Ann Dermatol.
2018 Dec;30(6):645-652. 10.5021/ad.2018.30.6.645.
The Effect of Adiponectin on the Regulation of Filaggrin Expression in Normal Human Epidermal Keratinocytes
- Affiliations
-
- 1Department of Dermatology, Inje University Seoul Paik Hospital, Inje University College of Medicine, Seoul, Korea.
- 2Department of Dermatology, Chung-Ang University Hospital, Chung-Ang University College of Medicine, Seoul, Korea. drseo@hanafos.com, kyky@caumc.or.kr
- 3Department of Laboratory Medicine, Chung-Ang University Hospital, Chung-Ang University College of Medicine, Seoul, Korea.
- KMID: 2428919
- DOI: http://doi.org/10.5021/ad.2018.30.6.645
Abstract
- BACKGROUND
Adiponectin, an adipokine secreted from adipocytes, affects energy metabolism and also shows anti-diabetic and anti-inflammatory properties. Recent studies have reported that adiponectin plays a role in regulating skin inflammation.
OBJECTIVE
This study aimed to investigate the effect of adiponectin on the expression of filaggrin (FLG) in normal human epidermal keratinocytes (NHEKs).
METHODS
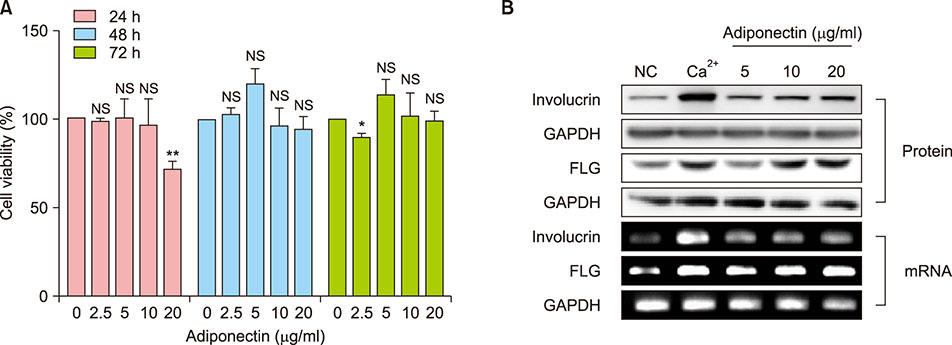
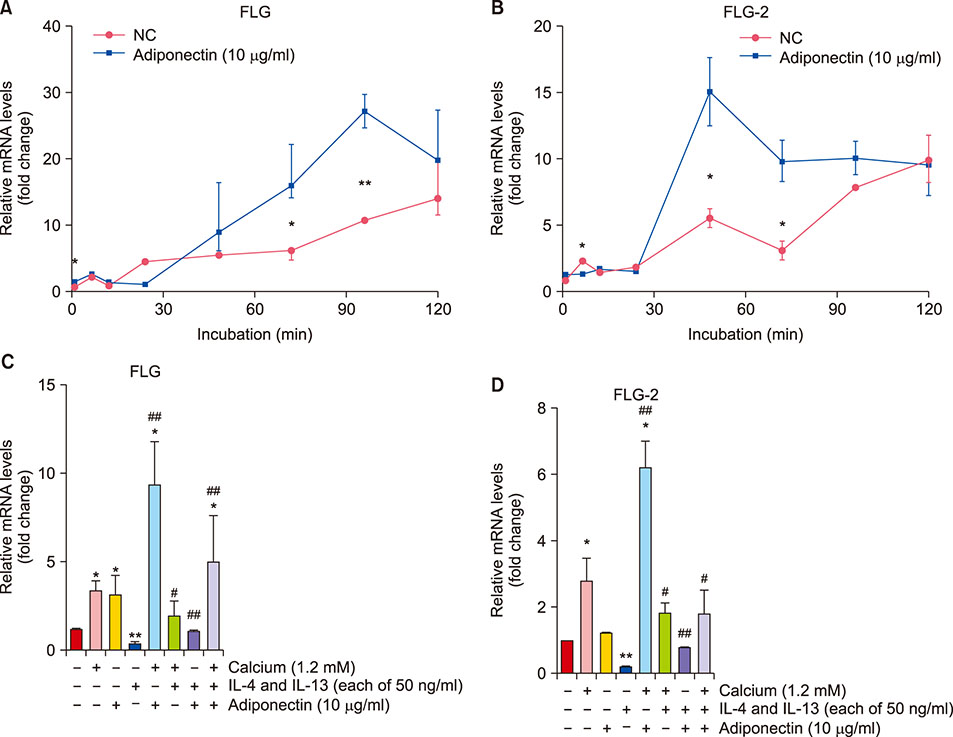
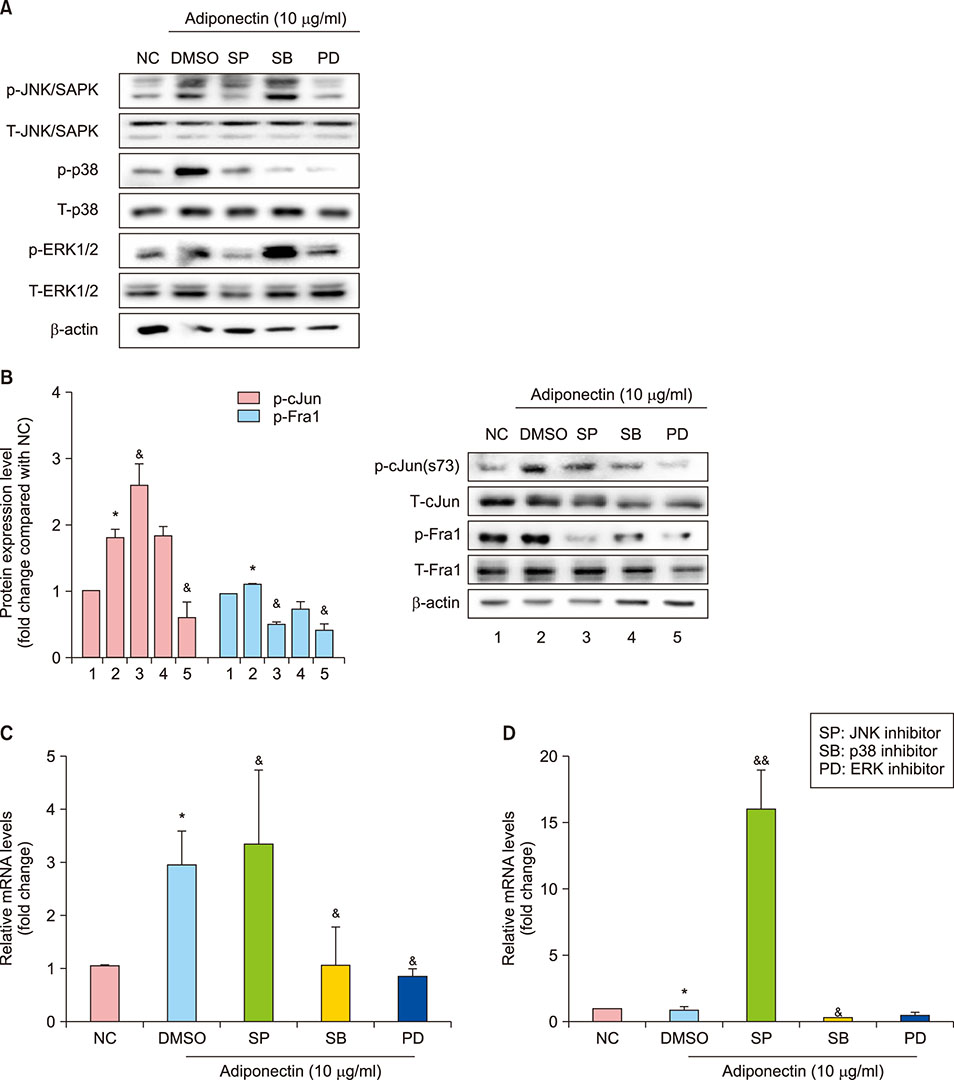
NHEKs were serum-starved for 6h before being treated with adiponectin. Afterward, cell viability was assessed by MTT assay. We also treated with calcium, interleukin (IL)-4, and IL-13 to provide positive and negative comparative controls, respectively. Gene mRNA expression was quantified using real time reverse transcription polymerase chain reaction, and protein expression was evaluated using Western blot. To evaluate the relationship among mitogen-activated protein kinases (MAPKs), activator protein 1 (AP-1), and FLG, we also treated cells with inhibitors for MAPKs JNK, p38, and ERK1/2.
RESULTS
FLG and FLG-2 mRNA expression in NHEKs significantly increased after treatment with 10 µg/ml adiponectin. Adiponectin also restored FLG and FLG-2 mRNA expression that was otherwise inhibited by treatment with IL-4 and IL-13. Adiponectin induced FLG expression via AP-1 and MAPK signaling.
CONCLUSION
Adiponectin positively regulated the expression of FLG and could be useful as a therapeutic agent to control diseases related to disrupted skin barrier function.
Keyword
MeSH Terms
-
Adipocytes
Adipokines
Adiponectin*
Blotting, Western
Calcium
Cell Differentiation
Cell Survival
Energy Metabolism
Humans*
Inflammation
Interleukin-13
Interleukin-4
Interleukins
Keratinocytes*
Mitogen-Activated Protein Kinases
Polymerase Chain Reaction
Reverse Transcription
RNA, Messenger
Skin
Transcription Factor AP-1
Adipokines
Adiponectin
Calcium
Interleukin-13
Interleukin-4
Interleukins
Mitogen-Activated Protein Kinases
RNA, Messenger
Transcription Factor AP-1
Figure
Cited by 1 articles
-
A Novel Effect of Lipids Extracted from Vernix Caseosa on Regulation of Filaggrin Expression in Human Epidermal Keratinocytes
Wu Qiao, Tinghan Jia, Hongjian Gu, Ruihua Guo, Ken Kaku, Wenhui Wu
Ann Dermatol. 2019;31(6):611-620. doi: 10.5021/ad.2019.31.6.611.
Reference
-
1. Scherer PE, Williams S, Fogliano M, Baldini G, Lodish HF. A novel serum protein similar to C1q, produced exclusively in adipocytes. J Biol Chem. 1995; 270:26746–26749.
Article2. Weyer C, Funahashi T, Tanaka S, Hotta K, Matsuzawa Y, Pratley RE, et al. Hypoadiponectinemia in obesity and type 2 diabetes: close association with insulin resistance and hyperinsulinemia. J Clin Endocrinol Metab. 2001; 86:1930–1935.
Article3. Weiss R, Taksali SE, Dufour S, Yeckel CW, Papademetris X, Cline G, et al. The “obese insulin-sensitive” adolescent: importance of adiponectin and lipid partitioning. J Clin Endocrinol Metab. 2005; 90:3731–3737.
Article4. Goldstein BJ, Scalia R. Adiponectin: a novel adipokine linking adipocytes and vascular function. J Clin Endocrinol Metab. 2004; 89:2563–2568.
Article5. Magge SN, Stettler N, Koren D, Levitt Katz LE, Gallagher PR, Mohler ER 3rd, et al. Adiponectin is associated with favorable lipoprotein profile, independent of BMI and insulin resistance, in adolescents. J Clin Endocrinol Metab. 2011; 96:1549–1554.
Article6. Ouchi N, Kihara S, Arita Y, Okamoto Y, Maeda K, Kuriyama H, et al. Adiponectin, an adipocyte-derived plasma protein, inhibits endothelial NF-kappaB signaling through a cAMP-dependent pathway. Circulation. 2000; 102:1296–1301.
Article7. Salathia NS, Shi J, Zhang J, Glynne RJ. An in vivo screen of secreted proteins identifies adiponectin as a regulator of murine cutaneous wound healing. J Invest Dermatol. 2013; 133:812–821.
Article8. Shibata S, Tada Y, Asano Y, Hau CS, Kato T, Saeki H, et al. Adiponectin regulates cutaneous wound healing by promoting keratinocyte proliferation and migration via the ERK signaling pathway. J Immunol. 2012; 189:3231–3241.
Article9. Takahashi H, Honma M, Ishida-Yamamoto A, Iizuka H. Adiponectin and leptin modulate cell proliferation and cytokine secretion of normal human keratinocytes and T lymphocytes. J Dermatol Sci. 2010; 59:143–145.
Article10. Kawai K, Kageyama A, Tsumano T, Nishimoto S, Fukuda K, Yokoyama S, et al. Effects of adiponectin on growth and differentiation of human keratinocytes--implication of impaired wound healing in diabetes. Biochem Biophys Res Commun. 2008; 374:269–273.
Article11. Shibata S, Tada Y, Hau CS, Mitsui A, Kamata M, Asano Y, et al. Adiponectin regulates psoriasiform skin inflammation by suppressing IL-17 production from γδ-T cells. Nat Commun. 2015; 6:7687.
Article12. Wolk K, Sabat R. Adipokines in psoriasis: an important link between skin inflammation and metabolic alterations. Rev Endocr Metab Disord. 2016; 17:305–317.
Article13. Fang CL, Huang LH, Tsai HY, Chang HI. Dermal lipogenesis inhibits adiponectin production in human dermal fibroblasts while exogenous adiponectin administration prevents against UVA-induced dermal matrix degradation in human skin. Int J Mol Sci. 2016; 17:pii:E1129.
Article14. Kim EJ, Kim YK, Kim MK, Kim S, Kim JY, Lee DH, et al. UV-induced inhibition of adipokine production in subcutaneous fat aggravates dermal matrix degradation in human skin. Sci Rep. 2016; 6:25616.
Article15. Harding CR, Aho S, Bosko CA. Filaggrin - revisited. Int J Cosmet Sci. 2013; 35:412–423.
Article16. Palmer CN, Irvine AD, Terron-Kwiatkowski A, Zhao Y, Liao H, Lee SP, et al. Common loss-of-function variants of the epidermal barrier protein filaggrin are a major predisposing factor for atopic dermatitis. Nat Genet. 2006; 38:441–446.
Article17. Thyssen JP, Kezic S. Causes of epidermal filaggrin reduction and their role in the pathogenesis of atopic dermatitis. J Allergy Clin Immunol. 2014; 134:792–799.
Article18. Jin T, Park KY, Seo SJ. Adiponectin upregulates filaggrin expression via SIRT1-mediated signaling in human normal keratinocytes. Ann Dermatol. 2017; 29:407–413.
Article19. Hansen MB, Nielsen SE, Berg K. Re-examination and further development of a precise and rapid dye method for measuring cell growth/cell kill. J Immunol Methods. 1989; 119:203–210.
Article20. Rajapakse N, Kim MM, Mendis E, Kim SK. Inhibition of inducible nitric oxide synthase and cyclooxygenase-2 in lipopolysaccharide-stimulated RAW264.7 cells by carboxybutyrylated glucosamine takes place via down-regulation of mitogen-activated protein kinase-mediated nuclear factor-kappaB signaling. Immunology. 2008; 123:348–357.
Article21. Candi E, Schmidt R, Melino G. The cornified envelope: a model of cell death in the skin. Nat Rev Mol Cell Biol. 2005; 6:328–340.
Article22. Howell MD, Kim BE, Gao P, Grant AV, Boguniewicz M, Debenedetto A, et al. Cytokine modulation of atopic dermatitis filaggrin skin expression. J Allergy Clin Immunol. 2009; 124:R7–R12.
Article23. Pellerin L, Henry J, Hsu CY, Balica S, Jean-Decoster C, Méchin MC, et al. Defects of filaggrin-like proteins in both lesional and nonlesional atopic skin. J Allergy Clin Immunol. 2013; 131:1094–1102.
Article24. Paus R, Klein J, Permana PA, Owecki M, Chaldakov GN, Böhm M, et al. What are subcutaneous adipocytes really good for...? Exp Dermatol. 2007; 16:45–47.25. Sandilands A, Sutherland C, Irvine AD, McLean WH. Filaggrin in the frontline: role in skin barrier function and disease. J Cell Sci. 2009; 122:1285–1294.
Article26. Hsu CY, Henry J, Raymond AA, Méchin MC, Pendaries V, Nassar D, et al. Deimination of human filaggrin-2 promotes its proteolysis by calpain 1. J Biol Chem. 2011; 286:23222–23233.
Article27. Wu Z, Hansmann B, Meyer-Hoffert U, Gläser R, Schröder JM. Molecular identification and expression analysis of filaggrin-2, a member of the S100 fused-type protein family. PLoS One. 2009; 4:e5227.
Article28. Kim M, Park KY, Lee MK, Jin T, Seo SJ. Adiponectin suppresses UVB-induced premature senescence and hBD2 overexpression in human keratinocytes. PLoS One. 2016; 11:e0161247.
Article29. Mehic D, Bakiri L, Ghannadan M, Wagner EF, Tschachler E. Fos and jun proteins are specifically expressed during differentiation of human keratinocytes. J Invest Dermatol. 2005; 124:212–220.
Article30. Hess J, Angel P, Schorpp-Kistner M. AP-1 subunits: quarrel and harmony among siblings. J Cell Sci. 2004; 117:5965–5973.
Article31. DiSepio D, Bickenbach JR, Longley MA, Bundman DS, Rothnagel JA, Roop DR. Characterization of loricrin regulation in vitro and in transgenic mice. Differentiation. 1999; 64:225–235.
Article32. Jang SI, Steinert PM, Markova NG. Activator protein 1 activity is involved in the regulation of the cell type-specific expression from the proximal promoter of the human profilaggrin gene. J Biol Chem. 1996; 271:24105–24114.
Article33. Ng DC, Shafaee S, Lee D, Bikle DD. Requirement of an AP-1 site in the calcium response region of the involucrin promoter. J Biol Chem. 2000; 275:24080–24088.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Adiponectin Upregulates Filaggrin Expression via SIRT1-Mediated Signaling in Human Normal Keratinocytes
- Adiponectin Promotes Caspase-14 Expression in Normal Human Epidermal Keratinocytes
- Adiponectin Attenuates the Inflammation in Atopic Dermatitis-Like Reconstructed Human Epidermis
- Immunohistochemical Study of Expression of Involucrin, Loricrin, Filaggrin and Bcl-2 in Psoriasis
- Epigenetic Regulation of Filaggrin Gene Expression in Human Epidermal Keratinocytes