J Vet Sci.
2016 Jun;17(2):159-170. 10.4142/jvs.2016.17.2.159.
Differential regulation of senescence and in vitro differentiation by 17β-estradiol between mesenchymal stem cells derived from male and female mini-pigs
- Affiliations
-
- 1College of Veterinary Medicine, Gyeongsang National University, Jinju 52828, Korea. sllee@gnu.ac.kr
- 2PWG Genetics Pte. Ltd., 15 Tech Park Crescent, Singapore 638117, Singapore.
- 3Advanced Therapy Products Research Division, National Institute of Food and Drug Safety Evaluation, Ministry of Food and Drug Safety, Cheongju 28159, Korea.
- 4Research Institute of Life Sciences, Gyeongsang National University, Jinju 52828, Korea.
- KMID: 2413165
- DOI: http://doi.org/10.4142/jvs.2016.17.2.159
Abstract
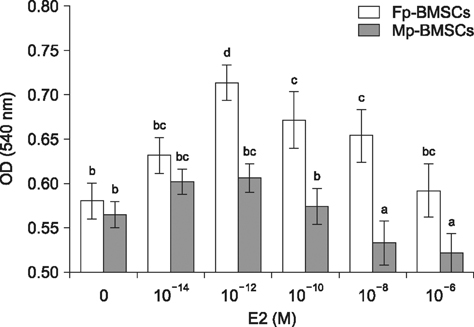
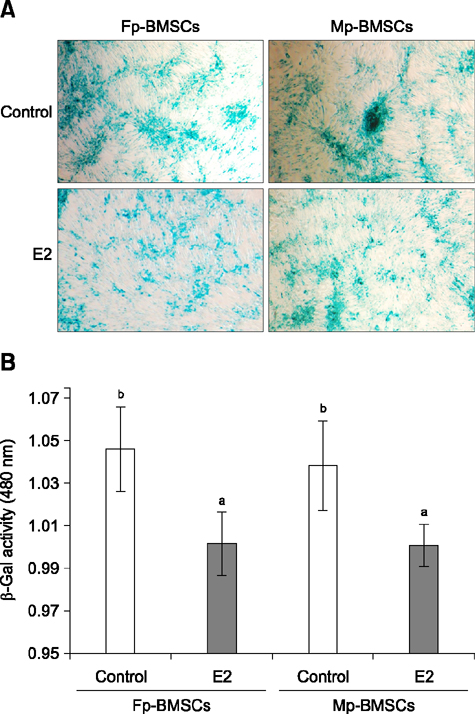
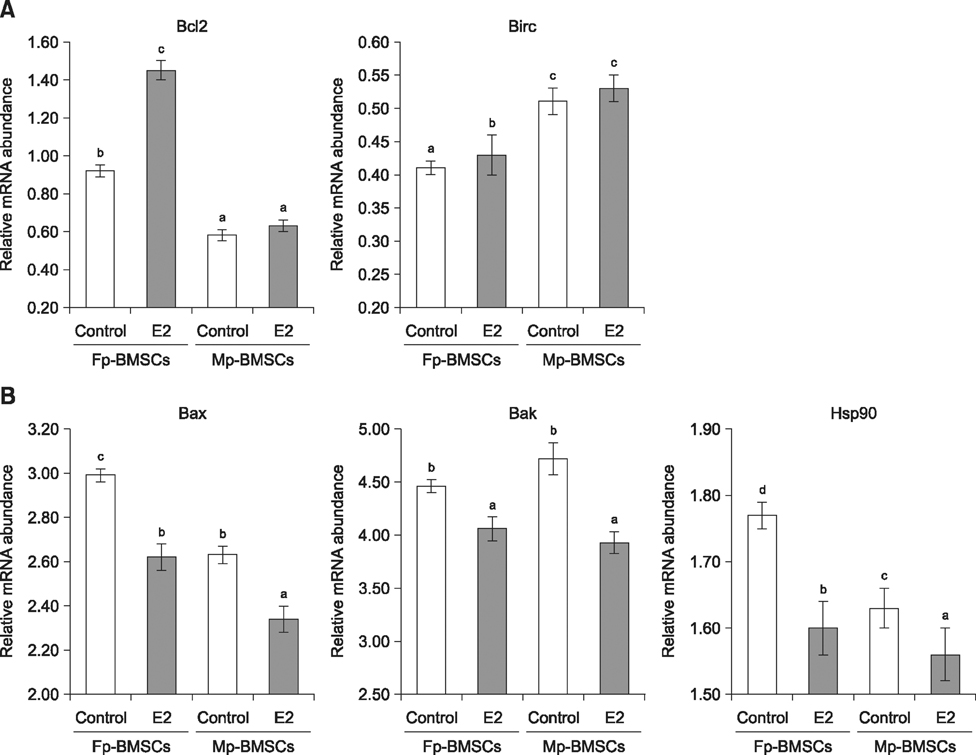
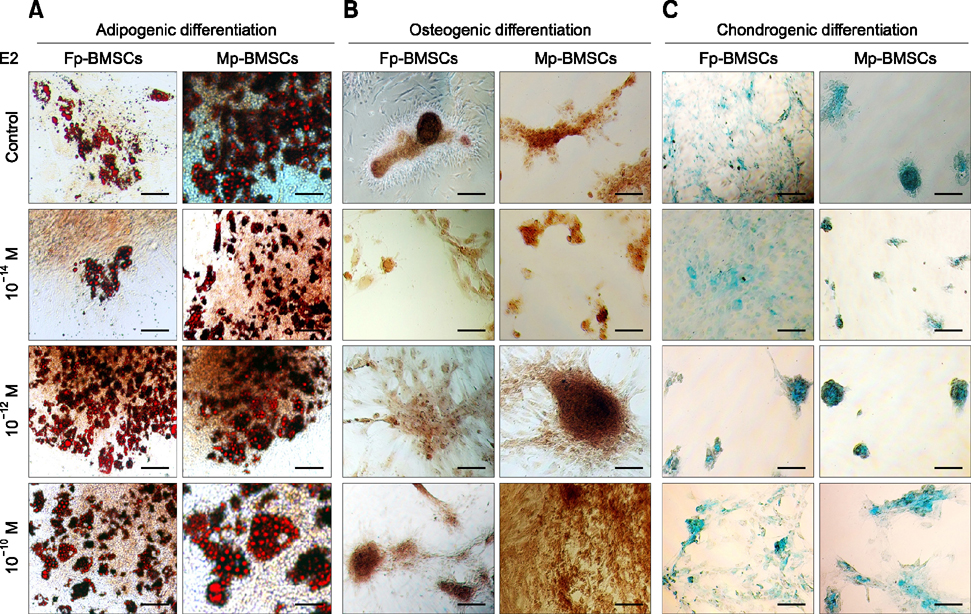
- The characterization and potential of mesenchymal stem cells (MSCs) are gender dependent and estrogen influences these properties. This study demonstrated that supplementation with 17β-estradiol (E2) increases the proliferation of bone marrow-MSCs derived from male and female mini-pigs (Mp- and Fp-BMSCs) in a concentration-dependent manner, with 10(-12) M E2 suggested as the optimal dose of E2 that led to the greatest improvement in BMSCs proliferation. Supplementation of 10(-12) M E2 resulted in down-regulation of β-galactosidase activity and pro-apoptotic activity in both BMSCs, while anti-apoptotic activity was up-regulated in only Fp-BMSCs. Further, E2 increased the osteogenic ability of Fp-BMSCs. Based on these findings, optimal utilization of E2 can improve cellular senescence and apoptosis, as well as in vitro osteogenesis of BMSCs, and could therefore be useful in stem cell therapy, particularly in bone regeneration for adult females.
MeSH Terms
Figure
Reference
-
1. Ab-Rahim S, Selvaratnam L, Kamarul T. The effect of TGFβ-1 and β-estradiol on glycosaminoglycan and type II collagen distribution in articular chondrocyte cultures. Cell Biol Int. 2008; 32:841–847.
Article2. Aksu AE, Rubin JP, Dudas JR, Marra KG. Role of gender and anatomical region on induction of osteogenic differentiation of human adipose-derived stem cells. Ann Plast Surg. 2008; 60:306–322.
Article3. Ayaloglu-Butun F, Terzioglu-Kara E, Tokcaer-Keskin Z, Akcali KC. The effect of estrogen on bone marrow-derived rat mesenchymal stem cell maintenance: inhibiting apoptosis through the expression of Bcl-xL and Bcl-2. Stem Cell Rev. 2012; 8:393–401.
Article4. Baba T, Shimizu T, Suzuki Y, Ogawara M, Isono K, Koseki H, Kurosawa H, Shirasawa T. Estrogen, insulin, and dietary signals cooperatively regulate longevity signals to enhance resistance to oxidative stress in mice. J Biol Chem. 2005; 280:16417–16426.
Article5. Claassen H, Schluter M, Schunke M, Kurz B. Influence of 17β-estradiol and insulin on type II collagen and protein synthesis of articular chondrocytes. Bone. 2006; 39:310–317.
Article6. Cooper DK, Gollackner B, Sachs DH. Will the pig solve the transplantation backlog? Annu Rev Med. 2002; 53:133–147.
Article7. Corsi KA, Pollett JB, Phillippi JA, Usas A, Li G, Huard J. Osteogenic potential of postnatal skeletal muscle-derived stem cells is influenced by donor sex. J Bone Miner Res. 2007; 22:1592–1602.
Article8. Crisostomo PR, Wang Y, Markel TA, Wang M, Lahm T, Meldrum DR. Human mesenchymal stem cells stimulated by TNF-α, LPS, or hypoxia produce growth factors by an NF-κB but not JNK-dependent mechanism. Am J Physiol Cell Physiol. 2008; 294:C675–C682.
Article9. Dang Z, Löwik CWGM. The balance between concurrent activation of ERs and PPARs determines daidzein-induced osteogenesis and adipogenesis. J Bone Miner Res. 2004; 19:853–861.
Article10. Eriksen EF, Colvard DS, Berg NJ, Graham ML, Mann KG, Spelsberg TC, Riggs BL. Evidence of estrogen receptors in normal human osteoblast-like cells. Science. 1988; 241:84–86.
Article11. Faast R, Harrison SJ, Beebe LFS, Mcilfatrick SM, Ashman RJ, Nottle MB. Use of adult mesenchymal stem cells isolated from bone marrow and blood for somatic cell nuclear transfer in pigs. Cloning Stem Cells. 2006; 8:166–173.
Article12. Fadini GP, de Kreutzenberg S, Albiero M, Coracina A, Pagnin E, Baesso I, Cignarella A, Bolego C, Plebani M, Nardelli GB, Sartore S, Agostini C, Avogaro A. Gender differences in endothelial progenitor cells and cardiovascular risk profile: the role of female estrogens. Arterioscler Thromb Vasc Biol. 2008; 28:997–1004.
Article13. Guo M, Hay BA. Cell proliferation and apoptosis. Curr Opin Cell Biol. 1999; 11:745–752.
Article14. Heim M, Frank O, Kampmann G, Sochocky N, Pennimpede T, Fuchs P, Hunziker W, Weber P, Martin I, Bendik I. The phytoestrogen genistein enhances osteogenesis and represses adipogenic differentiation of human primary bone marrow stromal cells. Endocrinology. 2004; 145:848–859.
Article15. Hong L, Colpan A, Peptan IA. Modulations of 17-β estradiol on osteogenic and adipogenic differentiations of human mesenchymal stem cells. Tissue Eng. 2006; 12:2747–2753.
Article16. Hong L, Sultana H, Paulius K, Zhang G. Steroid regulation of proliferation and osteogenic differentiation of bone marrow stromal cells: a gender difference. J Steroid Biochem Mol Biol. 2009; 114:180–185.
Article17. Jaiswal N, Haynesworth SE, Caplan AI, Bruder SP. Osteogenic differentiation of purified, culture expanded human mesenchymal stem cells in vitro. J Cell Biochem. 1997; 64:295–312.
Article18. Jenei-Lanzl Z, Straub RH, Dienstknecht T, Huber M, Hager M, Grässel S, Kujat R, Angele MK, Nerlich M, Angele P. Estradiol inhibits chondrogenic differentiation of mesenchymal stem cells via nonclassic signaling. Arthritis Rheum. 2010; 62:1088–1096.
Article19. Jog NR, Caricchio R. Differential regulation of cell death programs in males and females by Poly (ADP-ribose) polymerase-1 and 17β estradiol. Cell Death Dis. 2013; 4:e758.20. Johnstone B, Hering TM, Caplan AI, Goldberg VM, Yoo JU. In vitro chondrogenesis of bone marrow-derived mesenchymal progenitor cells. Exp Cell Res. 1998; 238:265–272.
Article21. Kluck RM, Bossy-Wetzel E, Green DR, Newmeyer DD. The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science. 1997; 275:1132–1136.
Article22. Lee SL, Kang EJ, Maeng GH, Kim MJ, Park JK, Kim TS, Hyun SH, Lee ES, Rho GJ. Developmental ability of miniature pig embryos cloned with mesenchymal stem cells. J Reprod Dev. 2010; 56:256–262.
Article23. Lee WJ, Hah YS, Ock SA, Lee JH, Jeon RH, Park JS, Lee SI, Rho NY, Rho GJ, Lee SL. Cell source-dependent in vivo immunosuppressive properties of mesenchymal stem cells derived from the bone marrow and synovial fluid of minipigs. Exp Cell Res. 2015; 333:273–288.
Article24. Lindsten T, Ross AJ, King A, Zong WX, Rathmell JC, Shiels HA, Ulrich E, Waymire KG, Mahar P, Frauwirth K, Chen Y, Wei M, Eng VM, Adelman DM, Simon MC, Ma A, Golden JA, Evan G, Korsmeyer SJ, MacGregor GR, Thompson CB. The combined functions of proapoptotic Bcl-2 family members Bak and Bax are essential for normal development of multiple tissues. Mol Cell. 2000; 6:1389–1399.
Article25. Nejadnik H, Hui JH, Feng Choong EP, Tai BC, Lee EH. Autologous bone marrow-derived mesenchymal stem cells versus autologous chondrocyte implantation: an observational cohort study. Am J Sports Med. 2010; 38:1110–1116.
Article26. Parker D, Hwa SY, Sambrook P, Ghosh P. Estrogen replacement therapy mitigates the loss of joint cartilage proteoglycans and bone mineral density induced by ovariectomy and osteoarthritis. APLAR J Rheumatol. 2003; 6:116–127.
Article27. Qu Q, Perälä-Heape M, Kapanen A, Dahllund J, Salo J, Väänänen HK, Härkänen P. Estrogen enhances differentiation of osteoblasts in mouse bone marrow culture. Bone. 1998; 22:201–209.
Article28. Sachs DH. The pig as a potential xenograft donor. Vet Immunol Immunopathol. 1994; 43:185–191.
Article29. Shukla D, Box GN, Edwards RA, Tyson DR. Bone marrow stem cells for urologic tissue engineering. World J Urol. 2008; 26:341–349.
Article30. Su J, Chen X, Huang Y, Li W, Li J, Cao K, Cao G, Zhang L, Li F, Roberts AI, Kang H, Yu P, Ren G, Ji W, Wang Y, Shi Y. Phylogenetic distinction of iNOS and IDO function in mesenchymal stem cell-mediated immunosuppression in mammalian species. Cell Death Differ. 2014; 21:388–396.
Article31. Syed F, Khosla S. Mechanisms of sex steroid effects on bone. Biochem Biophys Res Commun. 2005; 328:688–696.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Nervonic Acid Inhibits Replicative Senescence of Human Wharton’s Jelly-Derived Mesenchymal Stem Cells
- Characterization of Senescence of Culture-expanded Human Adipose-derived Mesenchymal Stem Cells
- Concise Review: Differentiation of Human Adult Stem Cells Into Hepatocyte-like Cells In vitro
- Differential Potential of Stem Cells Following Their Origin: Subacromial Bursa, Bone Marrow, Umbilical Cord Blood
- Cross-talk between BubR1 expression and the commitment to differentiate in adipose-derived mesenchymal stem cells