Int J Stem Cells.
2017 Nov;10(2):218-226. 10.15283/ijsc17003.
Hepatogenic Differentiation Capacity of Human Wharton’s Jelly Mesenchymal Stem Cell in a Co-culturing System with Endothelial Cells in Matrigel/collagen Scaffold in the Presence of Fetal Liver Extract
- Affiliations
-
- 1Laboratory for Stem Cell Research, Department of Anatomy, School of Medicine, Shiraz University of Medical Sciences, Shiraz, Iran. vojdaniz@sums.ac.ir
- 2Stem Cell Technology Research Center, Shiraz Institute for Stem Cell and Regenerative Medicine, Shiraz University of Medical Sciences, Shiraz, Iran.
- 3Tissue Engineering Lab, Department of Anatomy, School of Medicine, Shiraz University of Medical Sciences, Shiraz, Iran.
- KMID: 2400875
- DOI: http://doi.org/10.15283/ijsc17003
Abstract
- BACKGROUND
Human Wharton's jelly mesenchymal stem cells (HWJMSCs) isolated from medical waste product can be considered as an accessible source of cells in regenerative medicine. Stem cell-derived hepatocytes have poor function and need appropriate niche to reconstruct the liver structure. Therefore, we attempted to find a novel approach in differentiating HWJMSCs into functional hepatic cells using 3D culture conditions and liver extract that recapitulates vital stage in liver development.
MATERIALS AND METHODS
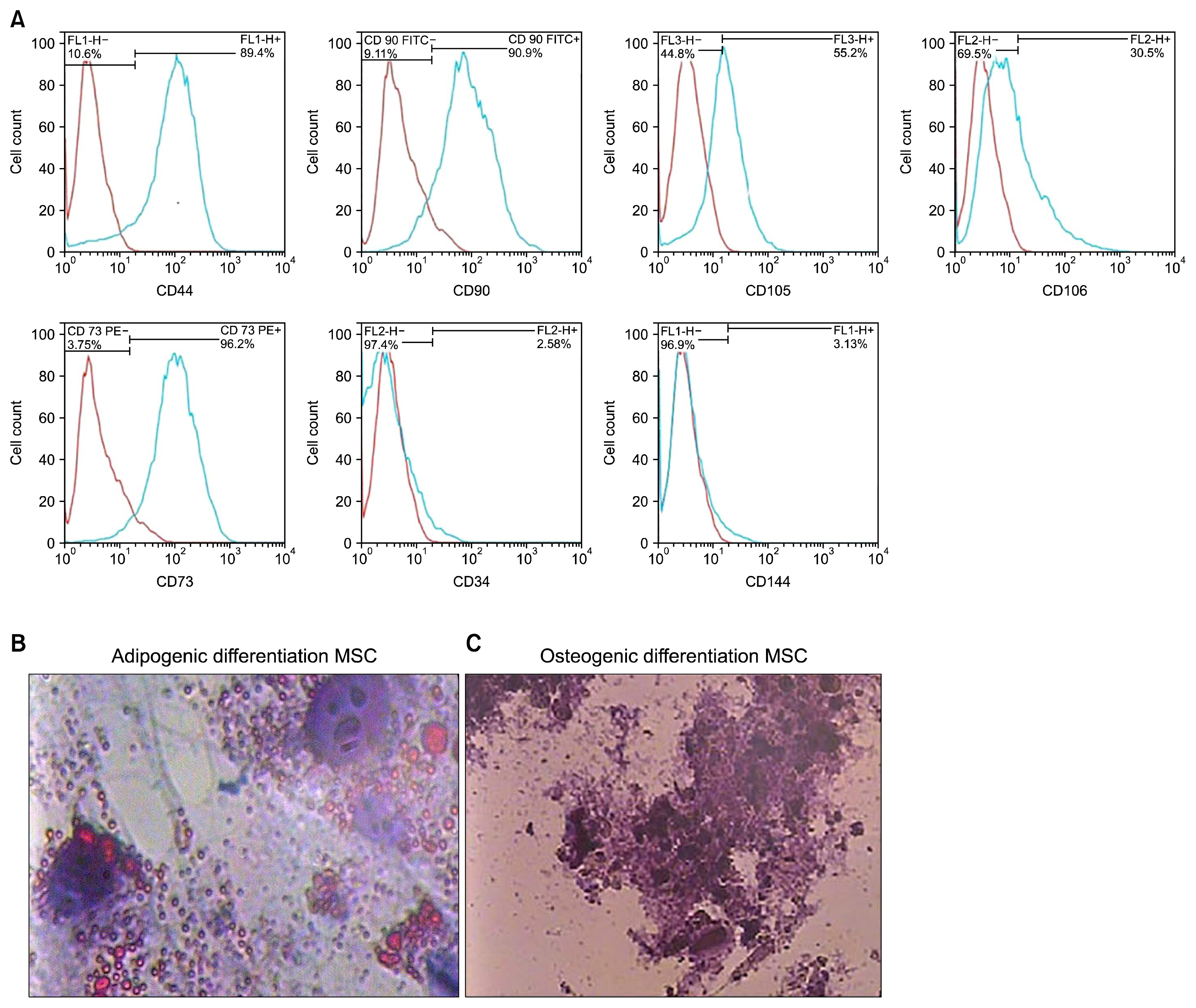
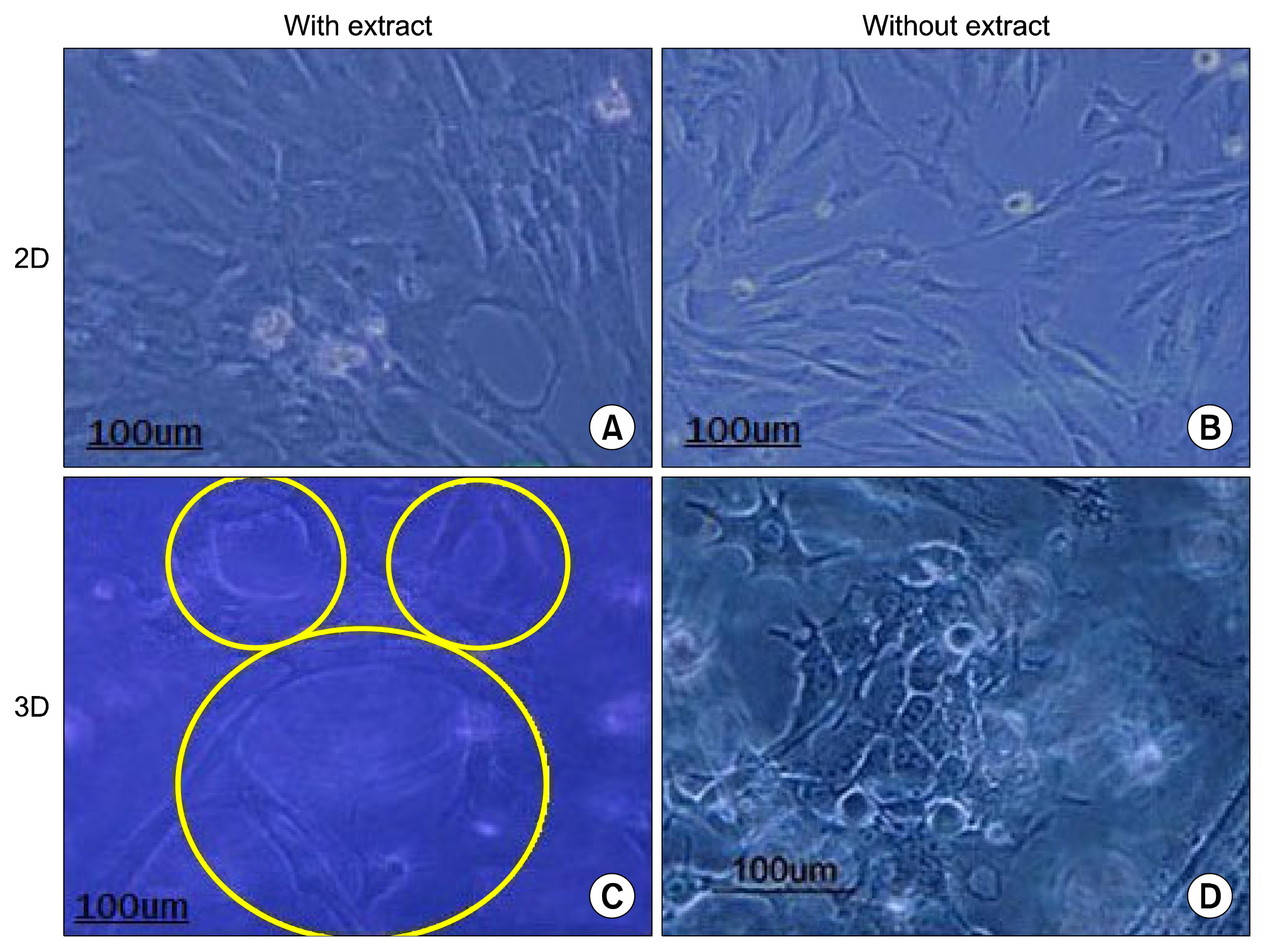
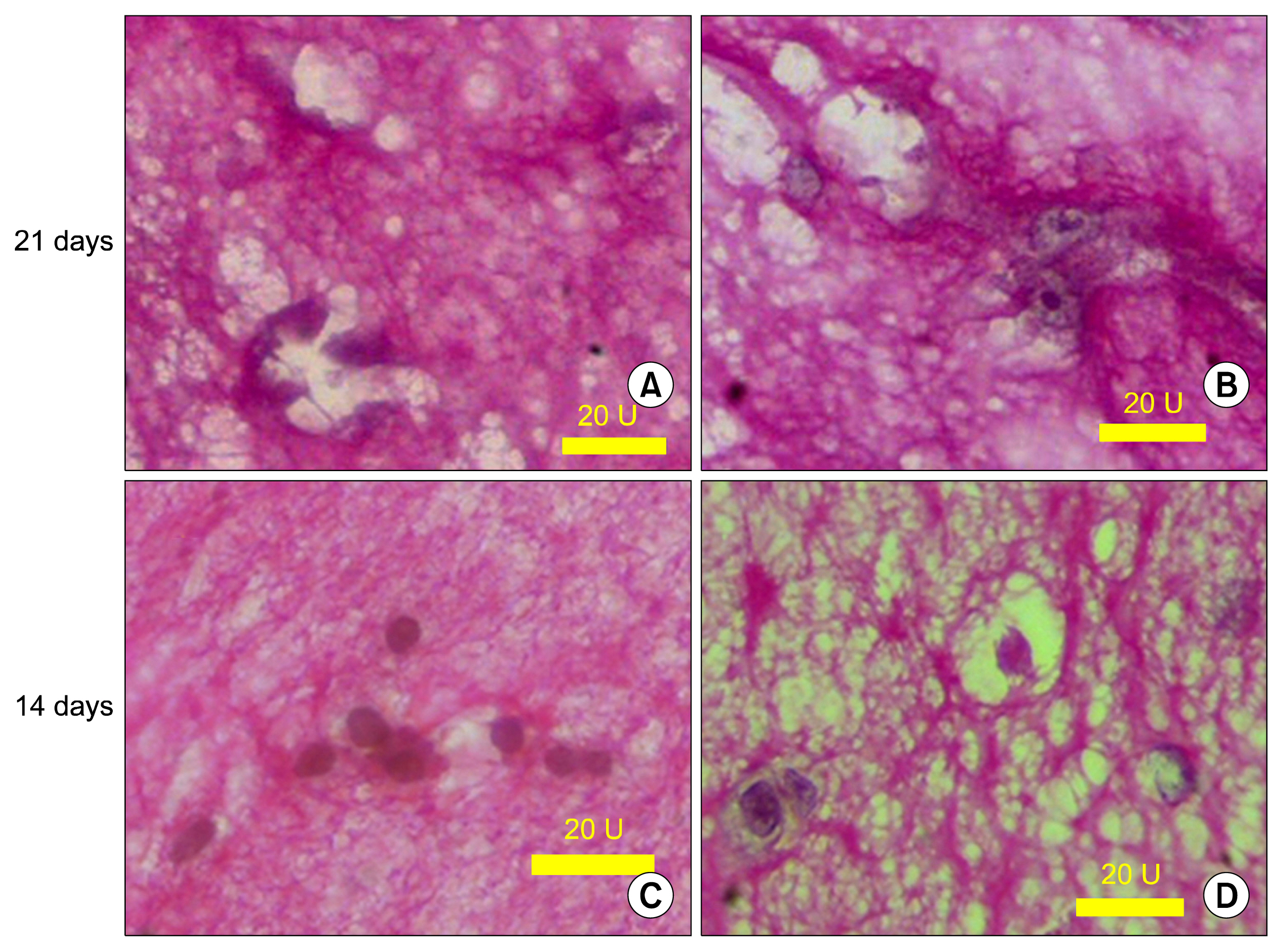
HWJMSCs were extracted from human Wharton's jelly, characterized by flow cytometry, and differentiated towards osteogenic and adipogenic lineages. HWJMSCs were co-cultured with HUVECs in 3D matrigel/collagen scaffolds in the presence of fetal liver extract for 14 days. The expression of specific liver genes were evaluated by lectins, PAS and immunocytochemistry.
RESULTS
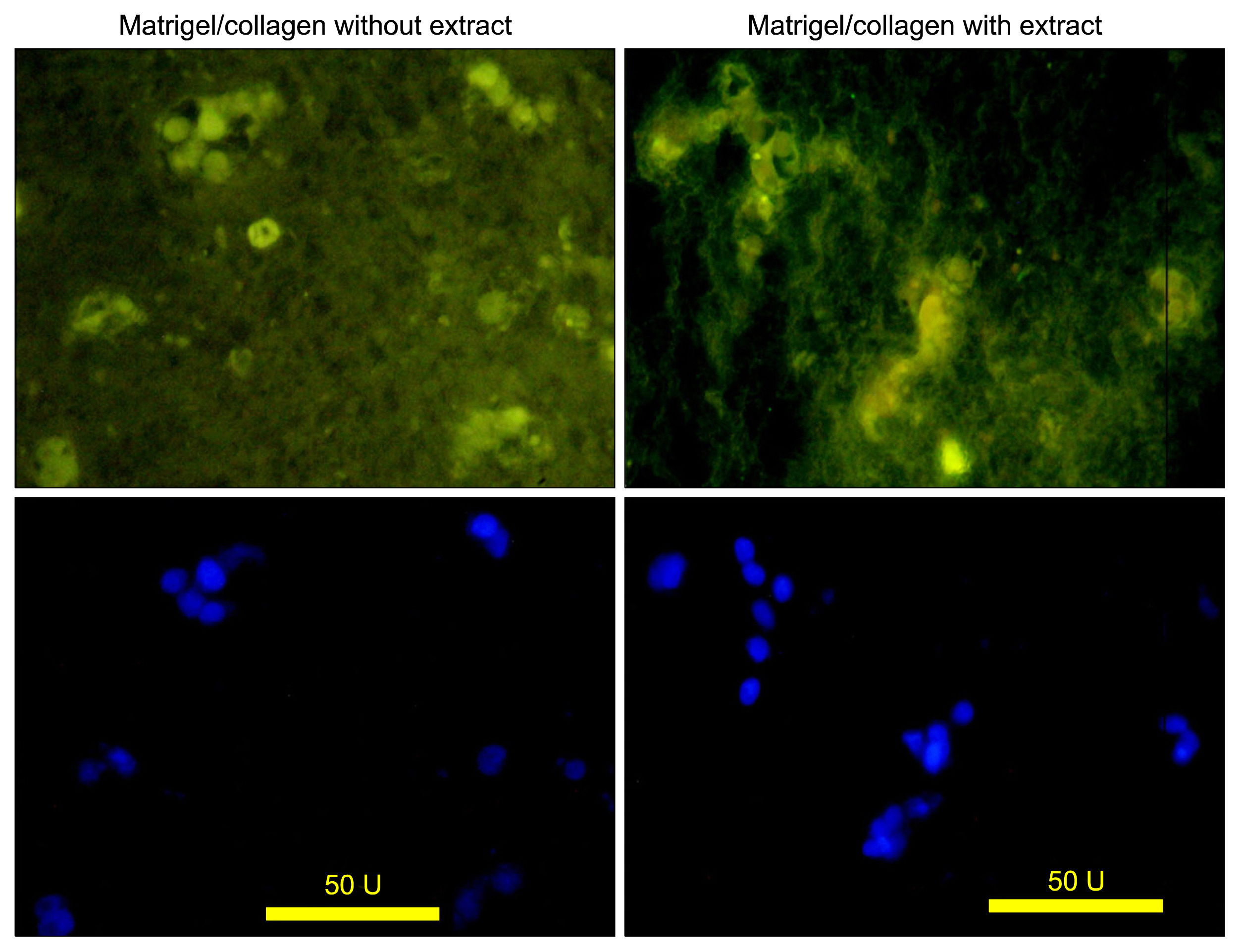
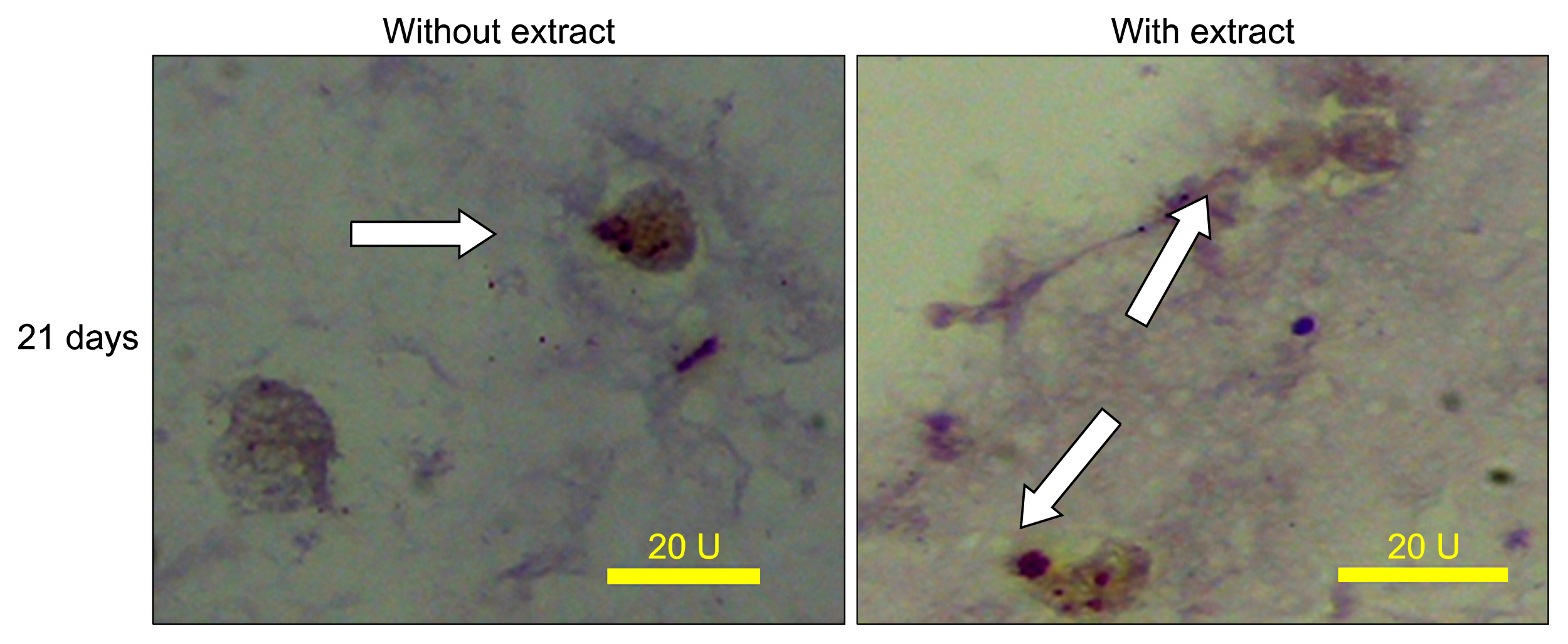
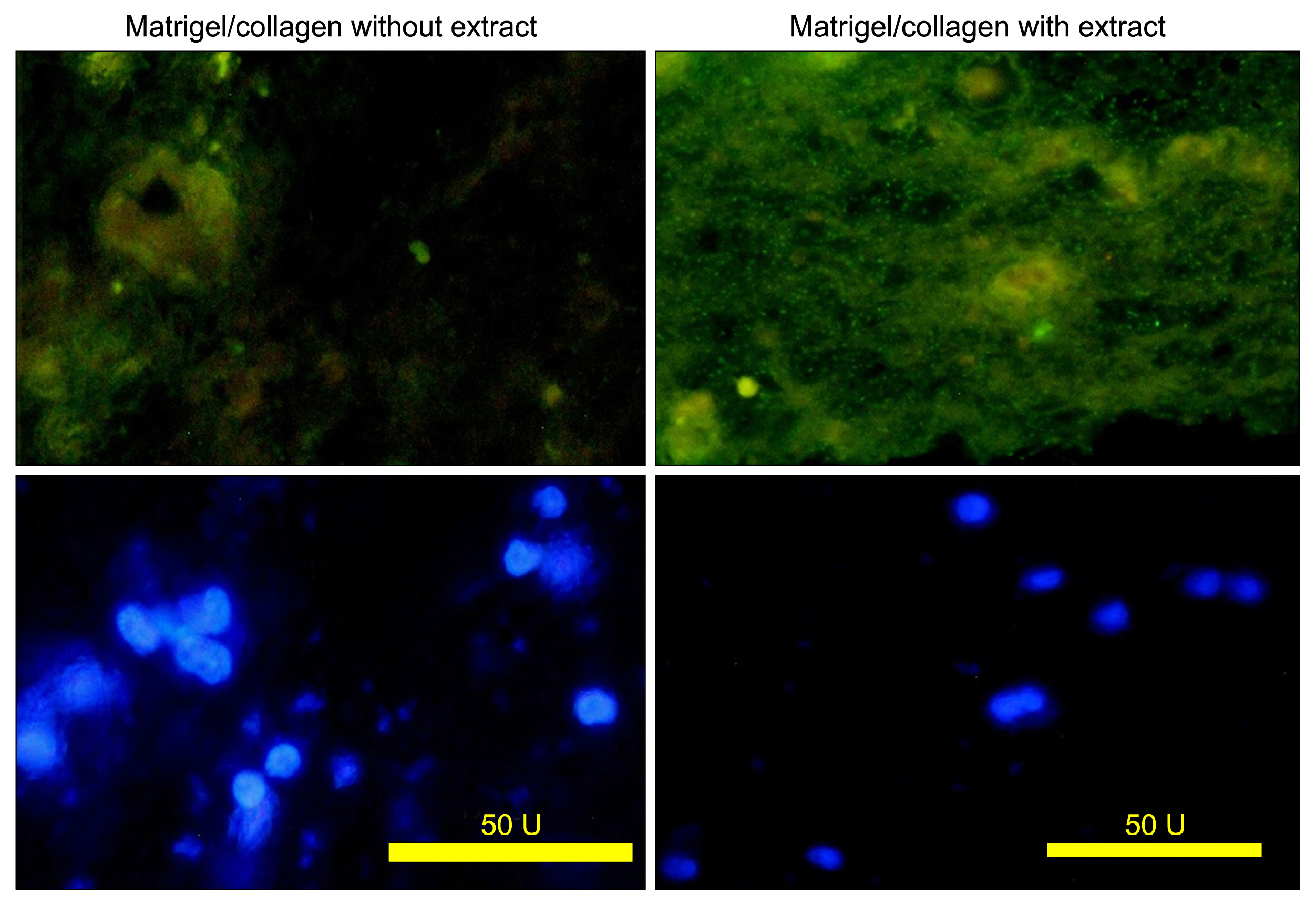
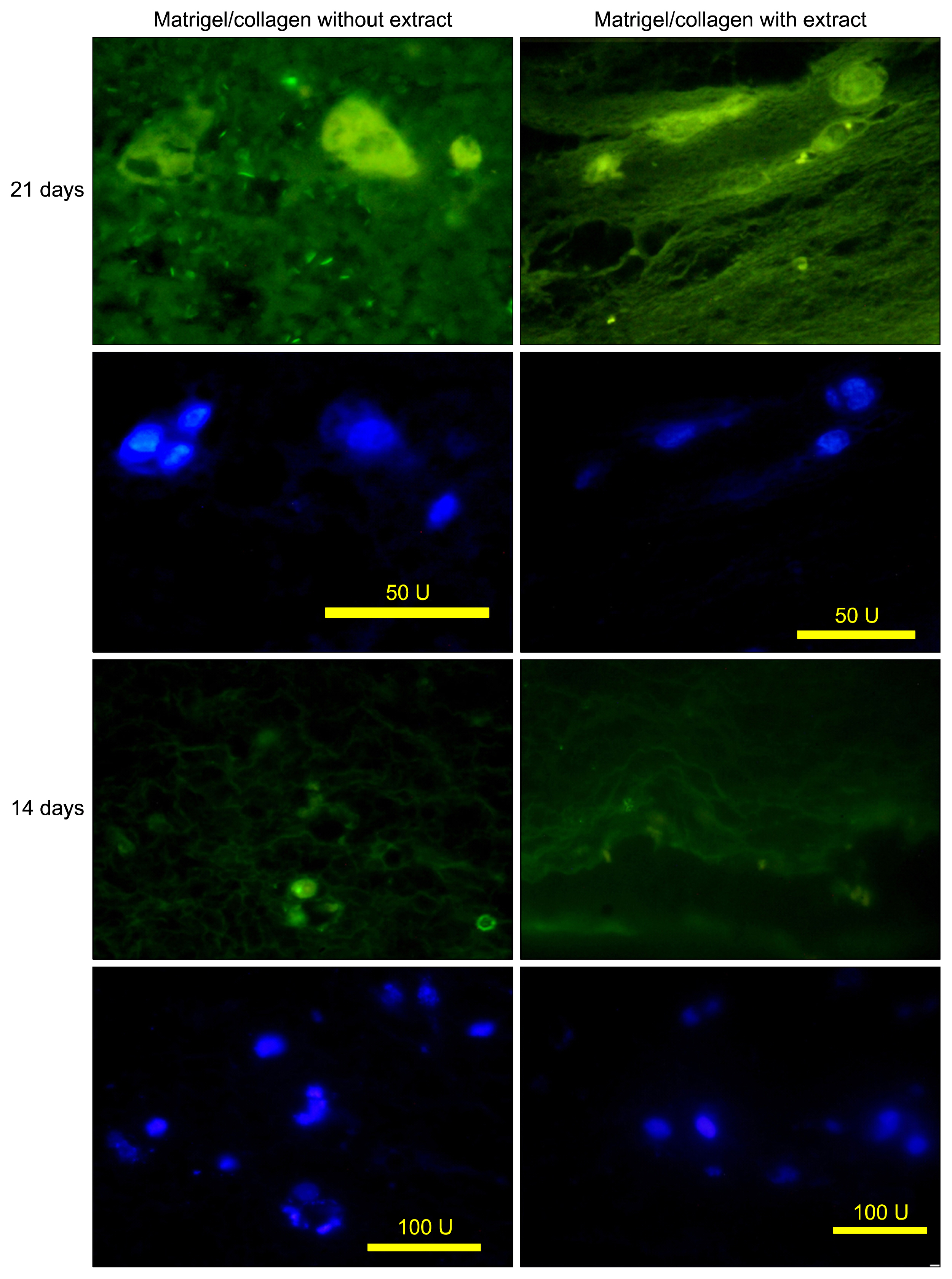
According to flow cytometry data, isolated cells from HWJMSCs were shown to express MSC markers. HWJMSCs co-cultured with HUVECs in matrigel/collagen scaffold with extract expressed albumin, lectins UEA and PNA. Immunohistochemistry of the cells in matrigel/collagen scaffold with or without extract exhibited a positive reaction for CK19.
CONCLUSIONS
Co-culturing of the HWJMSC/HUVEC in 3D matrigel/collagen scaffold is bimimicary of in vivo cell condition. The results showed that administration of the liver extract in 3D matrigel/collagen culture of HWJMSC/HUVEC can induce hepatocyte marker expression.
Keyword
MeSH Terms
Figure
Reference
-
References
1. Gaudio E, Carpino G, Cardinale V, Franchitto A, Onori P, Alvaro D. New insights into liver stem cells. Dig Liver Dis. 2009; 41:455–462. DOI: 10.1016/j.dld.2009.03.009. PMID: 19403350.
Article2. Sharma R, Greenhough S, Medine CN, Hay DC. Three-dimensional culture of human embryonic stem cell derived hepatic endoderm and its role in bioartificial liver construction. J Biomed Biotechnol. 2010; DOI: 10.1155/2010/236147. PMID: 20169088. PMCID: 2821762.
Article3. Broutier L, Andersson-Rolf A, Hindley CJ, Boj SF, Clevers H, Koo BK, Huch M. Culture and establishment of self-renewing human and mouse adult liver and pancreas 3D organoids and their genetic manipulation. Nat Protoc. 2016; 11:1724–1743. DOI: 10.1038/nprot.2016.097. PMID: 27560176.
Article4. Handa K, Matsubara K, Fukumitsu K, Guzman-Lepe J, Watson A, Soto-Gutierrez A. Assembly of human organs from stem cells to study liver disease. Am J Pathol. 2014; 184:348–357. DOI: 10.1016/j.ajpath.2013.11.003. PMCID: 3906514.
Article5. Wang ZZ, Au P, Chen T, Shao Y, Daheron LM, Bai H, Arzigian M, Fukumura D, Jain RK, Scadden DT. Endothelial cells derived from human embryonic stem cells form durable blood vessels in vivo. Nat Biotechnol. 2007; 25:317–318. DOI: 10.1038/nbt1287. PMID: 17322871.
Article6. Zwicky C, Gerber S, Gasparini D, Forestier F, Hohlfeld P, Tissot JD, Schneider P. Preparation and analysis of fetal liver extracts. Bone Marrow Transplant. 2000; 26:667–671. DOI: 10.1038/sj.bmt.1702564. PMID: 11035373.
Article7. Kang XQ, Zang WJ, Bao LJ, Li DL, Song TS, Xu XL, Yu XJ. Fibroblast growth factor-4 and hepatocyte growth factor induce differentiation of human umbilical cord blood-derived mesenchymal stem cells into hepatocytes. World J Gastroenterol. 2005; 11:7461–7465. DOI: 10.3748/wjg.v11.i47.7461.
Article8. Sellaro TL, Ranade A, Faulk DM, McCabe GP, Dorko K, Badylak SF, Strom SC. Maintenance of human hepatocyte function in vitro by liver-derived extracellular matrix gels. Tissue Eng Part A. 2010; 16:1075–1082. DOI: 10.1089/ten.tea.2008.0587. PMCID: 2863084.
Article9. Dawson E, Mapili G, Erickson K, Taqvi S, Roy K. Biomaterials for stem cell differentiation. Adv Drug Deliv Rev. 2008; 60:215–228. DOI: 10.1016/j.addr.2007.08.037.
Article10. Sodek KL, Brown TJ, Ringuette MJ. Collagen I but not Matrigel matrices provide an MMP-dependent barrier to ovarian cancer cell penetration. BMC Cancer. 2008; 8:223. DOI: 10.1186/1471-2407-8-223. PMID: 18681958. PMCID: 2519089.
Article11. Reed J, Walczak WJ, Petzold ON, Gimzewski JK. In situ mechanical interferometry of matrigel films. Langmuir. 2009; 25:36–39. DOI: 10.1021/la8033098.
Article12. Machado CB, Ventura JM, Lemos AF, Ferreira JM, Leite MF, Goes AM. 3D chitosan-gelatin-chondroitin porous scaffold improves osteogenic differentiation of mesenchymal stem cells. Biomed Mater. 2007; 2:124–131. DOI: 10.1088/1748-6041/2/2/010.
Article13. Liu H, Roy K. Biomimetic three-dimensional cultures significantly increase hematopoietic differentiation efficacy of embryonic stem cells. Tissue Eng. 2005; 11:319–330. DOI: 10.1089/ten.2005.11.319. PMID: 15738685.
Article14. Dunn JC, Yarmush ML, Koebe HG, Tompkins RG. Hepatocyte function and extracellular matrix geometry: long-term culture in a sandwich configuration. FASEB J. 1989; 3:174–177. PMID: 2914628.
Article15. Anzalone R, Lo Iacono M, Loria T, Di Stefano A, Giannuzzi P, Farina F, La Rocca G. Wharton’s jelly mesenchymal stem cells as candidates for beta cells regeneration: extending the differentiative and immunomodulatory benefits of adult mesenchymal stem cells for the treatment of type 1 diabetes. Stem Cell Rev. 2011; 7:342–363. DOI: 10.1007/s12015-010-9196-4.
Article16. Vojdani Z, Khodabandeh Z, Jaberipour M, Hosseini A, Bahmanpour S, Talaei-Khozani T. The influence of fibroblast growth factor 4 on hepatogenic capacity of Wharton’s jelly mesenchymal stromal cells. Rom J Morphol Embryol. 2015; 56:1043–1050.17. LeCouter J, Moritz DR, Li B, Phillips GL, Liang XH, Gerber HP, Hillan KJ, Ferrara N. Angiogenesis-independent endothelial protection of liver: role of VEGFR-1. Science. 2003; 299:890–893. DOI: 10.1126/science.1079562. PMID: 12574630.
Article18. Si-Tayeb K, Lemaigre FP, Duncan SA. Organogenesis and development of the liver. Dev Cell. 2010; 18:175–189. DOI: 10.1016/j.devcel.2010.01.011. PMID: 20159590.
Article19. Krause S, Maffini MV, Soto AM, Sonnenschein C. A novel 3D in vitro culture model to study stromal-epithelial interactions in the mammary gland. Tissue Eng Part C Methods. 2008; 14:261–271. DOI: 10.1089/ten.tec.2008.0030. PMID: 18694322.
Article20. Li L, Lu Y. Optimizing a 3D culture system to study the interaction between epithelial breast cancer and its surrounding fibroblasts. J Cancer. 2011; 2:458–466. DOI: 10.7150/jca.2.458. PMID: 21915190. PMCID: 3171896.
Article21. Dan YY, Yeoh GC. Liver stem cells: a scientific and clinical perspective. J Gastroenterol Hepatol. 2008; 23:687–698. DOI: 10.1111/j.1440-1746.2008.05383.x. PMID: 18410603.
Article22. Nagai H, Terada K, Watanabe G, Ueno Y, Aiba N, Shibuya T, Kawagoe M, Kameda T, Sato M, Senoo H, Sugiyama T. Differentiation of liver epithelial (stem-like) cells into hepatocytes induced by coculture with hepatic stellate cells. Biochem Biophys Res Commun. 2002; 293:1420–1425. DOI: 10.1016/S0006-291X(02)00406-0. PMID: 12054673.
Article23. Chen L, Chen XP, Zhang W, Liang HF, Lin YZ, Dong HH, Zhou QD. Differentiation of hepatic oval cell into mature hepatocyte induced by hepatic stellate cells. Zhonghua Gan Zang Bing Za Zhi. 2009; 17:765–770. PMID: 19874693.24. Lange C, Bruns H, Kluth D, Zander AR, Fiegel HC. Hepatocytic differentiation of mesenchymal stem cells in cocultures with fetal liver cells. World J Gastroenterol. 2006; 12:2394–2397. DOI: 10.3748/wjg.v12.i15.2394. PMID: 16688831. PMCID: 4088076.
Article25. Kleinman HK, Martin GR. Matrigel: basement membrane matrix with biological activity in Seminars in cancer biology. Elsevier;2005.26. Indramanee S, Silsirivanit A, Pairojkul C, Wongkham C, Wongkham S. Aberrant glycosylation in cholangiocarcinoma demonstrated by lectin-histochemistry. Asian Pac J Cancer Prev. 2012; 13(Suppl):119–124. PMID: 23480752.27. Donovan D, Brown NJ, Bishop ET, Lewis CE. Comparison of three in vitro human ‘angiogenesis’ assays with capillaries formed in vivo. Angiogenesis. 2001; 4:113–121. DOI: 10.1023/A:1012218401036.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Heparin/Collagen 3D Scaffold Accelerates Hepatocyte Differentiation of Wharton's Jelly-Derived Mesenchymal Stem Cells
- Comparative Evaluation for Potential Differentiation of Endothelial Progenitor Cells and Mesenchymal Stem Cells into Endothelial-Like Cells
- Fibrin Scaffolds Designing in order to Human Adipose-derived Mesenchymal Stem Cells Differentiation to Chondrocytes in the Presence of TGF-beta3
- Hepatogenic differentiation of human mesenchymal stem cells from peritoneal adipose tissue
- Endothelial progenitor cells and mesenchymal stem cells from human cord blood