Korean J Physiol Pharmacol.
2017 Jul;21(4):377-384. 10.4196/kjpp.2017.21.4.377.
Protein kinase C beta II upregulates intercellular adhesion molecule-1 via mitochondrial activation in cultured endothelial cells
- Affiliations
-
- 1Research Institute for Medical Sciences, Department of Physiology, School of Medicine, Chungnam National University, Daejeon 35015, Korea. bhjeon@cnu.ac.kr
- KMID: 2384451
- DOI: http://doi.org/10.4196/kjpp.2017.21.4.377
Abstract
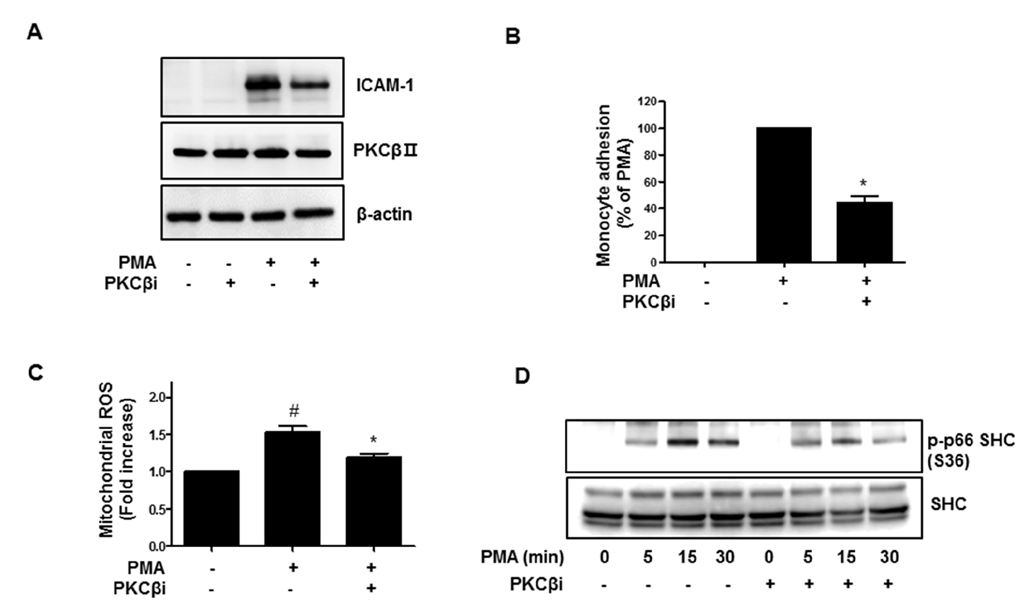
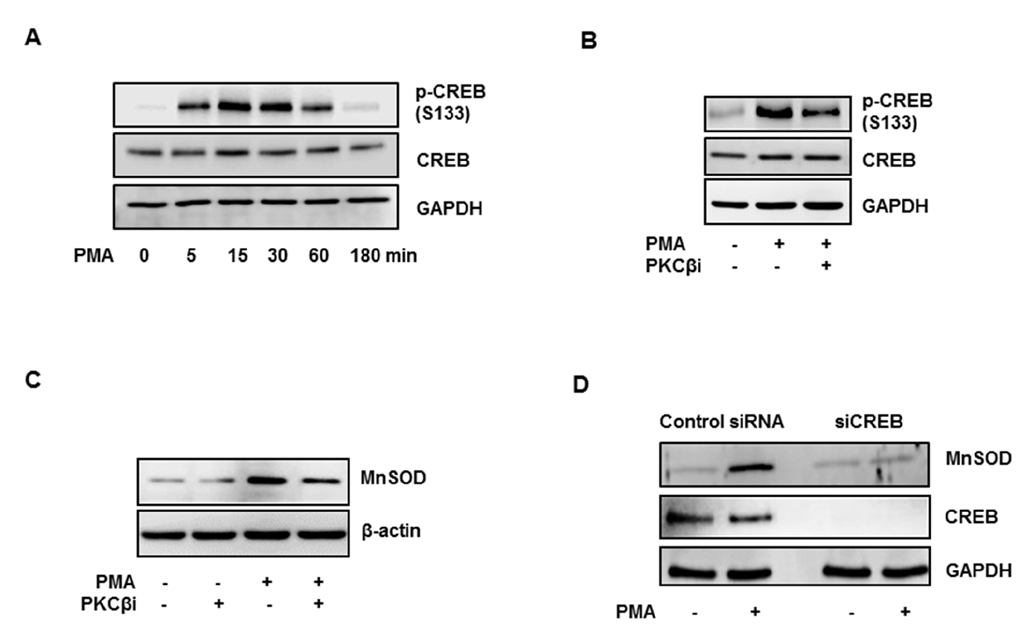
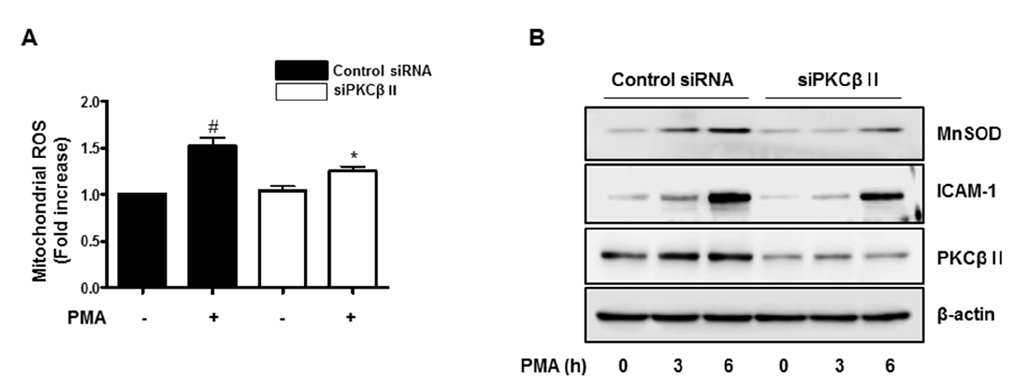
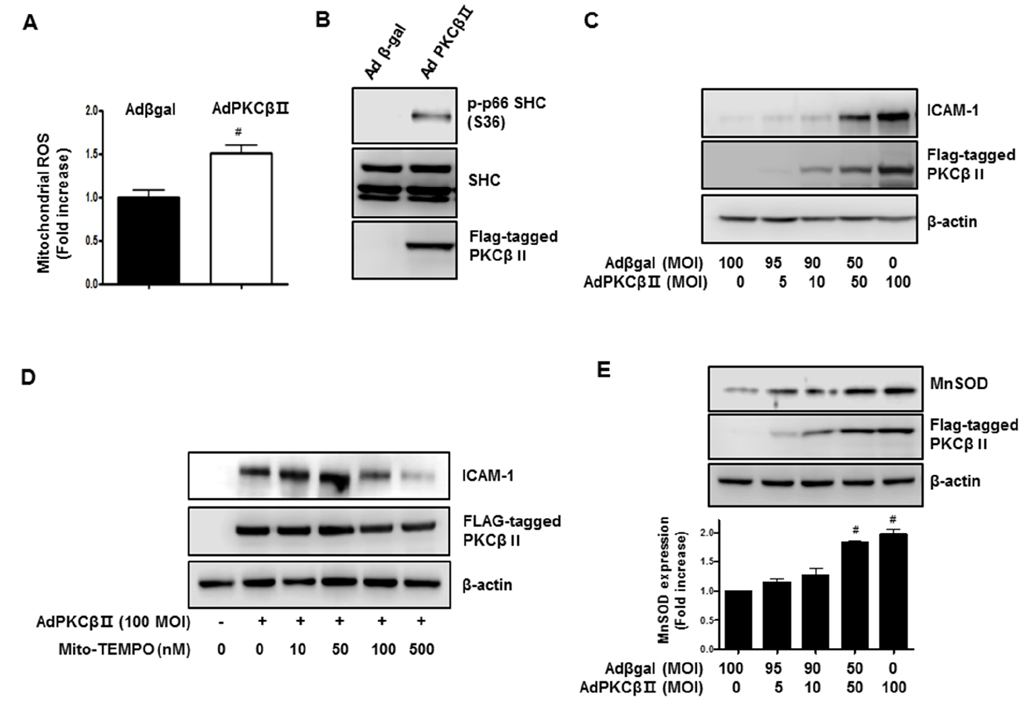
- Activation of protein kinase C (PKC) is closely linked with endothelial dysfunction. However, the effect of PKCβII on endothelial dysfunction has not been characterized in cultured endothelial cells. Here, using adenoviral PKCβII gene transfer and pharmacological inhibitors, the role of PKCβII on endothelial dysfucntion was investigated in cultured endothelial cells. Phorbol 12-myristate 13-acetate (PMA) increased reactive oxygen species (ROS), p66shc phosphorylation, intracellular adhesion molecule-1, and monocyte adhesion, which were inhibited by PKCβi (10 nM), a selective inhibitor of PKCβII. PMA increased the phosphorylation of CREB and manganese superoxide dismutase (MnSOD), which were also inhibited by PKCβi. Gene silencing of CREB inhibited PMA-induced MnSOD expression, suggesting that CREB plays a key role in MnSOD expression. Gene silencing of PKCβII inhibited PMA-induced mitochondrial ROS, MnSOD, and ICAM-1 expression. In contrast, overexpression of PKCβII using adenoviral PKCβII increased mitochondrial ROS, MnSOD, ICAM-1, and p66shc phosphorylation in cultured endothelial cells. Finally, PKCβII-induced ICAM-1 expression was inhibited by Mito-TEMPO, a mitochondrial ROS scavenger, suggesting the involvement of mitochondrial ROS in PKC-induced vascular inflammation. Taken together, the results suggest that PKCβII plays an important role in PMA-induced endothelial dysfunction, and that the inhibition of PKCβII-dependent p66shc signaling acts as a therapeutic target for vascular inflammatory diseases.
MeSH Terms
-
Endothelial Cells*
Gene Silencing
Inflammation
Intercellular Adhesion Molecule-1
Mitochondria
Monocytes
Phosphorylation
Protein Kinase C beta*
Protein Kinase C*
Protein Kinases*
Reactive Oxygen Species
Superoxide Dismutase
Intercellular Adhesion Molecule-1
Protein Kinase C
Protein Kinases
Reactive Oxygen Species
Superoxide Dismutase
Figure
Cited by 2 articles
-
Arginase Inhibition Suppresses Native Low-Density Lipoprotein-Stimulated Vascular Smooth Muscle Cell Proliferation by NADPH Oxidase Inactivation
Bon-Hyeock Koo, Bong-Gu Yi, Wi-Kwang Wang, In-Young Ko, Kwang-Lae Hoe, Young-Guen Kwon, Moo-Ho Won, Young-Myeong Kim, Hyun Kyo Lim, Sungwoo Ryoo
Yonsei Med J. 2018;59(3):366-375. doi: 10.3349/ymj.2018.59.3.366.Biphasic augmentation of alpha-adrenergic contraction by plumbagin in rat systemic arteries
Hae Jin Kim, Hae Young Yoo, Yin Hua Zhang, Woo Kyung Kim, Sung Joon Kim
Korean J Physiol Pharmacol. 2017;21(6):687-694. doi: 10.4196/kjpp.2017.21.6.687.
Reference
-
1. Giovannini C, Matarrese P, Scazzocchio B, Sanchez M, Masella R, Malorni W. Mitochondria hyperpolarization is an early event in oxidized low-density lipoprotein-induced apoptosis in Caco-2 intestinal cells. FEBS Lett. 2002; 523:200–206.2. Munusamy S, MacMillan-Crow LA. Mitochondrial superoxide plays a crucial role in the development of mitochondrial dysfunction during high glucose exposure in rat renal proximal tubular cells. Free Radic Biol Med. 2009; 46:1149–1157.3. Sommer SP, Sommer S, Sinha B, Wiedemann J, Otto C, Aleksic I, Schimmer C, Leyh RG. Ischemia-reperfusion injury-induced pulmonary mitochondrial damage. J Heart Lung Transplant. 2011; 30:811–818.4. Chen KH, Reece LM, Leary JF. Mitochondrial glutathione modulates TNF-alpha-induced endothelial cell dysfunction. Free Radic Biol Med. 1999; 27:100–109.5. Zinovkin RA, Romaschenko VP, Galkin II, Zakharova VV, Pletjushkina OY, Chernyak BV, Popova EN. Role of mitochondrial reactive oxygen species in age-related inflammatory activation of endothelium. Aging (Albany NY). 2014; 6:661–674.6. Kim HJ, Park KG, Yoo EK, Kim YH, Kim YN, Kim HS, Kim HT, Park JY, Lee KU, Jang WG, Kim JG, Kim BW, Lee IK. Effects of PGC-1alpha on TNF-alpha-induced MCP-1 and VCAM-1 expression and NF-kappaB activation in human aortic smooth muscle and endothelial cells. Antioxid Redox Signal. 2007; 9:301–307.7. Liao JK. Linking endothelial dysfunction with endothelial cell activation. J Clin Invest. 2013; 123:540–541.8. Ross R. Cell biology of atherosclerosis. Annu Rev Physiol. 1995; 57:791–804.9. Davidson SM, Duchen MR. Endothelial mitochondria: contributing to vascular function and disease. Circ Res. 2007; 100:1128–1141.10. Kluge MA, Fetterman JL, Vita JA. Mitochondria and endothelial function. Circ Res. 2013; 112:1171–1188.11. Joo HK, Lee YR, Park MS, Choi S, Park K, Lee SK, Kim CS, Park JB, Jeon BH. Mitochondrial APE1/Ref-1 suppressed protein kinase C-induced mitochondrial dysfunction in mouse endothelial cells. Mitochondrion. 2014; 17:42–49.12. Patterson CE, Davis HW, Schaphorst KL, Garcia JG. Mechanisms of cholera toxin prevention of thrombin- and PMA-induced endothelial cell barrier dysfunction. Microvasc Res. 1994; 48:212–235.13. Goel A, Thor D, Anderson L, Rahimian R. Sexual dimorphism in rabbit aortic endothelial function under acute hyperglycemic conditions and gender-specific responses to acute 17beta-estradiol. Am J Physiol Heart Circ Physiol. 2008; 294:H2411–H2420.14. Pinton P, Rimessi A, Marchi S, Orsini F, Migliaccio E, Giorgio M, Contursi C, Minucci S, Mantovani F, Wieckowski MR, Del Sal G, Pelicci PG, Rizzuto R. Protein kinase C beta and prolyl isomerase 1 regulate mitochondrial effects of the life-span determinant p66Shc. Science. 2007; 315:659–663.15. Giorgio M, Migliaccio E, Orsini F, Paolucci D, Moroni M, Contursi C, Pelliccia G, Luzi L, Minucci S, Marcaccio M, Pinton P, Rizzuto R, Bernardi P, Paolucci F, Pelicci PG. Electron transfer between cytochrome c and p66Shc generates reactive oxygen species that trigger mitochondrial apoptosis. Cell. 2005; 122:221–233.16. Kim HP, Roe JH, Chock PB, Yim MB. Transcriptional activation of the human manganese superoxide dismutase gene mediated by tetradecanoylphorbol acetate. J Biol Chem. 1999; 274:37455–37460.17. Bedogni B, Pani G, Colavitti R, Riccio A, Borrello S, Murphy M, Smith R, Eboli ML, Galeotti T. Redox regulation of cAMP-responsive element-binding protein and induction of manganous superoxide dismutase in nerve growth factor-dependent cell survival. J Biol Chem. 2003; 278:16510–16519.18. Naruse K, Rask-Madsen C, Takahara N, Ha SW, Suzuma K, Way KJ, Jacobs JR, Clermont AC, Ueki K, Ohshiro Y, Zhang J, Goldfine AB, King GL. Activation of vascular protein kinase C-beta inhibits Akt-dependent endothelial nitric oxide synthase function in obesity-associated insulin resistance. Diabetes. 2006; 55:691–698.19. Lei S, Li H, Xu J, Liu Y, Gao X, Wang J, Ng KF, Lau WB, Ma XL, Rodrigues B, Irwin MG, Xia Z. Hyperglycemia-induced protein kinase C β2 activation induces diastolic cardiac dysfunction in diabetic rats by impairing caveolin-3 expression and Akt/eNOS signaling. Diabetes. 2013; 62:2318–2328.20. Harja E, Chang JS, Lu Y, Leitges M, Zou YS, Schmidt AM, Yan SF. Mice deficient in PKCbeta and apolipoprotein E display decreased atherosclerosis. FASEB J. 2009; 23:1081–1091.21. Lee SK, Chung JI, Park MS, Joo HK, Lee EJ, Cho EJ, Park JB, Ryoo S, Irani K, Jeon BH. Apurinic/apyrimidinic endonuclease 1 inhibits protein kinase C-mediated p66shc phosphorylation and vasoconstriction. Cardiovasc Res. 2011; 91:502–509.22. Jeon BH, Gupta G, Park YC, Qi B, Haile A, Khanday FA, Liu YX, Kim JM, Ozaki M, White AR, Berkowitz DE, Irani K. Apurinic/apyrimidinic endonuclease 1 regulates endothelial NO production and vascular tone. Circ Res. 2004; 95:902–910.23. Sun L, Xiao L, Nie J, Liu FY, Ling GH, Zhu XJ, Tang WB, Chen WC, Xia YC, Zhan M, Ma MM, Peng YM, Liu H, Liu YH, Kanwar YS. p66Shc mediates high-glucose and angiotensin II-induced oxidative stress renal tubular injury via mitochondrial-dependent apoptotic pathway. Am J Physiol Renal Physiol. 2010; 299:F1014–F1025.24. Ono Y, Kurokawa T, Fujii T, Kawahara K, Igarashi K, Kikkawa U, Ogita K, Nishizuka Y. Two types of complementary DNAs of rat brain protein kinase C. Heterogeneity determined by alternative splicing. FEBS Lett. 1986; 206:347–352.25. Steinberg SF. Structural basis of protein kinase C isoform function. Physiol Rev. 2008; 88:1341–1378.26. Tanaka M, Sagawa S, Hoshi J, Shimoma F, Matsuda I, Sakoda K, Sasase T, Shindo M, Inaba T. Synthesis of anilino-monoindolylmaleimides as potent and selective PKCbeta inhibitors. Bioorg Med Chem Lett. 2004; 14:5171–5174.27. Lee SK, Lee JY, Joo HK, Cho EJ, Kim CS, Lee SD, Park JB, Jeon BH. Tat-mediated p66shc transduction decreased phosphorylation of endothelial nitric oxide synthase in endothelial cells. Korean J Physiol Pharmacol. 2012; 16:199–204.28. Chiasson VL, Quinn MA, Young KJ, Mitchell BM. Protein kinase CbetaII-mediated phosphorylation of endothelial nitric oxide synthase threonine 495 mediates the endothelial dysfunction induced by FK506 (tacrolimus). J Pharmacol Exp Ther. 2011; 337:718–723.29. Lee SK, Kim HS, Song YJ, Joo HK, Lee JY, Lee KH, Cho EJ, Cho CH, Park JB, Jeon BH. Alteration of p66shc is associated with endothelial dysfunction in the abdominal aortic coarctation of rats. FEBS Lett. 2008; 582:2561–2566.30. Kang G, Lee YR, Joo HK, Park MS, Kim CS, Choi S, Jeon BH. Trichostatin a modulates angiotensin II-induced vasoconstriction and blood pressure via inhibition of p66shc activation. Korean J Physiol Pharmacol. 2015; 19:467–472.31. Li Q, Park K, Li C, Rask-Madsen C, Mima A, Qi W, Mizutani K, Huang P, King GL. Induction of vascular insulin resistance and endothelin-1 expression and acceleration of atherosclerosis by the overexpression of protein kinase C-β isoform in the endothelium. Circ Res. 2013; 113:418–427.32. Kee BL, Arias J, Montminy MR. Adaptor-mediated recruitment of RNA polymerase II to a signal-dependent activator. J Biol Chem. 1996; 271:2373–2375.33. Chung YW, Kim HK, Kim IY, Yim MB, Chock PB. Dual function of protein kinase C (PKC) in 12-O-tetradecanoylphorbol-13-acetate (TPA)-induced manganese superoxide dismutase (MnSOD) expression: activation of CREB and FOXO3a by PKC-alpha phosphorylation and by PKC-mediated inactivation of Akt, respectively. J Biol Chem. 2011; 286:29681–29690.34. Trinei M, Giorgio M, Cicalese A, Barozzi S, Ventura A, Migliaccio E, Milia E, Padura IM, Raker VA, Maccarana M, Petronilli V, Minucci S, Bernardi P, Lanfrancone L, Pelicci PG. A p53-p66Shc signalling pathway controls intracellular redox status, levels of oxidation-damaged DNA and oxidative stress-induced apoptosis. Oncogene. 2002; 21:3872–3878.35. Chen Z, Wang G, Zhai X, Hu Y, Gao D, Ma L, Yao J, Tian X. Selective inhibition of protein kinase C β2 attenuates the adaptor P66 Shc-mediated intestinal ischemia-reperfusion injury. Cell Death Dis. 2014; 5:e1164.36. Dikalova AE, Bikineyeva AT, Budzyn K, Nazarewicz RR, McCann L, Lewis W, Harrison DG, Dikalov SI. Therapeutic targeting of mitochondrial superoxide in hypertension. Circ Res. 2010; 107:106–116.37. Yu JH, Kim CS, Yoo DG, Song YJ, Joo HK, Kang G, Jo JY, Park JB, Jeon BH. NADPH oxidase and mitochondrial ROS are involved in the TNF-alpha-induced vascular cell adhesion molecule-1 and monocyte adhesion in cultured endothelial cells. Korean J Physiol Pharmacol. 2006; 10:217–222.38. Madamanchi NR, Runge MS. Mitochondrial dysfunction in atherosclerosis. Circ Res. 2007; 100:460–473.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Expression of Intercellular Adhesion Molecule-1 and E-Selectin in Gastric Cancer and Their Clinical Significance
- The role of mapk and pkc-delta in phosphatidic acid-mediated intercellular adhesion molecule-1 expression
- Activated platelets induce secretion of interleukin-1beta, monocyte chemotactic protein-1, and macrophage inflammatory protein-1alpha and surface expression of intercellular adhesion molecule-1 on cultured endothelial cells
- Comparative Study on the Expression of ICAM-1 of Preimmune State Keratinocytes Induced by Trichophyton Antigen
- Allicin Reduces Adhesion Molecules and NO Production Induced by gamma irradiation in Human Endothelial Cells