 39:317–321.21. Kim H, Son D, Choi TH, Jung S, Kwon S, Kim J, Han K. Evaluation of an amniotic membrane-collagen dermal substitute in the management of full-thickness skin defects in a pig. Arch Plast Surg. 2013; 40:11–18.22. Chung KI, Kim HK, Kim WS, Bae TH. The effects of polydeoxyribonucleotide on the survival of random pattern skin flaps in rats. Arch Plast Surg. 2013; 40:181–186.23. Bitto A, Galeano M, Squadrito F, Minutoli L, Polito F, Dye JF, Clayton EA, Calò M, Venuti FS, Vaccaro M, et al. Polydeoxyribonucleotide improves angiogenesis and wound healing in experimental thermal injury. Crit Care Med. 2008; 36:1594–1602.24. Thellung S, Florio T, Maragliano A, Cattarini G, Schettini G. Polydeoxyribonucleotides enhance the proliferation of human skin fibroblasts: involvement of A2 purinergic receptor subtypes. Life Sci. 1999; 64:1661–1674.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical Updates in Polydeoxyribonucleotide Injection
- Conservative Treatment of Stage of Pressure Ulcers with Ulmus
- The Wound Healing Effect of PDRN(polydeoxyribonucleotide) Material on Full Thickness Skin Defect in the Mouse
- Evaluation of Risk for Pressure Ulcers Using the Braden Scale in Elderly Patients Receiving Long-Term Care
- Combined Treatment of Negative Pressure Wound Therapy and Temporary Tension Suture for Intractable Hindfoot Ulcer with Chronic Osteomyelitis: A Case Report