Korean J Physiol Pharmacol.
2016 Mar;20(2):185-192. 10.4196/kjpp.2016.20.2.185.
The neuroprotective mechanism of ampicillin in a mouse model of transient forebrain ischemia
- Affiliations
-
- 1Department of Pharmacology, Catholic Neuroscience Institute, College of Medicine, The Catholic University of Korea, Seoul 137-701, Korea. syk@catholic.ac.kr
- 2Department of Pharmaceutical Science and Technology, College of Health and Medical Science, Catholic University of Daegu, Daegu 712-702, Korea.
- KMID: 2388675
- DOI: http://doi.org/10.4196/kjpp.2016.20.2.185
Abstract
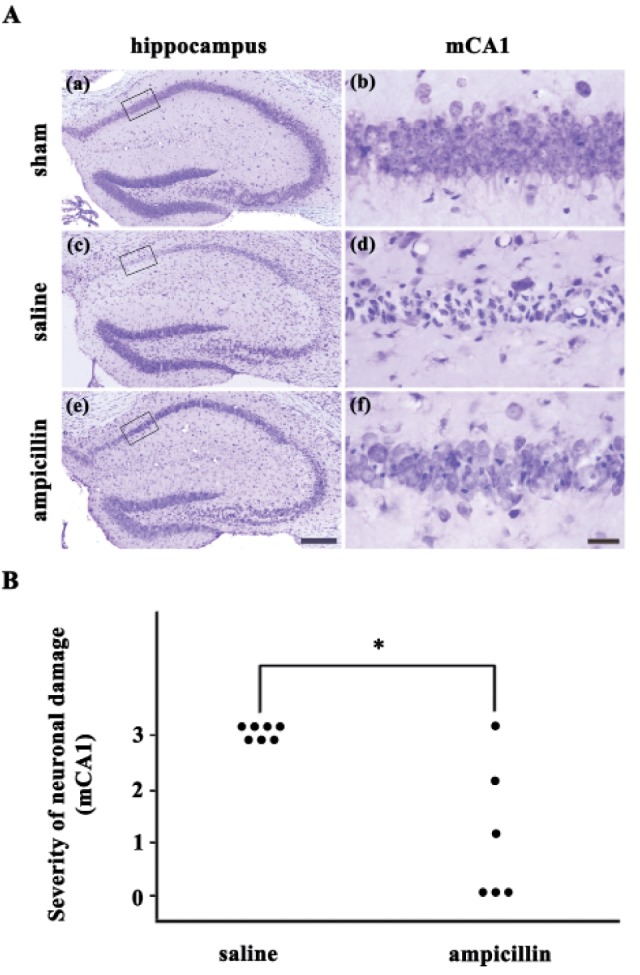
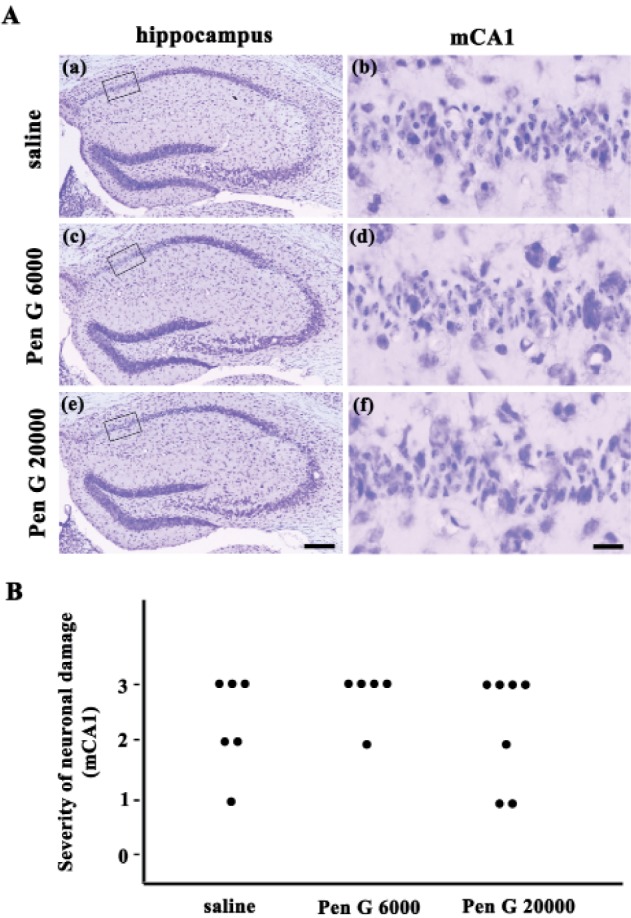
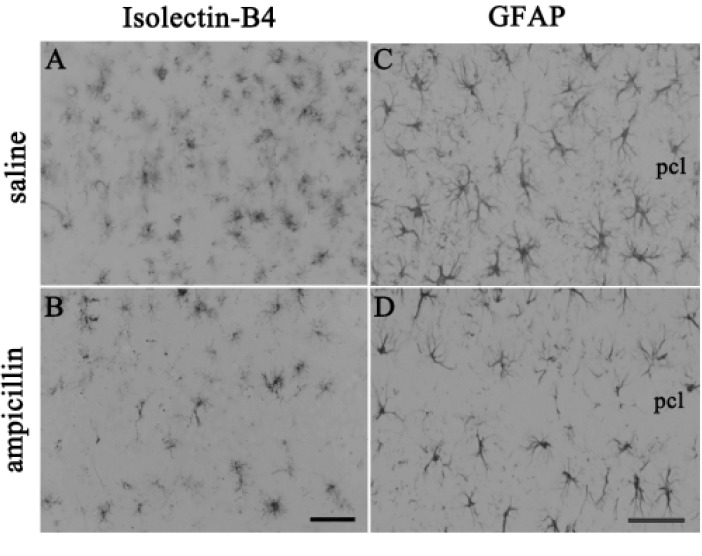
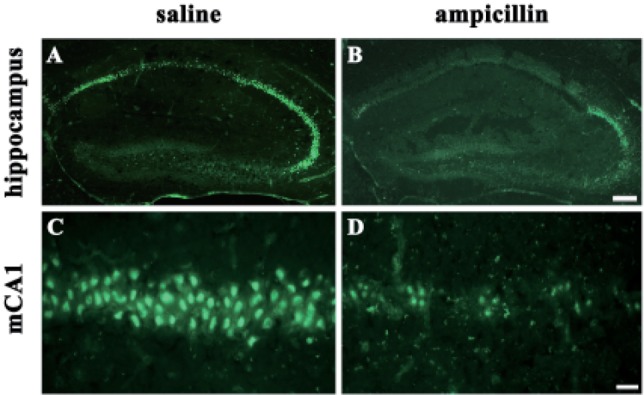
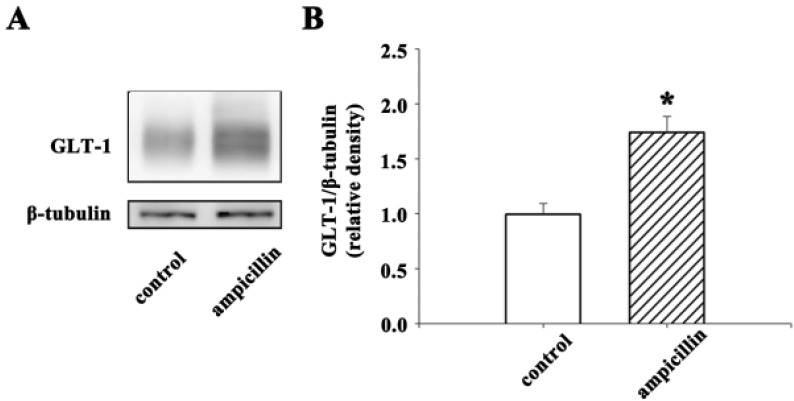
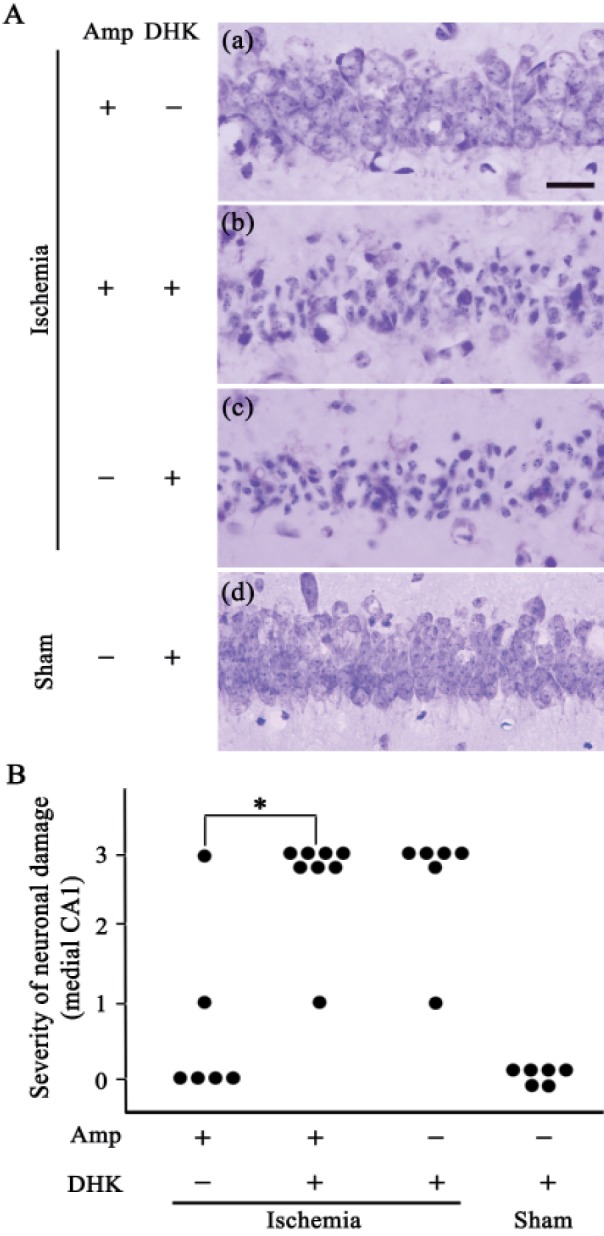
- Ampicillin, a beta-lactam antibiotic, dose-dependently protects neurons against ischemic brain injury. The present study was performed to investigate the neuroprotective mechanism of ampicillin in a mouse model of transient global forebrain ischemia. Male C57BL/6 mice were anesthetized with halothane and subjected to bilateral common carotid artery occlusion for 40 min. Before transient forebrain ischemia, ampicillin (200 mg/kg, intraperitoneally [i.p.]) or penicillin G (6,000 U/kg or 20,000 U/kg, i.p.) was administered daily for 5 days. The pretreatment with ampicillin but not with penicillin G signifi cantly attenuated neuronal damage in the hippocampal CA1 subfield. Mechanistically, the increased activity of matrix metalloproteinases (MMPs) following forebrain ischemia was also attenuated by ampicillin treatment. In addition, the ampicillin treatment reversed increased immunoreactivities to glial fibrillary acidic protein and isolectin B4, markers of astrocytes and microglia, respectively. Furthermore, the ampicillin treatment significantly increased the level of glutamate transporter-1, and dihydrokainic acid (DHK, 10 mg/kg, i.p.), an inhibitor of glutamate transporter-1 (GLT-1), reversed the neuroprotective effect of ampicillin. Taken together, these data indicate that ampicillin provides neuroprotection against ischemia-reperfusion brain injury, possibly through inducing the GLT-1 protein and inhibiting the activity of MMP in the mouse hippocampus.
Keyword
MeSH Terms
-
Ampicillin*
Animals
Astrocytes
Brain Injuries
Carotid Artery, Common
Glial Fibrillary Acidic Protein
Glutamic Acid
Halothane
Hippocampus
Humans
Ischemia*
Lectins
Male
Matrix Metalloproteinases
Mice*
Microglia
Neurons
Neuroprotective Agents
Penicillin G
Prosencephalon*
Ampicillin
Glial Fibrillary Acidic Protein
Glutamic Acid
Halothane
Lectins
Matrix Metalloproteinases
Neuroprotective Agents
Penicillin G
Figure
Reference
-
1. Kirino T. Delayed neuronal death in the gerbil hippocampus following ischemia. Brain Res. 1982; 239:57–69. PMID: 7093691.
Article2. Lipton P. Ischemic cell death in brain neurons. Physiol Rev. 1999; 79:1431–1568. PMID: 10508238.
Article3. Chong W, Kim SN, Han SK, Lee SY, Ryu PD. Low non-NMDA receptor current density as possible protection mechanism from neurotoxicity of circulating glutamate on subfornical organ neurons in rats. Korean J Physiol Pharmacol. 2015; 19:177–181. PMID: 25729280.
Article4. Melani A, Dettori I, Corti F, Cellai L, Pedata F. Time-course of protection by the selective A2A receptor antagonist SCH58261 after transient focal cerebral ischemia. Neurol Sci. 2015; 36:1441–1448. PMID: 25805704.
Article5. Yu G, Wu F, Wang ES. BQ-869, a novel NMDA receptor antagonist, protects against excitotoxicity and attenuates cerebral ischemic injury in stroke. Int J Clin Exp Pathol. 2015; 8:1213–1225. PMID: 25973006.6. Kong Q, Takahashi K, Schulte D, Stouffer N, Lin Y, Lin CL. Increased glial glutamate transporter EAAT2 expression reduces epileptogenic processes following pilocarpine-induced status epilepticus. Neurobiol Dis. 2012; 47:145–154. PMID: 22513140.
Article7. Zumkehr J, Rodriguez-Ortiz CJ, Cheng D, Kieu Z, Wai T, Hawkins C, Kilian J, Lim SL, Medeiros R, Kitazawa M. Ceftriaxone ameliorates tau pathology and cognitive decline via restoration of glial glutamate transporter in a mouse model of Alzheimer's disease. Neurobiol Aging. 2015; 36:2260–2271. PMID: 25964214.
Article8. Zhang M, Li WB, Liu YX, Liang CJ, Liu LZ, Cui X, Gong JX, Gong SJ, Hu YY, Xian XH. High expression of GLT-1 in hippocampal CA3 and dentate gyrus subfields contributes to their inherent resistance to ischemia in rats. Neurochem Int. 2011; 59:1019–1028. PMID: 21925558.
Article9. Fontana AC. Current approaches to enhance glutamate transporter function and expression. J Neurochem. 2015; 134:982–1007. PMID: 26096891.
Article10. Massie A, Cnops L, Smolders I, McCullumsmith R, Kooijman R, Kwak S, Arckens L, Michotte Y. High-affinity Na+/K+-dependent glutamate transporter EAAT4 is expressed throughout the rat fore- and midbrain. J Comp Neurol. 2008; 511:155–172. PMID: 18770868.11. Scofield MD, Kalivas PW. Astrocytic dysfunction and addiction: consequences of impaired glutamate homeostasis. Neuroscientist. 2014; 20:610–622. PMID: 24496610.12. Mookherjee P, Green PS, Watson GS, Marques MA, Tanaka K, Meeker KD, Meabon JS, Li N, Zhu P, Olson VG, Cook DG. GLT-1 loss accelerates cognitive deficit onset in an Alzheimer's disease animal model. J Alzheimers Dis. 2011; 26:447–455. PMID: 21677376.
Article13. Soni N, Reddy BV, Kumar P. GLT-1 transporter: an effective pharmacological target for various neurological disorders. Pharmacol Biochem Behav. 2014; 127:70–81. PMID: 25312503.
Article14. Zhou J, Sutherland ML. Glutamate transporter cluster formation in astrocytic processes regulates glutamate uptake activity. J Neurosci. 2004; 24:6301–6306. PMID: 15254085.
Article15. Meeker KD, Meabon JS, Cook DG. Partial loss of the glutamate transporter GLT-1 alters brain Akt and insulin signaling in a mouse model of Alzheimer's disease. J Alzheimers Dis. 2015; 45:509–520. PMID: 25589729.
Article16. Petr GT, Sun Y, Frederick NM, Zhou Y, Dhamne SC, Hameed MQ, Miranda C, Bedoya EA, Fischer KD, Armsen W, Wang J, Danbolt NC, Rotenberg A, Aoki CJ, Rosenberg PA. Conditional deletion of the glutamate transporter GLT-1 reveals that astrocytic GLT-1 protects against fatal epilepsy while neuronal GLT-1 contributes significantly to glutamate uptake into synaptosomes. J Neurosci. 2015; 35:5187–5201. PMID: 25834045.
Article17. Hu YY, Xu J, Zhang M, Wang D, Li L, Li WB. Ceftriaxone modulates uptake activity of glial glutamate transporter-1 against global brain ischemia in rats. J Neurochem. 2015; 132:194–205. PMID: 25270764.
Article18. Chu K, Lee ST, Sinn DI, Ko SY, Kim EH, Kim JM, Kim SJ, Park DK, Jung KH, Song EC, Lee SK, Kim M, Roh JK. Pharmacological induction of ischemic tolerance by glutamate transporter-1 (EAAT2) upregulation. Stroke. 2007; 38:177–182. PMID: 17122424.
Article19. Lee KE, Kim SK, Cho KO, Kim SY. Pre-ischemic treatment with ampicillin reduces neuronal damage in the mouse hippocampus and neostriatum after transient forebrain ischemia. Korean J Physiol Pharmacol. 2008; 12:287–291. PMID: 19967069.
Article20. Rothstein JD, Patel S, Regan MR, Haenggeli C, Huang YH, Bergles DE, Jin L, Dykes Hoberg M, Vidensky S, Chung DS, Toan SV, Bruijn LI, Su ZZ, Gupta P, Fisher PB. Beta-lactam antibiotics offer neuroprotection by increasing glutamate transporter expression. Nature. 2005; 433:73–77. PMID: 15635412.21. Cho KO, Kim SK, Cho YJ, Sung KW, Kim SY. A simple method for predicting hippocampal neurodegeneration in a mouse model of transient global forebrain ischemia. Korean J Physiol Pharmacol. 2006; 10:167–172.22. Zhang M, Li WB, Geng JX, Li QJ, Sun XC, Xian XH, Qi J, Li SQ. The upregulation of glial glutamate transporter-1 participates in the induction of brain ischemic tolerance in rats. J Cereb Blood Flow Metab. 2007; 27:1352–1368. PMID: 17228332.
Article23. Murakami K, Kondo T, Kawase M, Chan PH. The development of a new mouse model of global ischemia: focus on the relationships between ischemia duration, anesthesia, cerebral vasculature, and neuronal injury following global ischemia in mice. Brain Res. 1998; 780:304–310. PMID: 9507171.
Article24. Lee SR, Tsuji K, Lee SR, Lo EH. Role of matrix metalloproteinases in delayed neuronal damage after transient global cerebral ischemia. J Neurosci. 2004; 24:671–678. PMID: 14736853.
Article25. Pandey AK, Bhattacharya P, Shukla SC, Paul S, Patnaik R. Resveratrol inhibits matrix metalloproteinases to attenuate neuronal damage in cerebral ischemia: a molecular docking study exploring possible neuroprotection. Neural Regen Res. 2015; 10:568–575. PMID: 26170816.
Article26. Asahi M, Asahi K, Jung JC, del Zoppo GJ, Fini ME, Lo EH. Role for matrix metalloproteinase 9 after focal cerebral ischemia: effects of gene knockout and enzyme inhibition with BB-94. J Cereb Blood Flow Metab. 2000; 20:1681–1689. PMID: 11129784.
Article27. Spector R. Nature and consequences of mammalian brain and CSF efflux transporters: four decades of progress. J Neurochem. 2010; 112:13–23. PMID: 19860860.
Article28. Hu YY, Xu J, Zhang M, Wang D, Li L, Li WB. Ceftriaxone modulates uptake activity of glial glutamate transporter-1 against global brain ischemia in rats. J Neurochem. 2015; 132:194–205. PMID: 25270764.
Article29. Kelsey JE, Neville C. The effects of the β-lactam antibiotic, ceftriaxone, on forepaw stepping and L-DOPA-induced dyskinesia in a rodent model of Parkinson's disease. . Psychopharmacology (Berl). 2014; 231:2405–2415. PMID: 24402134.
Article30. Gao D, Huang T, Jiang X, Hu S, Zhang L, Fei Z. Resveratrol protects primary cortical neuron cultures from transient oxygen-glucose deprivation by inhibiting MMP-9. Mol Med Rep. 2014; 9:2197–2204. PMID: 24682241.
Article31. Kim SJ, Lee SR. Protective effect of melatonin against transient global cerebral ischemia-induced neuronal cell damage via inhibition of matrix metalloproteinase-9. Life Sci. 2014; 94:8–16. PMID: 24269215.
Article32. Harkness KA, Adamson P, Sussman JD, Davies-Jones GA, Greenwood J, Woodroofe MN. Dexamethasone regulation of matrix metalloproteinase expression in CNS vascular endothelium. Brain. 2000; 123:698–709. PMID: 10734001.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Pre-ischemic Treatment with Ampicillin Reduces Neuronal Damage in the Mouse Hippocampus and Neostriatum after Transient Forebrain Ischemia
- Corrigendum to: Pre-ischemic treatment with ampicillin reduces neuronal damage in the mouse hippocampus and neostriatum after transient forebrain ischemia
- Neuroprotective Effects of Propofol, Ketamine and Propofol-ketamine after Transient Forebrain Ischemia in the Rat
- The neuroprotective effect of colloids in forebrain ischemia model
- Neuroprotective Effect of Focal Ischemic Preconditioning in Transient Focal Ischemia Animal Model