J Rheum Dis.
2017 Oct;24(5):271-278. 10.4078/jrd.2017.24.5.271.
Interleukin-17 Enhances Germinal Center Formation and Immunoglobulin G1 Production in Mice
- Affiliations
-
- 1The Rheumatism Research Center, Catholic Research Institute of Medical Science, The Catholic University of Korea, Seoul, Korea. seungki73@catholic.ac.kr
- 2Division of Rheumatology, Department of Internal Medicine, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- KMID: 2394342
- DOI: http://doi.org/10.4078/jrd.2017.24.5.271
Abstract
OBJECTIVE
Interleukin (IL)-17 is a pro-inflammatory cytokine that has pleiotropic effects on multiple target cells and thereby contributes to the development of immune-mediated inflammatory disorders. However, the role of IL-17 in the humoral immune response has not been clearly elucidated.
METHODS
Mice deficient in IL-17A (IL-17A knockout [KO] mice) and wild type (WT) C57BL/6 mice were compared. Distinct B cell (mature/precursor and marginal zone/follicular) and plasma cell populations were compared using fluorescence-activated cell sorting (FACS) and confocal immunostaining. Immunoglobulin production was assessed by enzyme-linked immunosorbent assay.
RESULTS
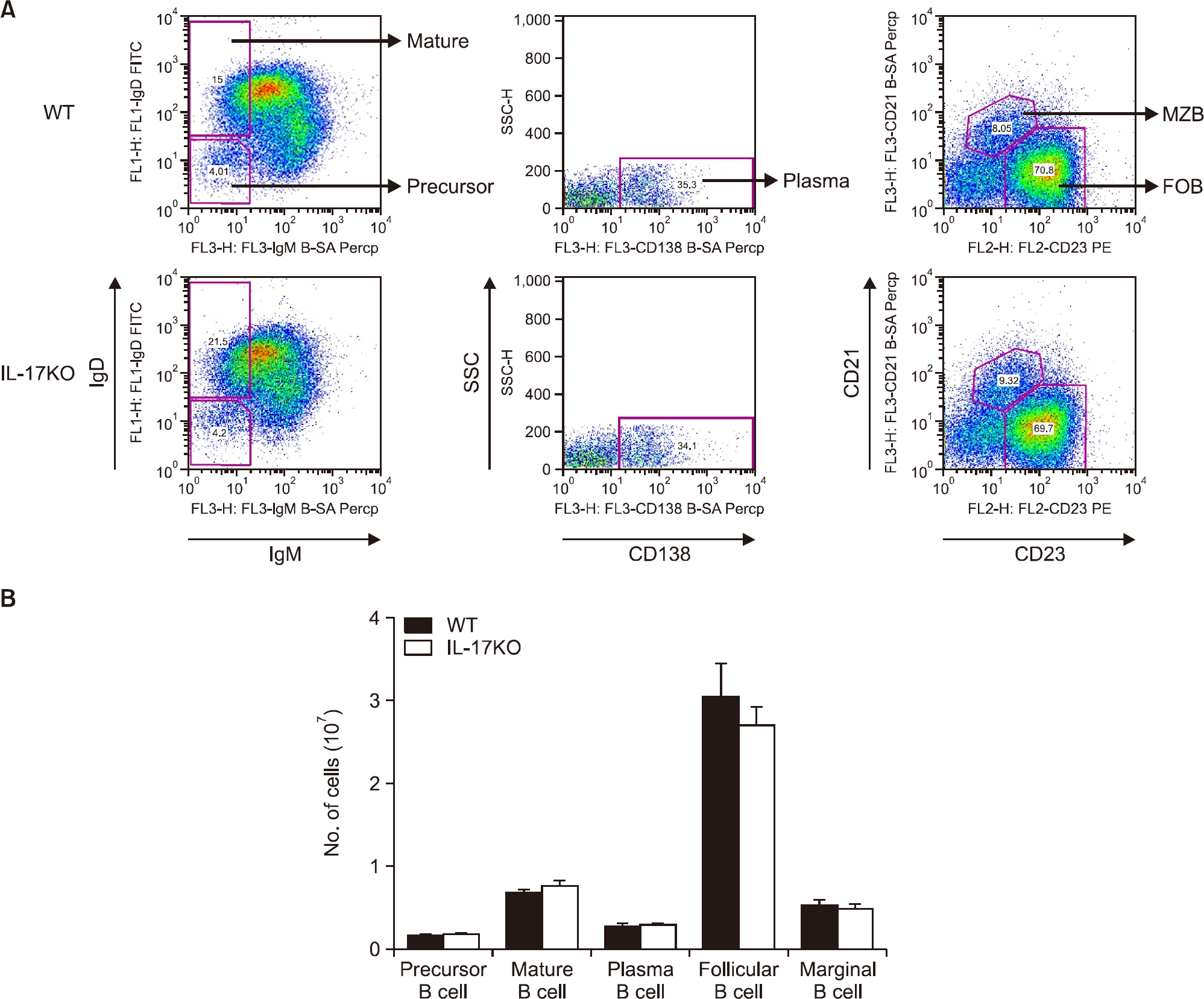
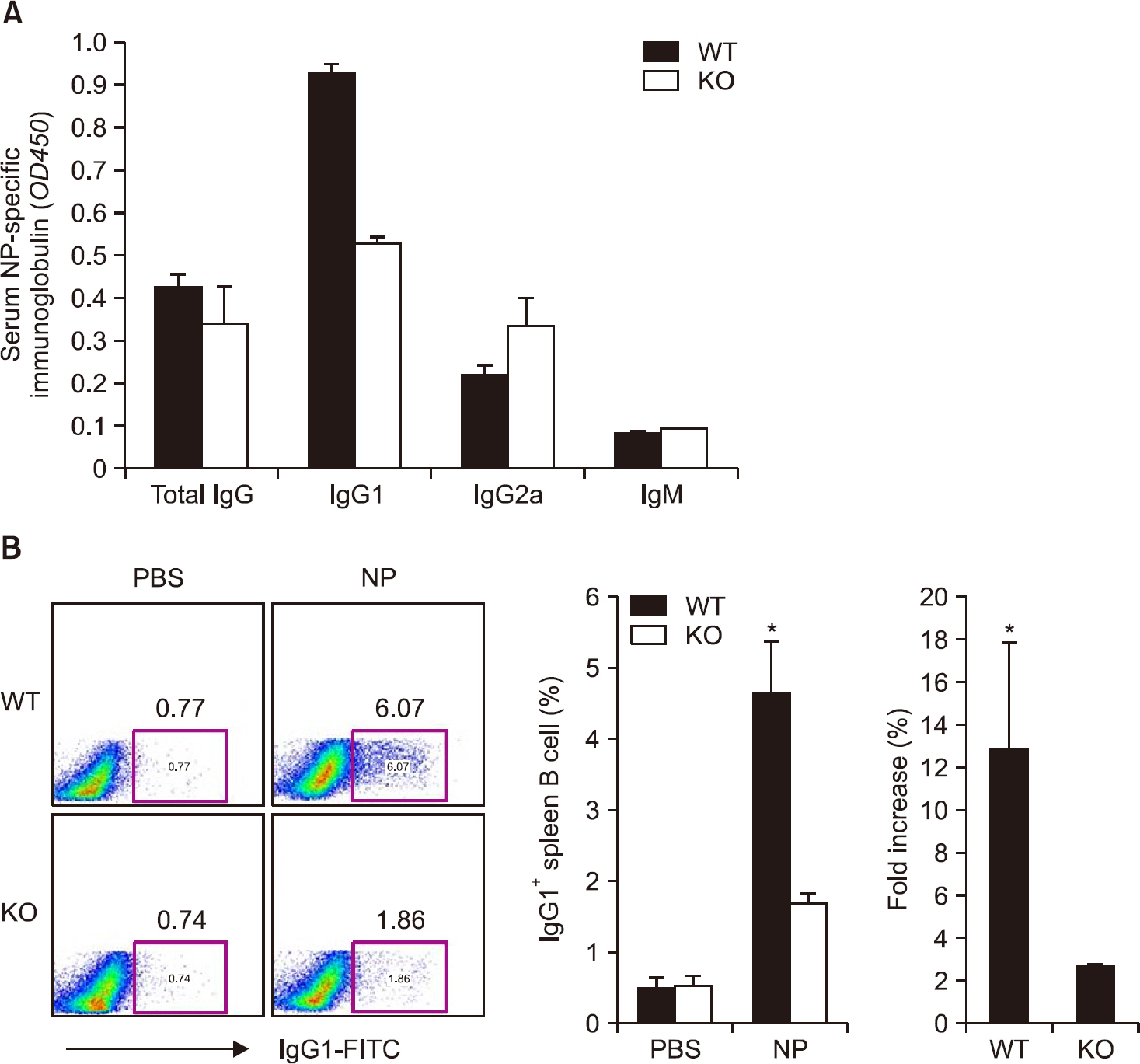
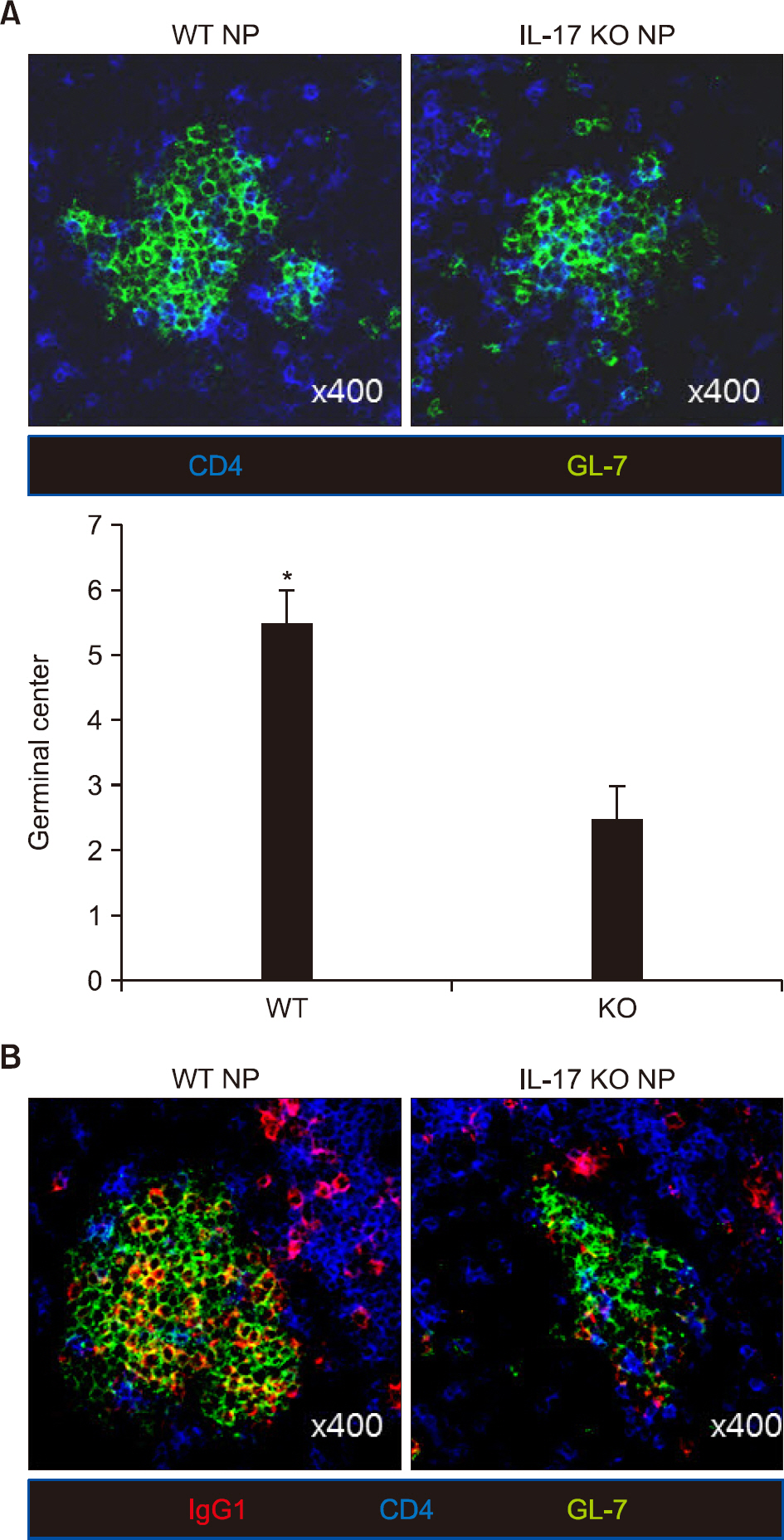
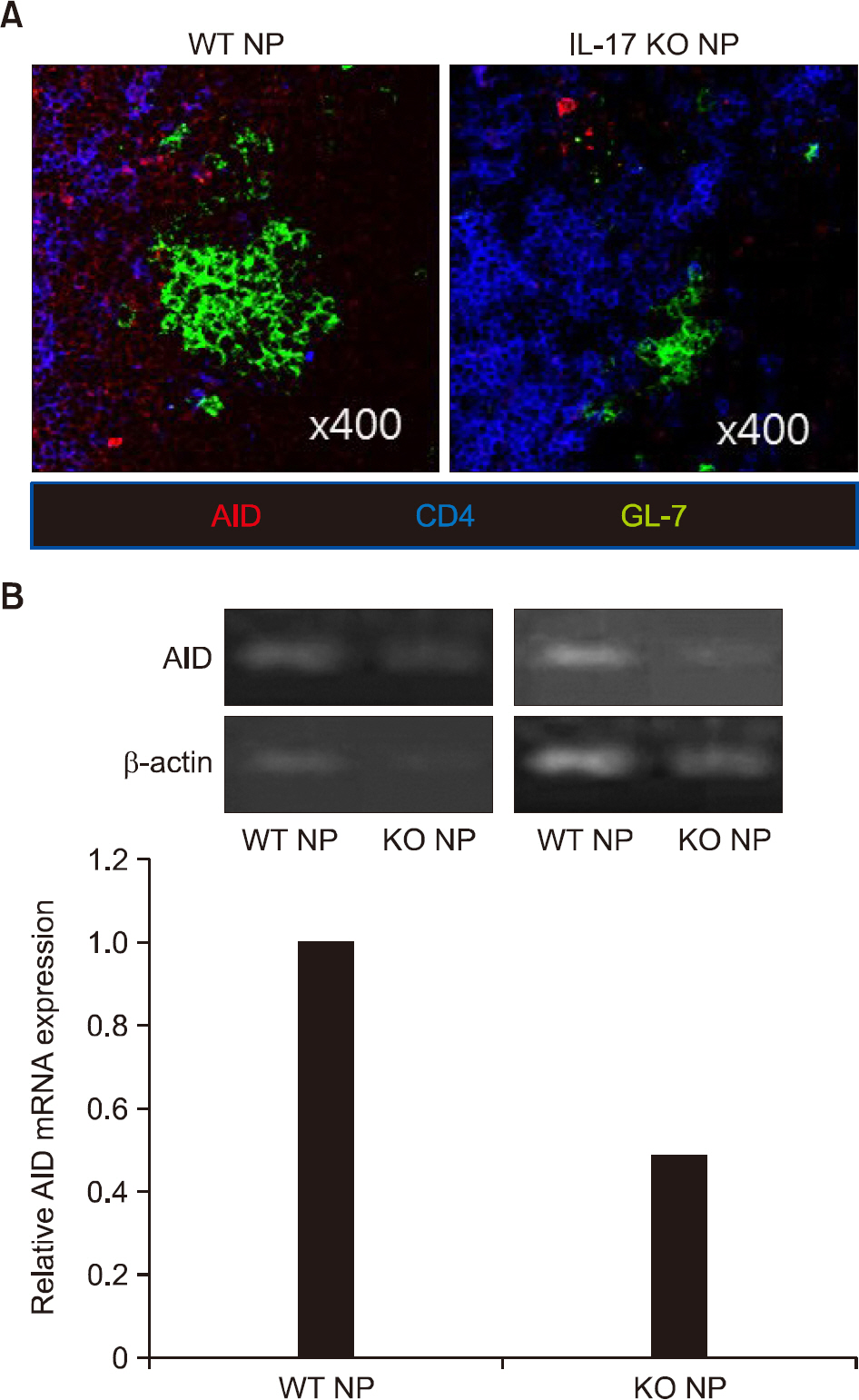
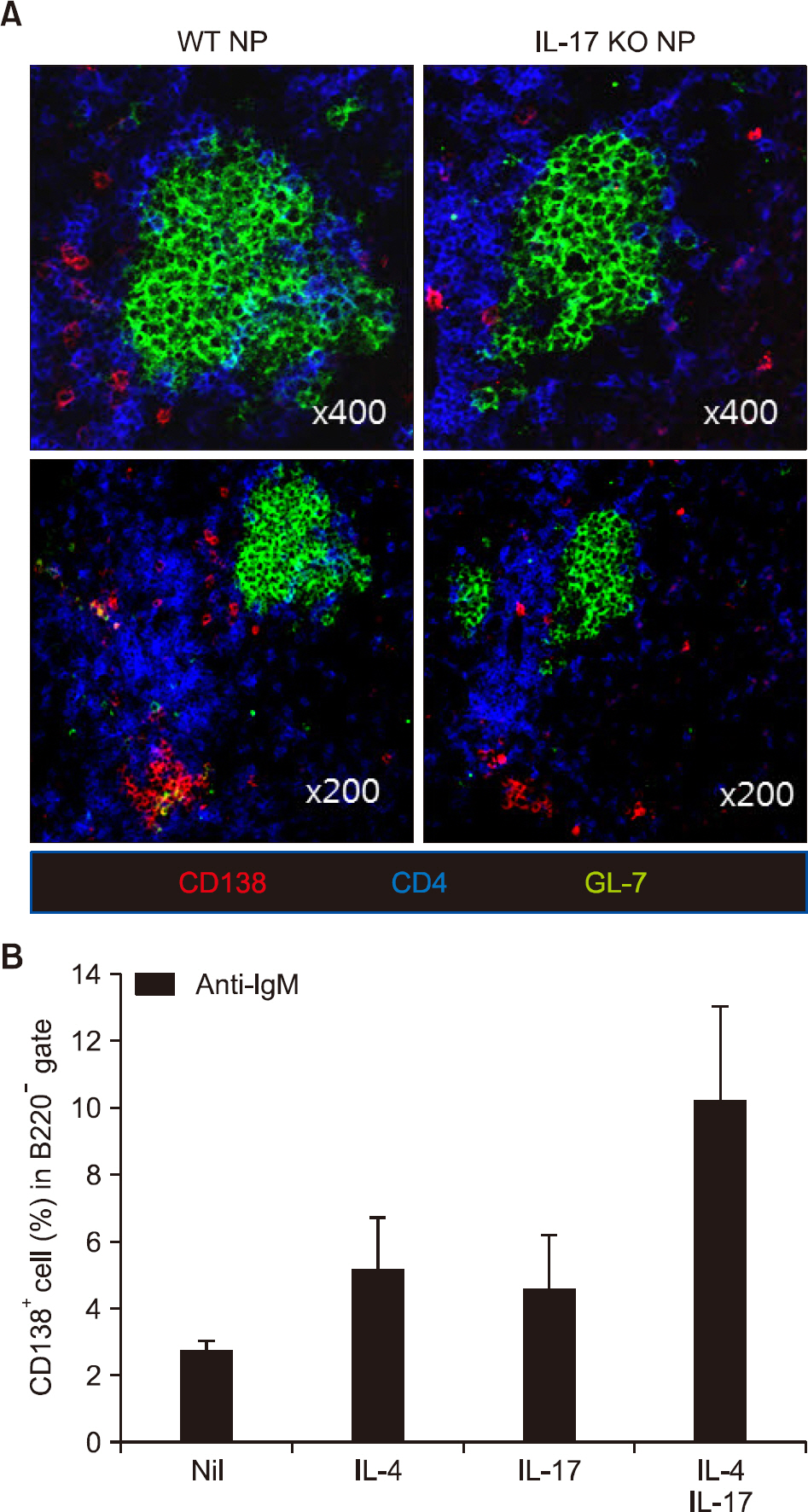
There was no difference in B cell and plasma cell populations between IL-17A KO and WT mice. However, after T cell-dependent antigen challenge, IL-17A KO mice produced lower levels of immunoglobulin (Ig)G1 than wild-type animals. IL-17A KO mice also showed reduced germinal center (GC) formation and lower expression of activation-induced cytidine deaminase, the essential enzyme for class switch recombination (CSR). IL-17 had no effect on the proliferation or survival of naïve B cells in in vitro functional studies. However, IL-17 treatment promoted naïve B cell differentiation into plasma cells in synergy with IL-4, although IL-17 alone had no effect.
CONCLUSION
Our findings suggest that IL-17 contributes to the humoral immune response by enhancing GC formation, CSR to IgG1, and plasma cell differentiation in synergy with IL-4.
Keyword
MeSH Terms
-
Animals
B-Lymphocytes
Cell Differentiation
Cytidine Deaminase
Enzyme-Linked Immunosorbent Assay
Flow Cytometry
Germinal Center*
Immunity, Humoral
Immunoglobulin G
Immunoglobulins*
In Vitro Techniques
Interleukin-17*
Interleukin-4
Interleukins
Mice*
Plasma Cells
Recombination, Genetic
Cytidine Deaminase
Immunoglobulin G
Immunoglobulins
Interleukin-17
Interleukin-4
Interleukins
Figure
Reference
-
1. Jin W, Dong C. IL-17 cytokines in immunity and inflammation. Emerg Microbes Infect. 2013; 2:e60.
Article2. McInnes IB, Schett G. The pathogenesis of rheumatoid arthritis. N Engl J Med. 2011; 365:2205–19.
Article3. Raychaudhuri SP. Role of IL-17 in psoriasis and psoriatic arthritis. Clin Rev Allergy Immunol. 2013; 44:183–93.
Article4. Matusevicius D, Kivisäkk P, He B, Kostulas N, Ozenci V, Fredrikson S, et al. Interleukin-17 mRNA expression in blood and CSF mononuclear cells is augmented in multiple sclerosis. Mult Scler. 1999; 5:101–4.
Article5. Lock C, Hermans G, Pedotti R, Brendolan A, Schadt E, Garren H, et al. Gene-microarray analysis of multiple sclerosis lesions yields new targets validated in autoimmune encephalomyelitis. Nat Med. 2002; 8:500–8.
Article6. Nakae S, Nambu A, Sudo K, Iwakura Y. Suppression of immune induction of collagen-induced arthritis in IL-17-defi-cient mice. J Immunol. 2003; 171:6173–7.
Article7. Komiyama Y, Nakae S, Matsuki T, Nambu A, Ishigame H, Kakuta S, et al. IL-17 plays an important role in the development of experimental autoimmune encephalomyelitis. J Immunol. 2006; 177:566–73.
Article8. Patel DD, Lee DM, Kolbinger F, Antoni C. Effect of IL-17A blockade with secukinumab in autoimmune diseases. Ann Rheum Dis. 2013; 72(Suppl 2):ii116–23.
Article9. Lopez-Olivo MA, Amezaga Urruela M, McGahan L, Pollono EN, Suarez-Almazor ME. Rituximab for rheumatoid arthritis. Cochrane Database Syst Rev. 2015; 1:CD007356.
Article10. Guillevin L, Pagnoux C, Karras A, Khouatra C, Aumaître O, Cohen P, et al. Rituximab versus azathioprine for maintenance in ANCA-associated vasculitis. N Engl J Med. 2014; 371:1771–80.
Article11. Xie S, Li J, Wang JH, Wu Q, Yang P, Hsu HC, et al. IL-17 activates the canonical NF-kappaB signaling pathway in autoimmune B cells of BXD2 mice to upregulate the expression of regulators of G-protein signaling 16. J Immunol. 2010; 184:2289–96.12. Ding Y, Li J, Wu Q, Yang P, Luo B, Xie S, et al. IL-17RA is essential for optimal localization of follicular Th cells in the germinal center light zone to promote autoantibody-pro-ducing B cells. J Immunol. 2013; 191:1614–24.
Article13. Mitsdoerffer M, Lee Y, Jäger A, Kim HJ, Korn T, Kolls JK, et al. Proinflammatory T helper type 17 cells are effective B-cell helpers. Proc Natl Acad Sci U S A. 2010; 107:14292–7.
Article14. Corneth OB, Mus AM, Asmawidjaja PS, Klein Wolterink RG, van Nimwegen M, Brem MD, et al. Absence of interleukin-17 receptor a signaling prevents autoimmune inflammation of the joint and leads to a Th2-like phenotype in collagen-induced arthritis. Arthritis Rheumatol. 2014; 66:340–9.
Article15. Patakas A, Benson RA, Withers DR, Conigliaro P, McInnes IB, Brewer JM, et al. Th17 effector cells support B cell responses outside of germinal centres. PLoS One. 2012; 7:e49715.
Article16. Hsu HC, Yang P, Wang J, Wu Q, Myers R, Chen J, et al. Interleukin 17-producing T helper cells and interleukin 17 orchestrate autoreactive germinal center development in autoimmune BXD2 mice. Nat Immunol. 2008; 9:166–75.17. Snapper CM, Paul WE. Interferon-gamma and B cell stimulatory factor-1 reciprocally regulate Ig isotype production. Science. 1987; 236:944–7.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Regulation of IgG1 and IgE synthesis by interleukin 4 in mouse B cells
- Curcumin Elevates T(FH) Cells and Germinal Center B Cell Response for Antibody Production in Mice
- Anti-inflammatory Effect of Alloferon on Ovalbumin-induced Asthma
- Immunohistochemical study on the formation of germinal centers in the spleen of mouse injected with sheep red blood cells
- Effect of pseudomonas aeruginosa infection on production of IL-2 and IL-6, and other parameters of immunocompetency in mice