Nutr Res Pract.
2017 Aug;11(4):290-299. 10.4162/nrp.2017.11.4.290.
Monocyte chemoattractant protein-1 polymorphism interaction with spirulina immunomodulatory effects in healthy Korean elderly: A 16 week, double-blind randomized clinical trial
- Affiliations
-
- 1Department of Foods and Nutrition, Kookmin University, Seoul 02707, Korea.
- 2Department of Food Science and Nutrition, Dongseo University, 47 Jurye-ro, Sasang-gu, Busan 47011, Korea. hyunlee@gdsu.dongseo.ac.kr
- KMID: 2385467
- DOI: http://doi.org/10.4162/nrp.2017.11.4.290
Abstract
- BACKGROUND/OBJECTIVES
Spirulina is a known a functional food related to lipid profiles, immune functions, and antioxidant capacity. Circulating monocyte chemoattractant protein-1 (MCP-1) level is associated with inflammation markers. Single nucleotide polymorphism in the MCP-1 promoter region -2518 have been identified and shown to affect gene transcription. Gene variation may also impact functional food supplementary effects. The current study investigated the interaction of MCP-1 -2518 polymorphism with spirulina supplements on anti-inflammatory capacity in Korean elderly.
SUBJECTS/METHODS
After genotyping, healthy elderly subjects (n = 78) were included in a randomized, double blind, and placebo controlled study. Baseline characteristic, body composition, and dietary intake were measured twice (baseline vs. week 16). For 16 weeks, subjects consumed 8 g either spirulina or placebo daily. Plasma MCP-1, interleukin (IL) -2, IL-6, tumor necrosis factor (TNF)-α, complement (C) 3, immunoglobulin (Ig) G, and Ig A concentrations and lymphocyte proliferation rate (LPR) were analyzed as inflammatory markers.
RESULTS
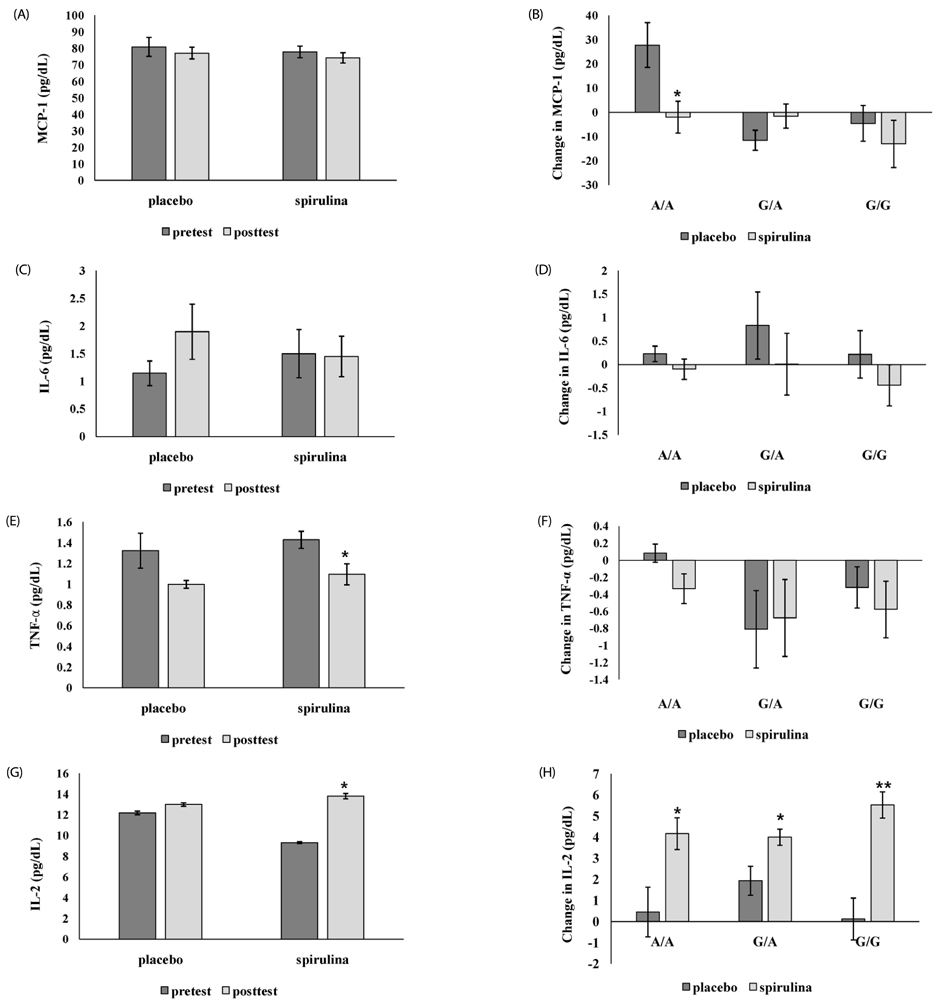
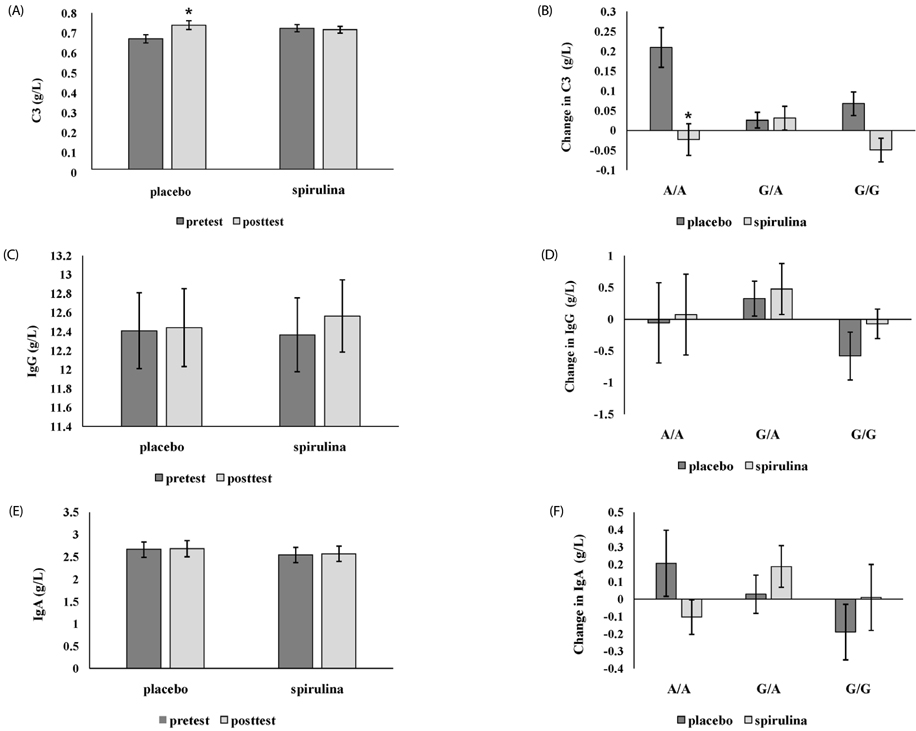
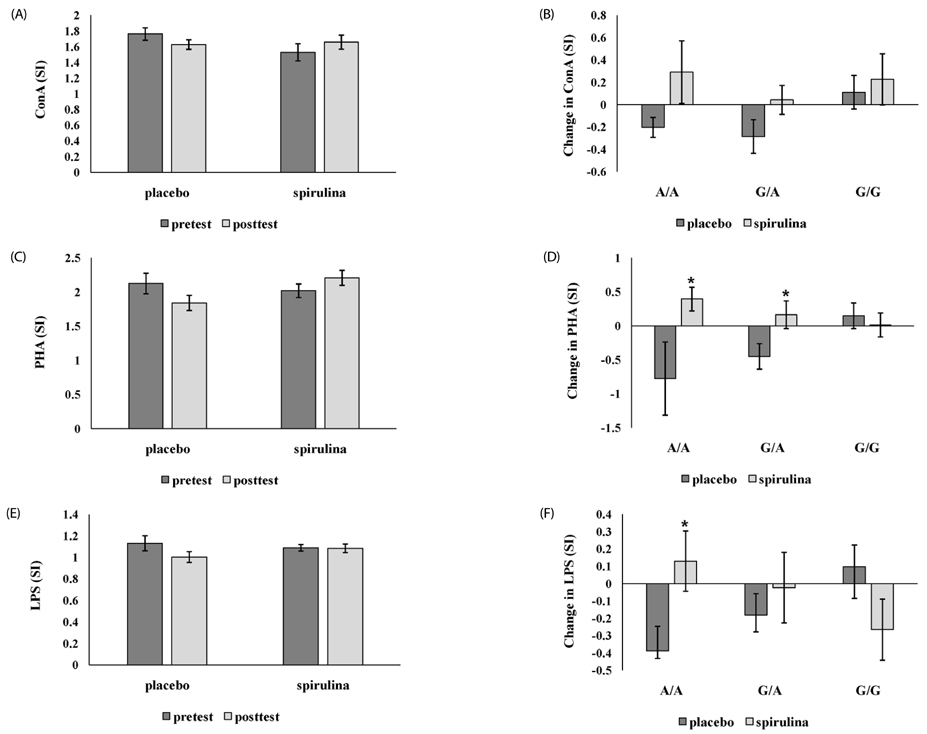
In the placebo group with A/A genotype, MCP-1 level was significantly increased, but the spirulina group with A/A genotype was unchanged. IL-2 was significantly increased only in subjects with spirulina supplementation. TNF-α was significantly reduced in subjects with the G carrier. C3 was significantly increased in the placebo group, particularly when A/A increased more than G, but not when spirulina was ingested. LPR was significantly different only in subjects with A/A genotype; there was a significant increase in phytohemagglutinin and lipopolysaccharide induced LPR in the spirulina group.
CONCLUSION
In healthy Korean elderly, spirulina supplementation may influence different inflammatory markers by the MCP-1 genotype. These results may be useful for customized dietary guidelines to improve immune function in Koreans.
Keyword
MeSH Terms
-
Aged*
Body Composition
Chemokine CCL2*
Complement System Proteins
Functional Food
Genotype
Humans
Immune System
Immunoglobulins
Inflammation
Interleukin-2
Interleukin-6
Interleukins
Lymphocytes
Monocytes*
Nutrition Policy
Plasma
Polymorphism, Single Nucleotide
Promoter Regions, Genetic
Spirulina*
Tumor Necrosis Factor-alpha
Chemokine CCL2
Complement System Proteins
Immunoglobulins
Interleukin-2
Interleukin-6
Interleukins
Tumor Necrosis Factor-alpha
Figure
Reference
-
1. Muller WA. Getting leukocytes to the site of inflammation. Vet Pathol. 2013; 50:7–22.
Article2. Xu L, Rocnik E, Rahlpour R, Hunter N, Pickering G, Kelvin DJ. MCP-1 induces proliferation and migration of vascular smooth muscle cells. FASEB J. 1996; 10:A1932.3. Luster AD. Chemokines--chemotactic cytokines that mediate inflammation. N Engl J Med. 1998; 338:436–445.
Article4. Rossi D, Zlotnik A. The biology of chemokines and their receptors. Annu Rev Immunol. 2000; 18:217–242.
Article5. Salcedo R, Ponce ML, Young HA, Wasserman K, Ward JM, Kleinman HK, Oppenheim JJ, Murphy WJ. Human endothelial cells express CCR2 and respond to MCP-1: direct role of MCP-1 in angiogenesis and tumor progression. Blood. 2000; 96:34–40.
Article6. Inadera H, Egashira K, Takemoto M, Ouchi Y, Matsushima K. Increase in circulating levels of monocyte chemoattractant protein-1 with aging. J Interferon Cytokine Res. 1999; 19:1179–1182.
Article7. Rovin BH, Lu L, Saxena R. A novel polymorphism in the MCP-1 gene regulatory region that influences MCP-1 expression. Biochem Biophys Res Commun. 1999; 259:344–348.
Article8. Martin ES, Schneeberger EE, Aranda FM, Peres SW, Del Carmen Valerio M, de Los Angeles Correa M, Pra FD, Martinez L, Remondino G, de Larrañaga G, Citera G. The -2518 A/G polymorphism in the monocyte chemoattractant protein 1 gene (MCP-1) is associated with an increased risk of rheumatoid arthritis in Argentine patients. Clin Rheumatol. 2016; 35:3057–3061.
Article9. Wang W, He M, Huang W. Association of monocyte chemoattractant protein-1 gene 2518A/G polymorphism with diabetic retinopathy in type 2 diabetes mellitus: a meta-analysis. Diabetes Res Clin Pract. 2016; 120:40–46.
Article10. Niu J, Kolattukudy PE. Role of MCP-1 in cardiovascular disease: molecular mechanisms and clinical implications. Clin Sci (Lond). 2009; 117:95–109.
Article11. Mansego ML, De Marco G, Ivorra C, Lopez-Izquierdo R, Morcillo S, Rojo-Martínez G, González-Albert V, Martinez F, Soriguer F, Martín-Escudero JC, Redon J, Chaves FJ. The nutrigenetic influence of the interaction between dietary vitamin E and TXN and COMT gene polymorphisms on waist circumference: a case control study. J Transl Med. 2015; 13:286.
Article12. Pase CS, Teixeira AM, Roversi K, Dias VT, Calabrese F, Molteni R, Franchi S, Panerai AE, Riva MA, Burger ME. Olive oil-enriched diet reduces brain oxidative damages and ameliorates neurotrophic factor gene expression in different life stages of rats. J Nutr Biochem. 2015; 26:1200–1207.
Article13. Pietropaolo S, Goubran MG, Joffre C, Aubert A, Lemaire-Mayo V, Crusio WE, Layé S. Dietary supplementation of omega-3 fatty acids rescues fragile X phenotypes in Fmr1-Ko mice. Psychoneuroendocrinology. 2014; 49:119–129.
Article14. Nigam A, Talajic M, Roy D, Nattel S, Lambert J, Nozza A, Jones P, Ramprasath VR, O'Hara G, Kopecky S, Brophy JM, Tardif JC. AFFORD Investigators. Fish oil for the reduction of atrial fibrillation recurrence, inflammation, and oxidative stress. J Am Coll Cardiol. 2014; 64:1441–1448.
Article15. Yanaka A, Fahey JW, Fukumoto A, Nakayama M, Inoue S, Zhang S, Tauchi M, Suzuki H, Hyodo I, Yamamoto M. Dietary sulforaphane-rich broccoli sprouts reduce colonization and attenuate gastritis in Helicobacter pylori-infected mice and humans. Cancer Prev Res (Phila). 2009; 2:353–360.
Article16. Vicari E, La Vignera S, Castiglione R, Condorelli RA, Vicari LO, Calogero AE. Chronic bacterial prostatitis and irritable bowel syndrome: effectiveness of treatment with rifaximin followed by the probiotic VSL#3. Asian J Androl. 2014; 16:735–739.
Article17. Ortega-Azorín C, Sorlí JV, Asensio EM, Coltell O, Martínez-González MA, Salas-Salvadó J, Covas MI, Arós F, Lapetra J, Serra-Majem L, Gómez-Gracia E, Fiol M, Sáez-Tormo G, Pintó X, Muñoz MA, Ros E, Ordovás JM, Estruch R, Corella D. Associations of the FTO rs9939609 and the MC4R rs17782313 polymorphisms with type 2 diabetes are modulated by diet, being higher when adherence to the Mediterranean diet pattern is low. Cardiovasc Diabetol. 2012; 11:137.
Article18. Serrano JC, De Lorenzo D, Cassanye A, Martín-Gari M, Espinel A, Delgado MA, Pamplona R, Portero-Otin M. Vitamin D receptor BsmI polymorphism modulates soy intake and 25-hydroxyvitamin D supplementation benefits in cardiovascular disease risk factors profile. Genes Nutr. 2013; 8:561–569.
Article19. Giacconi R, Costarelli L, Malavolta M, Cardelli M, Galeazzi R, Piacenza F, Gasparini N, Basso A, Mariani E, Fulop T, Rink L, Dedoussis G, Herbein G, Jajte J, Provinciali M, Busco F, Mocchegiani E. Effect of ZIP2 Gln/Arg/Leu (rs2234632) polymorphism on zinc homeostasis and inflammatory response following zinc supplementation. Biofactors. 2015; 41:414–423.
Article20. Park S, Zhang X, Lee NR, Jin HS. TRPV1 gene polymorphisms are associated with type 2 diabetes by their interaction with fat consumption in the Korean genome epidemiology study. J Nutrigenet Nutrigenomics. 2016; 9:47–61.
Article21. Thengodkar RR, Sivakami S. Degradation of Chlorpyrifos by an alkaline phosphatase from the cyanobacterium Spirulina platensis. Biodegradation. 2010; 21:637–644.
Article22. Lee JB, Srisomporn P, Hayashi K, Tanaka T, Sankawa U, Hayashi T. Effects of structural modification of calcium spirulan, a sulfated polysaccharide from Spirulina platensis, on antiviral activity. Chem Pharm Bull (Tokyo). 2001; 49:108–110.
Article23. Marcel AK, Ekali LG, Eugene S, Arnold OE, Sandrine ED, von der Weid D, Gbaguidi E, Ngogang J, Mbanya JC. The effect of Spirulina platensis versus soybean on insulin resistance in HIV-infected patients: a randomized pilot study. Nutrients. 2011; 3:712–724.
Article24. Nkengfack G, Torimiro J, Ngogang J, Heike E. Effects of nutritional education and lifestyle modification on the nutritional status of HIV positive patients: results of a cluster randomized intervention over a period of six months. Glob Epidemi Obes. 2013; 1:5.
Article25. Moura LP, Puga GM, Beck WR, Teixeira IP, Ghezzi AC, Silva GA, Mello MA. Exercise and Spirulina control non-alcoholic hepatic steatosis and lipid profile in diabetic Wistar rats. Lipids Health Dis. 2011; 10:77.
Article26. Layam A, Reddy CL. Antidiabetic property of Spirulina. Diabetol Croat. 2007; 35:29–33.27. Hayashi O, Katoh T, Okuwaki Y. Enhancement of antibody production in mice by dietary Spirulina platensis. J Nutr Sci Vitaminol (Tokyo). 1994; 40:431–441.
Article28. Watanuki H, Ota K, Tassakka AC, Kato T, Sakai M. Immunostimulant effects of dietary Spirulina platensis on carp, Cyprinus carpio. Aquaculture. 2006; 258:157–163.
Article29. Qureshi MA, Kidd MT, Ali RA. Spirulina platensis extract enhances chicken macrophages functions after in vitro exposure. J Nutr Immunol. 1996; 3:35–45.30. Hirahashi T, Matsumoto M, Hazeki K, Saeki Y, Ui M, Seya T. Activation of the human innate immune system by Spirulina: augmentation of interferon production and NK cytotoxicity by oral administration of hot water extract of Spirulina platensis. Int Immunopharmacol. 2002; 2:423–434.
Article31. Selmi C, Leung PS, Fischer L, German B, Yang CY, Kenny TP, Cysewski GR, Gershwin ME. The effects of Spirulina on anemia and immune function in senior citizens. Cell Mol Immunol. 2011; 8:248–254.
Article32. Park HJ, Lee YJ, Ryu HK, Kim MH, Chung HW, Kim WY. A randomized double-blind, placebo-controlled study to establish the effects of Spirulina in elderly Koreans. Ann Nutr Metab. 2008; 52:322–328.
Article33. Park HJ, Lee HS. The influence of obesity on the effects of Spirulina supplementation in the human metabolic response of Korean elderly. Nutr Res Pract. 2016; 10:418–423.
Article34. The Korean Nutrition Society. CAN-Pro 4.0: nutritional assessment program. Seoul: The Korean Nutrition Society;2010.35. Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972; 18:499–502.
Article36. Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983; 65:55–63.
Article37. Bielinski SJ, Pankow JS, Miller MB, Hopkins PN, Eckfeldt JH, Hixson J, Liu Y, Register T, Myers RH, Arnett DK. Circulating MCP-1 levels shows linkage to chemokine receptor gene cluster on chromosome 3: the NHLBI family heart study follow-up examination. Genes Immun. 2007; 8:684–690.
Article38. Deshmane SL, Kremlev S, Amini S, Sawaya BE. Monocyte chemoattractant protein-1 (MCP-1): an overview. J Interferon Cytokine Res. 2009; 29:313–326.
Article39. Yamamoto T, Eckes B, Mauch C, Hartmann K, Krieg T. Monocyte chemoattractant protein-1 enhances gene expression and synthesis of matrix metalloproteinase-1 in human fibroblasts by an autocrine IL-1 alpha loop. J Immunol. 2000; 164:6174–6179.
Article40. Xia M, Sui Z. Recent developments in CCR2 antagonists. Expert Opin Ther Pat. 2009; 19:295–303.
Article41. Rollins BJ. Chemokines. Blood. 1997; 90:909–928.
Article42. de Lemos JA, Morrow DA, Sabatine MS, Murphy SA, Gibson CM, Antman EM, McCabe CH, Cannon CP, Braunwald E. Association between plasma levels of monocyte chemoattractant protein-1 and long-term clinical outcomes in patients with acute coronary syndromes. Circulation. 2003; 107:690–695.
Article43. Deo R, Khera A, McGuire DK, Murphy SA, Meo Neto, Morrow DA, de Lemos JA. Association among plasma levels of monocyte chemoattractant protein-1, traditional cardiovascular risk factors, and subclinical atherosclerosis. J Am Coll Cardiol. 2004; 44:1812–1818.
Article44. Navratilova Z. Polymorphisms in CCL2&CCL5 chemokines/chemokine receptors genes and their association with diseases. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2006; 150:191–204.
Article45. Szalai C, Kozma GT, Nagy A, Bojszkó A, Krikovszky D, Szabó T, Falus A. Polymorphism in the gene regulatory region of MCP-1 is associated with asthma susceptibility and severity. J Allergy Clin Immunol. 2001; 108:375–381.
Article46. Arakelyan A, Zakharyan R, Hambardzumyan M, Petrkova J, Olsson MC, Petrek M, Boyajyan A. Functional genetic polymorphisms of monocyte chemoattractant protein 1 and C-C chemokine receptor type 2 in ischemic stroke. J Interferon Cytokine Res. 2014; 34:100–105.
Article47. Bjarnadottir K, Eiriksdottir G, Aspelund T, Gudnason V. Examination of genetic effects of polymorphisms in the MCP-1 and CCR2 genes on MI in the Icelandic population. Atherosclerosis. 2006; 188:341–346.
Article48. Angeles-Martínez J, Posadas-Sánchez R, Álvarez-León E, Villarreal-Molina T, Cardoso-Saldaña G, Fragoso JM, Juárez-Rojas JG, Medina-Urrutia A, Posadas-Romero C, Vargas-Alarcón G. Monocyte chemoattractant protein-1 gene (MCP-1) polymorphisms are associated with risk of premature coronary artery disease in Mexican patients from the Genetics of Atherosclerotic Disease (GEA) study. Immunol Lett. 2015; 167:125–130.
Article49. Xu J, Liao YF, Zhou WP, Ming HL, Wang QH. The MCP-1 gene A-2518G polymorphism confers an increased risk of vascular complications in type 2 diabetes mellitus patients. Genet Test Mol Biomarkers. 2015; 19:411–417.
Article50. Hou S, Yang P, Du L, Jiang Z, Mao L, Shu Q, Zhou H, Kijlstra A. Monocyte chemoattractant protein-1 -2518 A/G single nucleotide polymorphism in Chinese Han patients with ocular Behçet's disease. Hum Immunol. 2010; 71:79–82.
Article51. Zuo S, Wang H, Wang B. Association of monocyte chemoattractant protein-1-2518A/G polymorphism and risk of coronary artery disease among the Chinese population: a meta-analysis. Int J Clin Exp Med. 2015; 8:15182–15189.52. Jemaa R, Rojbani H, Kallel A, Ben Ali S, Feki M, Chabrak S, Elasmi M, Taieb SH, Sanhaji H, Souheil O, Mechmeche R, Kaabachi N. Association between the -2518G/A polymorphism in the monocyte chemoattractant protein-1 (MCP-1) gene and myocardial infarction in Tunisian patients. Clin Chim Acta. 2008; 390:122–125.
Article53. Kaur R, Matharoo K, Arora P, Bhanwer AJ. Association of -2518A>G promoter polymorphism in the monocyte chemoattractant protein-1 (MCP-1) gene with type 2 diabetes and coronary artery disease. Genet Test Mol Biomarkers. 2013; 17:750–755.
Article54. Okopień B, Haberka M, Cwalina L, Kowalski J, Belowski D, Madej A, Zieliński M, Krysiak R, Labuzek K, Kalina Z, Herman ZS. Plasma cytokines as predictors of coronary heart disease. Res Commun Mol Pathol Pharmacol. 2002; 112:5–15.55. Gao HH, Gao LB, Wen JM. Correlations of MCP-1 -2518A>G polymorphism and serum levels with cerebral infarction risk: a meta-analysis. DNA Cell Biol. 2014; 33:522–530.
Article56. Park HJ. Association of MCP-1 polymorphism with cardiovascular disease risk factors in Korean elderly. J Nutr Health. 2013; 46:511–520.
Article57. Nelken NA, Coughlin SR, Gordon D, Wilcox JN. Monocyte chemoattractant protein-1 in human atheromatous plaques. J Clin Invest. 1991; 88:1121–1127.
Article58. Nikolich-Žugich J. T cell aging: naive but not young. J Exp Med. 2005; 201:837–840.59. Halkes CJ, van Dijk H, de Jaegere PP, Plokker HW, van Der Helm Y, Erkelens DW, Castro Cabezas M. Postprandial increase of complement component 3 in normolipidemic patients with coronary artery disease: effects of expanded-dose simvastatin. Arterioscler Thromb Vasc Biol. 2001; 21:1526–1530.
Article60. Muscari A, Bozzoli C, Puddu GM, Sangiorgi Z, Dormi A, Rovinetti C, Descovich GC, Puddu P. Association of serum C3 levels with the risk of myocardial infarction. Am J Med. 1995; 98:357–364.
Article61. Hoskinson CD, Chew BP, Wong TS. Age-related changes in mitogen-induced lymphocyte proliferation and polymorphonuclear neutrophil function in the piglet. J Anim Sci. 1990; 68:2471–2478.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Potential Role of Monocyte Chemoattractant Protein-1 for Major Depressive Disorder
- The influence of obesity on the effects of spirulina supplementation in the human metabolic response of Korean elderly
- Analysis of the Monocyte Chemoattractant Protein 1 -2518 Promoter Polymorphism in Korean Patients with Alopecia Areata
- Gene - Gene Interactions Among MCP Genes Polymorphisms in Asthma
- Monocyte Chemoattractant Protein-1(MCP1)-2518 Polymorphism and Major Depressive Disorder