Korean J Physiol Pharmacol.
2016 Mar;20(2):201-211. 10.4196/kjpp.2016.20.2.201.
NecroX-5 protects mitochondrial oxidative phosphorylation capacity and preserves PGC1alpha expression levels during hypoxia/reoxygenation injury
- Affiliations
-
- 1National Research Laboratory for Mitochondrial Signaling, Department of Physiology, College of Medicine, Cardiovascular and Metabolic Disease Center, Inje University, Busan 47392, Korea. phyhanj@inje.ac.kr
- 2Faculty of Biology, VNU University of Science, Hanoi 120036, Vietnam.
- 3Product Strategy and Development, LG Life Sciences Ltd, Seoul 03184, Korea.
- KMID: 2388677
- DOI: http://doi.org/10.4196/kjpp.2016.20.2.201
Abstract
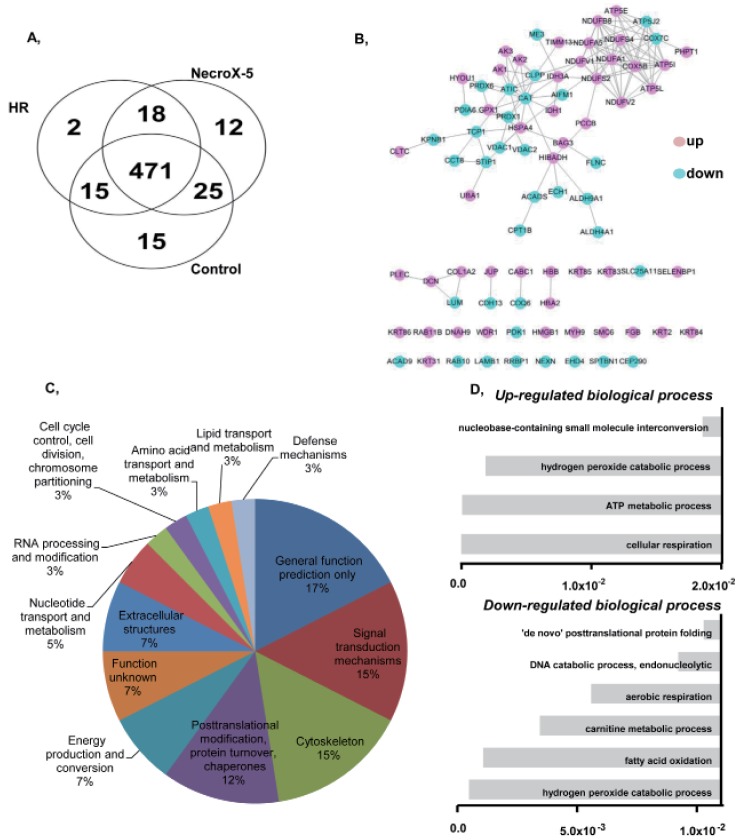
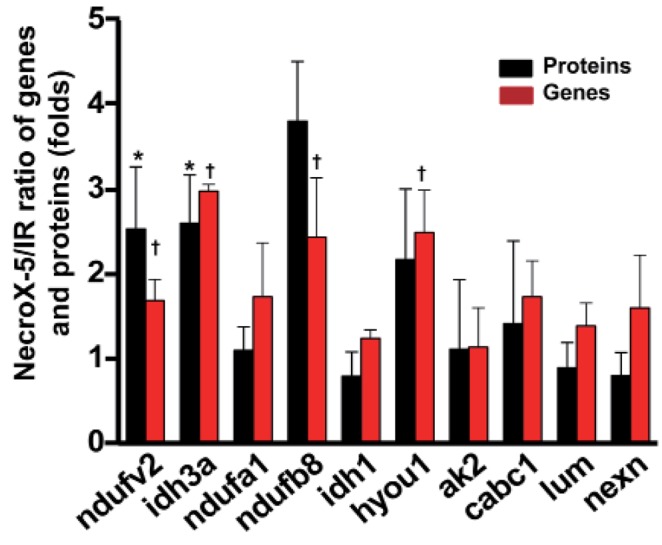
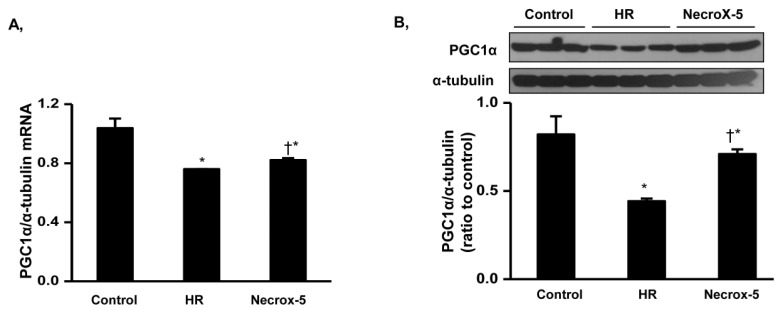
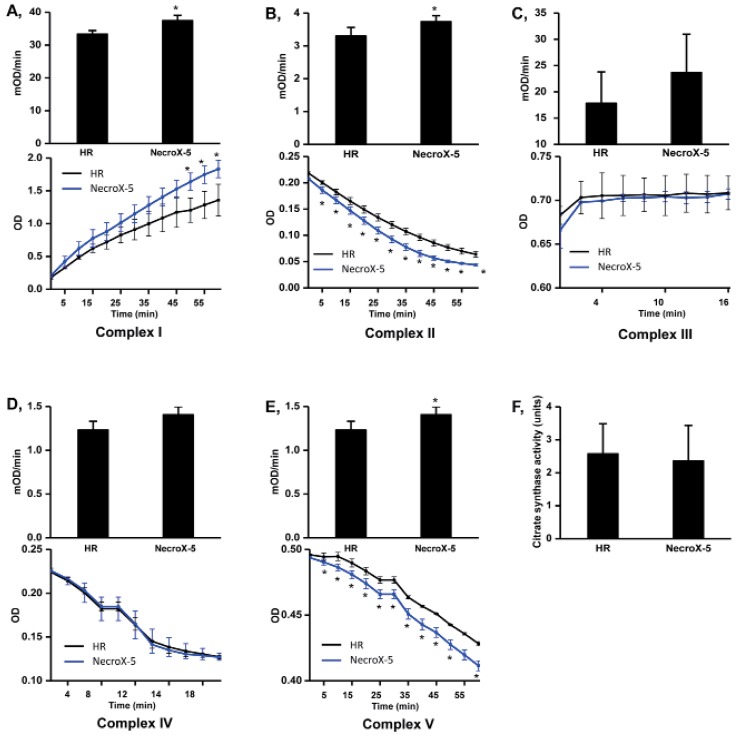
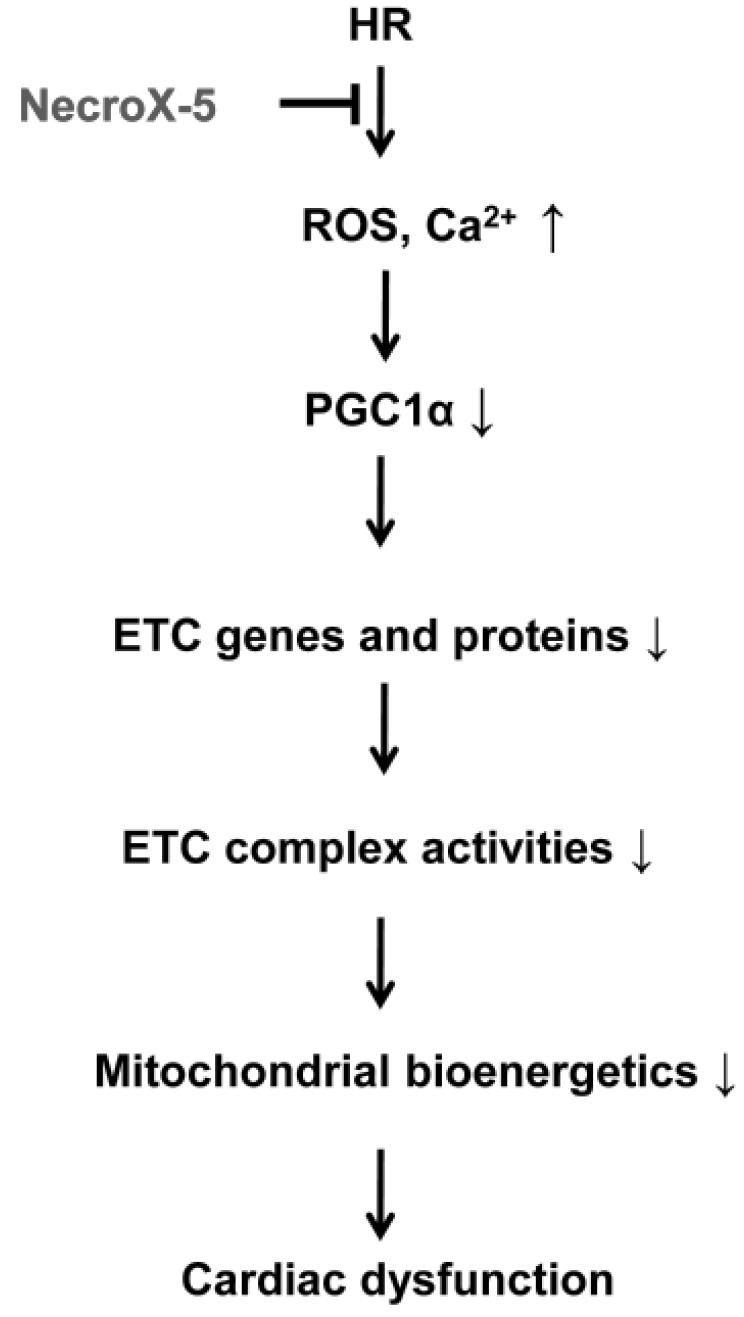
- Although the antioxidant and cardioprotective effects of NecroX-5 on various in vitro and in vivo models have been demonstrated, the action of this compound on the mitochondrial oxidative phosphorylation system remains unclear. Here we verify the role of NecroX-5 in protecting mitochondrial oxidative phosphorylation capacity during hypoxia-reoxygenation (HR). Necrox-5 treatment (10 microM) and non-treatment were employed on isolated rat hearts during hypoxia/reoxygenation treatment using an ex vivo Langendorff system. Proteomic analysis was performed using liquid chromatography-mass spectrometry (LC-MS) and non-labeling peptide count protein quantification. Real-time PCR, western blot, citrate synthases and mitochondrial complex activity assays were then performed to assess heart function. Treatment with NecroX-5 during hypoxia significantly preserved electron transport chain proteins involved in oxidative phosphorylation and metabolic functions. NecroX-5 also improved mitochondrial complex I, II, and V function. Additionally, markedly higher peroxisome proliferator-activated receptor-gamma coactivator-1alpha (PGC1alpha) expression levels were observed in NecroX-5-treated rat hearts. These novel results provide convincing evidence for the role of NecroX-5 in protecting mitochondrial oxidative phosphorylation capacity and in preserving PGC1alpha during cardiac HR injuries.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
NecroX-5 exerts anti-inflammatory and anti-fibrotic effects via modulation of the TNFα/Dcn/TGFβ1/Smad2 pathway in hypoxia/reoxygenation-treated rat hearts
Vu Thi Thu, Hyoung Kyu Kim, Le Thanh Long, To Thanh Thuy, Nguyen Quang Huy, Soon Ha Kim, Nari Kim, Kyung Soo Ko, Byoung Doo Rhee, Jin Han
Korean J Physiol Pharmacol. 2016;20(3):305-314. doi: 10.4196/kjpp.2016.20.3.305.
Reference
-
1. Carden DL, Granger DN. Pathophysiology of ischaemia-reperfusion injury. J Pathol. 2000; 190:255–266. PMID: 10685060.
Article2. Halestrap AP. A pore way to die: the role of mitochondria in reperfusion injury and cardioprotection. Biochem Soc Trans. 2010; 38:841–860. PMID: 20658967.
Article3. Kleinbongard P, Schulz R, Heusch G. TNFα in myocardial ischemia/reperfusion, remodeling and heart failure. Heart Fail Rev. 2011; 16:49–69. PMID: 20571888.
Article4. Ribichini F, Wijns W. Acute myocardial infarction: reperfusion treatment. Heart. 2002; 88:298–305. PMID: 12181231.
Article5. Xia A, Xue Z, Wang W, Zhang T, Wei T, Sha X, Ding Y, Zhou W. Naloxone postconditioning alleviates rat myocardial ischemia reperfusion injury by inhibiting JNK activity. Korean J Physiol Pharmacol. 2014; 18:67–72. PMID: 24634599.
Article6. Brand MD, Nicholls DG. Assessing mitochondrial dysfunction in cells. Biochem J. 2011; 435:297–312. PMID: 21726199.
Article7. Murphy E, Steenbergen C. Mechanisms underlying acute protection from cardiac ischemia-reperfusion injury. Physiol Rev. 2008; 88:581–609. PMID: 18391174.
Article8. Piot C, Croisille P, Staat P, Thibault H, Rioufol G, Mewton N, Elbelghiti R, Cung TT, Bonnefoy E, Angoulvant D, Macia C, Raczka F, Sportouch C, Gahide G, Finet G, André-Fouët X, Revel D, Kirkorian G, Monassier JP, Derumeaux G, Ovize M. Effect of cyclosporine on reperfusion injury in acute myocardial infarction. N Engl J Med. 2008; 359:473–481. PMID: 18669426.
Article9. Heller A, Brockhoff G, Goepferich A. Targeting drugs to mitochondria. Eur J Pharm Biopharm. 2012; 82:1–18. PMID: 22687572.10. McLeod CJ, Pagel I, Sack MN. The mitochondrial biogenesis regulatory program in cardiac adaptation to ischemia--a putative target for therapeutic intervention. Trends Cardiovasc Med. 2005; 15:118–123. PMID: 16039972.
Article11. Kim HK, Thu VT, Heo HJ, Kim N, Han J. Cardiac proteomic responses to ischemia-reperfusion injury and ischemic preconditioning. Expert Rev Proteomics. 2011; 8:241–261. PMID: 21501017.12. Ventura-Clapier R, Garnier A, Veksler V. Transcriptional control of mitochondrial biogenesis: the central role of PGC-1alpha. Cardiovasc Res. 2008; 79:208–217. PMID: 18430751.13. Sun CK, Chang LT, Sheu JJ, Wang CY, Youssef AA, Wu CJ, Chua S, Yip HK. Losartan preserves integrity of cardiac gap junctions and PGC-1 alpha gene expression and prevents cellular apoptosis in remote area of left ventricular myocardium following acute myocardial infarction. Int Heart J. 2007; 48:533–546. PMID: 17827825.14. Gundewar S, Calvert JW, Jha S, Toedt-Pingel I, Ji SY, Nunez D, Ramachandran A, Anaya-Cisneros M, Tian R, Lefer DJ. Activation of AMP-activated protein kinase by metformin improves left ventricular function and survival in heart failure. Circ Res. 2009; 104:403–411. PMID: 19096023.
Article15. Kim HJ, Koo SY, Ahn BH, Park O, Park DH, Seo DO, Won JH, Yim HJ, Kwak HS, Park HS, Chung CW, Oh YL, Kim SH. NecroX as a novel class of mitochondrial reactive oxygen species and ONOO– scavenger. Arch Pharm Res. 2010; 33:1813–1823. PMID: 21116785.16. Thu VT, Kim HK, Long le T, Lee SR, Hanh TM, Ko TH, Heo HJ, Kim N, Kim SH, Ko KS, Rhee BD, Han J. NecroX-5 prevents hypoxia/reoxygenation injury by inhibiting the mitochondrial calcium uniporter. Cardiovasc Res. 2012; 94:342–350. PMID: 22425903.
Article17. Lee SR, Lee SJ, Kim SH, Ko KS, Rhee BD, Xu Z, Kim N, Han J. NecroX-5 suppresses sodium nitroprusside-induced cardiac cell death through inhibition of JNK and caspase-3 activation. Cell Biol Int. 2014; 38:702–707. PMID: 24446382.
Article18. Choi JM, Park KM, Kim SH, Hwang DW, Chon SH, Lee JH, Lee SY, Lee YJ. Effect of necrosis modulator necrox-7 on hepatic ischemia-reperfusion injury in beagle dogs. Transplant Proc. 2010; 42:3414–3421. PMID: 21094788.
Article19. Takaseya T, Ishimatsu M, Tayama E, Nishi A, Akasu T, Aoyagi S. Mechanical unloading improves intracellular Ca2+ regulation in rats with doxorubicin-induced cardiomyopathy. J Am Coll Cardiol. 2004; 44:2239–2246. PMID: 15582323.20. Kim N, Lee Y, Kim H, Joo H, Youm JB, Park WS, Warda M, Cuong DV, Han J. Potential biomarkers for ischemic heart damage identified in mitochondrial proteins by comparative proteomics. Proteomics. 2006; 6:1237–1249. PMID: 16402359.
Article21. Thu VT, Kim HK, Ha SH, Yoo JY, Park WS, Kim N, Oh GT, Han J. Glutathione peroxidase 1 protects mitochondria against hypoxia/reoxygenation damage in mouse hearts. Pflugers Arch. 2010; 460:55–68. PMID: 20306076.
Article22. Kim HK, Song IS, Lee SY, Jeong SH, Lee SR, Heo HJ, Thu VT, Kim N, Ko KS, Rhee BD, Jeong DH, Kim YN, Han J. B7-H4 downregulation induces mitochondrial dysfunction and enhances doxorubicin sensitivity via the cAMP/CREB/PGC1-α signaling pathway in HeLa cells. Pflugers Arch. 2014; 466:2323–2338. PMID: 24658911.
Article23. Kim HK, Kim YK, Song IS, Lee SR, Jeong SH, Kim MH, Seo DY, Kim N, Rhee BD, Ko KS, Tark KC, Park CG, Cho JY, Han J. Human giant congenital melanocytic nevus exhibits potential proteomic alterations leading to melanotumorigenesis. Proteome Sci. 2012; 10:50. PMID: 22906024.
Article24. Bindea G, Mlecnik B, Hackl H, Charoentong P, Tosolini M, Kirilovsky A, Fridman WH, Pagès F, Trajanoski Z, Galon J. Clue-GO: a Cytoscape plug-in to decipher functionally grouped gene ontology and pathway annotation networks. Bioinformatics. 2009; 25:1091–1093. PMID: 19237447.
Article25. Jensen LJ, Kuhn M, Stark M, Chaffron S, Creevey C, Muller J, Doerks T, Julien P, Roth A, Simonovic M, Bork P, von Mering C. STRING 8--a global view on proteins and their functional interactions in 630 organisms. Nucleic Acids Res. 2009; 37:D412–D416. PMID: 18940858.
Article26. Distelmaier F, Koopman WJ, van den Heuvel LP, Rodenburg RJ, Mayatepek E, Willems PH, Smeitink JA. Mitochondrial complex I deficiency: from organelle dysfunction to clinical disease. Brain. 2009; 132:833–842. PMID: 19336460.
Article27. Morán M, Rivera H, Sánchez-Aragó M, Blázquez A, Merinero B, Ugalde C, Arenas J, Cuezva JM, Martín MA. Mitochondrial bioenergetics and dynamics interplay in complex I-deficient fibroblasts. Biochim Biophys Acta. 2010; 1802:443–453. PMID: 20153825.
Article28. Bénit P, Beugnot R, Chretien D, Giurgea I, De Lonlay-Debeney P, Issartel JP, Corral-Debrinski M, Kerscher S, Rustin P, Rötig A, Munnich A. Mutant NDUFV2 subunit of mitochondrial complex I causes early onset hypertrophic cardiomyopathy and encephalopathy. Hum Mutat. 2003; 21:582–586. PMID: 12754703.
Article29. Mbaya E, Oulès B, Caspersen C, Tacine R, Massinet H, Pennuto M, Chrétien D, Munnich A, Rötig A, Rizzuto R, Rutter GA, Paterlini-Bréchot P, Chami M. Calcium signalling-dependent mitochondrial dysfunction and bioenergetics regulation in respiratory chain Complex II deficiency. Cell Death Differ. 2010; 17:1855–1866. PMID: 20489732.
Article30. Gill RS, Lee TF, Manouchehri N, Liu JQ, Lopaschuk G, Bigam DL, Cheung PY. Postresuscitation cyclosporine treatment attenuates myocardial and cardiac mitochondrial injury in newborn piglets with asphyxia-reoxygenation. Crit Care Med. 2013; 41:1069–1074. PMID: 23385100.
Article31. Arany Z, Foo SY, Ma Y, Ruas JL, Bommi-Reddy A, Girnun G, Cooper M, Laznik D, Chinsomboon J, Rangwala SM, Baek KH, Rosenzweig A, Spiegelman BM. HIF-independent regulation of VEGF and angiogenesis by the transcriptional coactivator PGC-1alpha. Nature. 2008; 451:1008–1012. PMID: 18288196.32. Ma X, Liu H, Foyil SR, Godar RJ, Weinheimer CJ, Hill JA, Diwan A. Impaired autophagosome clearance contributes to cardiomyocyte death in ischemia/reperfusion injury. Circulation. 2012; 125:3170–3181. PMID: 22592897.
Article33. Ma X, Liu H, Murphy JT, Foyil SR, Godar RJ, Abuirqeba H, Weinheimer CJ, Barger PM, Diwan A. Regulation of the transcription factor EB-PGC1α axis by beclin-1 controls mitochondrial quality and cardiomyocyte death under stress. Mol Cell Biol. 2015; 35:956–976. PMID: 25561470.
Article34. Settembre C, De Cegli R, Mansueto G, Saha PK, Vetrini F, Visvikis O, Huynh T, Carissimo A, Palmer D, Klisch TJ, Wollenberg AC, Di Bernardo D, Chan L, Irazoqui JE, Ballabio A. TFEB controls cellular lipid metabolism through a starvation-induced autoregulatory loop. Nat Cell Biol. 2013; 15:647–658. PMID: 23604321.
Article35. Rasbach KA, Green PT, Schnellmann RG. Oxidants and Ca2+ induce PGC-1alpha degradation through calpain. Arch Biochem Biophys. 2008; 478:130–135. PMID: 18718443.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- NecroX-5 exerts anti-inflammatory and anti-fibrotic effects via modulation of the TNFα/Dcn/TGFβ1/Smad2 pathway in hypoxia/reoxygenation-treated rat hearts
- Phosphorylation in Novel Mitochondrial Creatine Kinase Tyrosine Residues Render Cardioprotection against Hypoxia/Reoxygenation Injury
- Diastolic Dysfunction Induced by a High-Fat Diet Is Associated with Mitochondrial Abnormality and Adenosine Triphosphate Levels in Rats
- Cardioprotection Via Modulation of Calcium Homeostasis by Thiopental in Hypoxia-Reoxygenated Neonatal Rat Cardiomyocytes
- Reducing Oxidative Stress and Inflammation by Pyruvate Dehydrogenase Kinase 4 Inhibition Is Important in Prevention of Renal Ischemia-Reperfusion Injury in Diabetic Mice