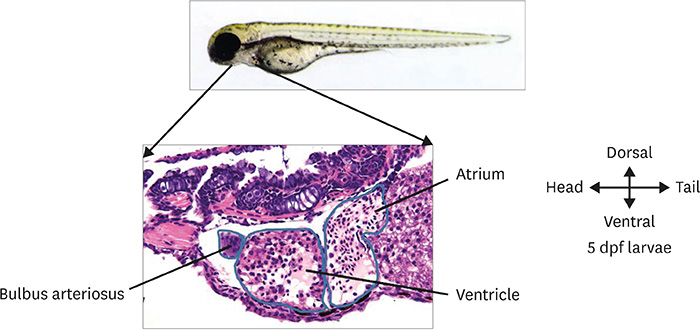
Korean Circ J.
2017 Nov;47(6):960-969. 10.4070/kcj.2017.0080.
Zebrafish Larvae Model of Dilated Cardiomyopathy Induced by Terfenadine
- Affiliations
-
- 1Department of Microbiology and Immunology, Institute of Endemic Disease, Seoul National University College of Medicine, Seoul, Korea. hylee612@snu.ac.kr lamseok@snu.ac.kr
- 2Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea. hylee612@snu.ac.kr lamseok@snu.ac.kr
- KMID: 2396489
- DOI: http://doi.org/10.4070/kcj.2017.0080
Abstract
- BACKGROUND AND OBJECTIVES
Dilated cardiomyopathy can be the end-stage of severe cardiac disorders and directly affects the cardiac muscle, inducing cardiomegaly and heart failure (HF). Although a wide variety of animal models are available to study dilated cardiomyopathy, there is no model to assess dilated cardiomyopathy with non-invasive, simple, and large screening methods.
METHODS
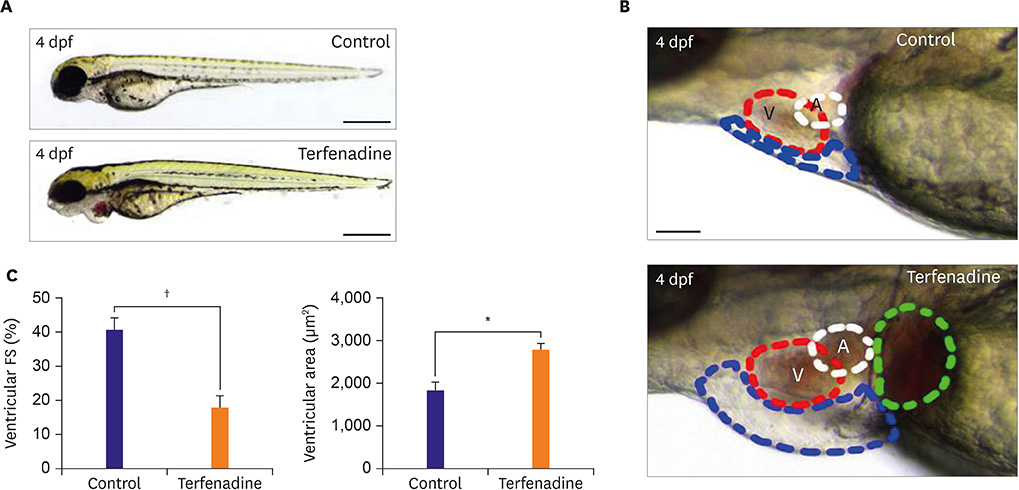
We developed a dilated cardiomyopathy model in zebrafish larvae using short duration terfenadine, a known cardiotoxic drug that induces ventricular size dilation. Fractional shortening of zebrafish hearts was calculated.
RESULTS
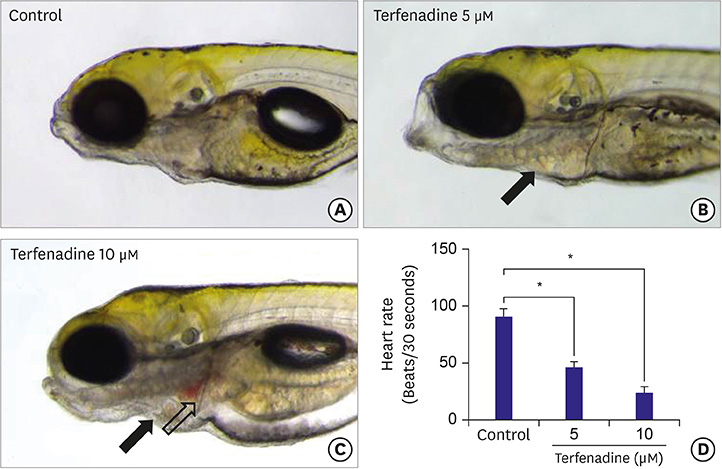
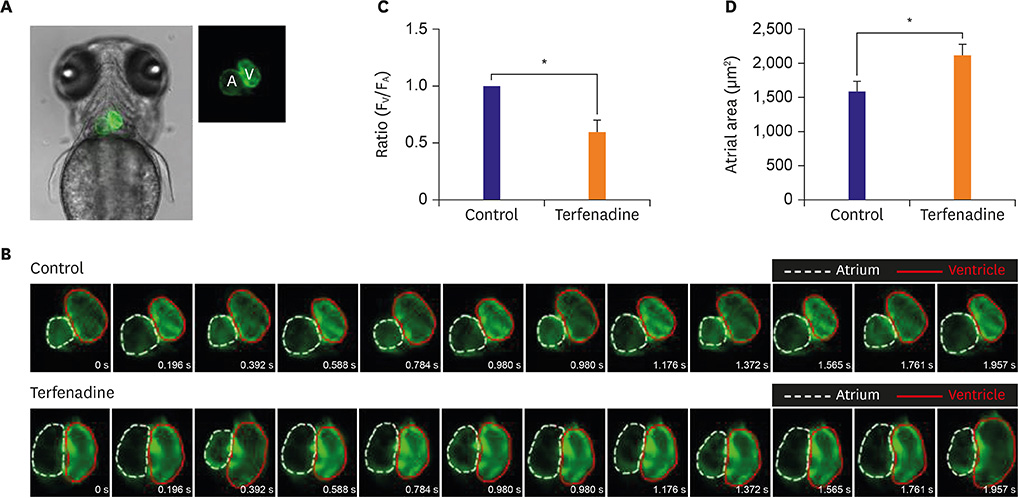
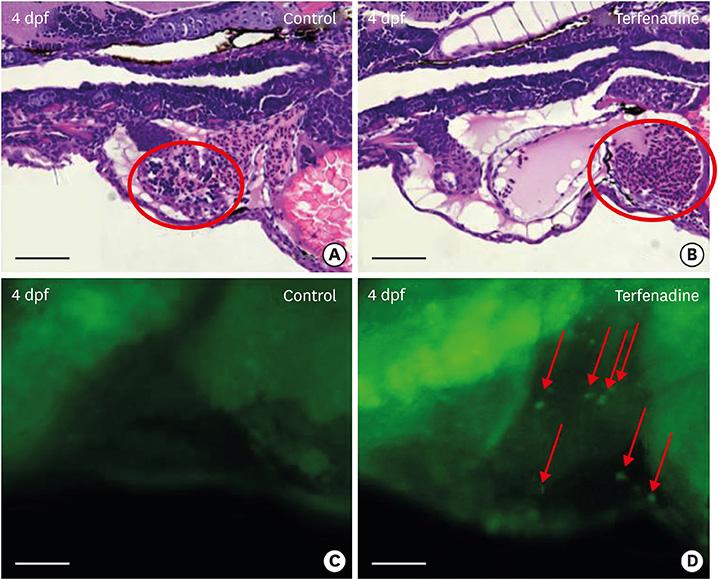
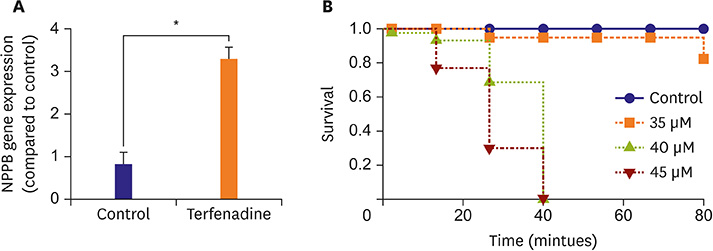
We treated zebrafish with 5 to 10 µM terfenadine for 24 hours. In terfenadine-treated zebrafish, blood frequently pooled and clotted in the chamber, and circulation was remarkably reduced. Atria and ventricles were swollen, and fluid was deposited around the heart, mimicking edema. Cardiac contractility was significantly reduced, and ventricular area was significantly enlarged. Heart rate was markedly reduced even after terfenadine withdrawal. Acridine orange staining also showed that terfenadine increased cardiomyocyte apoptosis. A significant increase of natriuretic peptide B (NPPB) mRNA was found in terfenadine-treated zebrafish. A low dose of terfenadine (5-10 µM) did not show mortality in short-term treatment (24 hours). However, moderate dose (35-45 µM) terfenadine treatment reduced zebrafish survival within 1 hour.
CONCLUSION
With advantages of rapid sample preparation procedure and transparent observation of the live heart, this model can potentially be applied to large-scale drug screening and toxicity assays for non-ischemic HF.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
A Small Animal Model of Diabetic Heart Failure With Reduced Ejection Fraction
Seung-Hyun Jung, Hyun-Taek Kim
Korean Circ J. 2023;53(1):47-48. doi: 10.4070/kcj.2022.0349.
Reference
-
1. Tang C, Xie D, Feng B. Zebrafish as a new model for phenotype-based screening of positive inotropic agents. Chem Biol Drug Des. 2015; 85:253–258.2. Huang CC, Monte A, Cook JM, Kabir MS, Peterson KP. Zebrafish heart failure models for the evaluation of chemical probes and drugs. Assay Drug Dev Technol. 2013; 11:561–572.3. Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC)Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2016; 37:2129–2200.4. Lee SE, Cho HJ, Lee HY, et al. A multicentre cohort study of acute heart failure syndromes in Korea: rationale, design, and interim observations of the Korean Acute Heart Failure (KorAHF) registry. Eur J Heart Fail. 2014; 16:700–708.5. Report of the WHO/ISFC task force on the definition and classification of cardiomyopathies. Br Heart J. 1980; 44:672–673.6. Beltrami CA, Finato N, Rocco M, et al. Structural basis of end-stage failure in ischemic cardiomyopathy in humans. Circulation. 1994; 89:151–163.7. Du CK, Morimoto S, Nishii K, et al. Knock-in mouse model of dilated cardiomyopathy caused by troponin mutation. Circ Res. 2007; 101:185–194.8. Mahmoudabady M, Niazmand S, Shafei MN, McEntee K. Investigation of apoptosis in a canine model of chronic heart failure induced by tachycardia. Acta Physiol Hung. 2013; 100:435–444.9. Tanaka Y, Rahmutula D, Duggirala S, et al. Diffuse fibrosis leads to a decrease in unipolar voltage: validation in a swine model of premature ventricular contraction-induced cardiomyopathy. Heart Rhythm. 2016; 13:547–554.10. Gava FN, Zacché E, Ortiz EM, et al. Doxorubicin induced dilated cardiomyopathy in a rabbit model: an update. Res Vet Sci. 2013; 94:115–121.11. Robert J. Long-term and short-term models for studying anthracycline cardiotoxicity and protectors. Cardiovasc Toxicol. 2007; 7:135–139.12. Teerlink JR, Pfeffer JM, Pfeffer MA. Progressive ventricular remodeling in response to diffuse isoproterenol-induced myocardial necrosis in rats. Circ Res. 1994; 75:105–113.13. Shih YH, Zhang Y, Ding Y, et al. Cardiac transcriptome and dilated cardiomyopathy genes in zebrafish. Circ Cardiovasc Genet. 2015; 8:261–269.14. Bakkers J. Zebrafish as a model to study cardiac development and human cardiac disease. Cardiovasc Res. 2011; 91:279–288.15. Jung DW, Oh ES, Park SH, et al. A novel zebrafish human tumor xenograft model validated for anti-cancer drug screening. Mol Biosyst. 2012; 8:1930–1939.16. Asnani A, Peterson RT. The zebrafish as a tool to identify novel therapies for human cardiovascular disease. Dis Model Mech. 2014; 7:763–767.17. Zünkler BJ, Kühne S, Rustenbeck I, Ott T. Mechanism of terfenadine block of ATP-sensitive K(+) channels. Br J Pharmacol. 2000; 130:1571–1574.18. Hove-Madsen L, Llach A, Molina CE, et al. The proarrhythmic antihistaminic drug terfenadine increases spontaneous calcium release in human atrial myocytes. Eur J Pharmacol. 2006; 553:215–221.19. Fearnley CJ, Roderick HL, Bootman MD. Calcium signaling in cardiac myocytes. Cold Spring Harb Perspect Biol. 2011; 3:a004242.20. Huang CJ, Tu CT, Hsiao CD, Hsieh FJ, Tsai HJ. Germ-line transmission of a myocardium-specific GFP transgene reveals critical regulatory elements in the cardiac myosin light chain 2 promoter of zebrafish. Dev Dyn. 2003; 228:30–40.21. Paquet D, Bhat R, Sydow A, et al. A zebrafish model of tauopathy allows in vivo imaging of neuronal cell death and drug evaluation. J Clin Invest. 2009; 119:1382–1395.22. Milan DJ, Peterson TA, Ruskin JN, Peterson RT, MacRae CA. Drugs that induce repolarization abnormalities cause bradycardia in zebrafish. Circulation. 2003; 107:1355–1358.23. Kooij V, Venkatraman V, Tra J, et al. Sizing up models of heart failure: proteomics from flies to humans. Proteomics Clin Appl. 2014; 8:653–664.24. Tsang M. Zebrafish: a tool for chemical screens. Birth Defects Res C Embryo Today. 2010; 90:185–192.25. Peterson RT, Link BA, Dowling JE, Schreiber SL. Small molecule developmental screens reveal the logic and timing of vertebrate development. Proc Natl Acad Sci USA. 2000; 97:12965–12969.26. Schoenebeck JJ, Yelon D. Illuminating cardiac development: advances in imaging add new dimensions to the utility of zebrafish genetics. Semin Cell Dev Biol. 2007; 18:27–35.27. Bühler A, Kustermann M, Bummer T, Rottbauer W, Sandri M, Just S. Atrogin-1 deficiency leads to myopathy and heart failure in zebrafish. Int J Mol Sci. 2016; 17:E187.28. Cui G, Chen H, Cui W, et al. FGF2 prevents sunitinib-induced cardiotoxicity in zebrafish and cardiomyoblast H9c2 cells. Cardiovasc Toxicol. 2016; 16:46–53.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Development of a Zebrafish Larvae Model for Diabetic Heart Failure With Reduced Ejection Fraction
- Fimasartan, an angiotensin II receptor antagonist, ameliorates an in vivo zebrafish model of heart failure
- Zebrafish and Mycobacterial infection
- A Case of Hypocalcemia-Induced Dilated Cardiomyopathy
- Possibility of Zebrafish as New infection model for Leprosy