Korean J Physiol Pharmacol.
2016 Sep;20(5):539-545. 10.4196/kjpp.2016.20.5.539.
Nafamostat mesilate promotes endothelium-dependent vasorelaxation via the Akt-eNOS dependent pathway
- Affiliations
-
- 1Department of physiology & BK21Plus CNU Integrative Biomedical Education Initiative, School of Medicine, Chungnam National University, Daejeon 35015, Korea. cskim@cnu.ac.kr
- 2Department of Neurosurgery, Regional Cerebrovascular Center, Chungnam National University Hospital, Daejeon 35015, Korea.
- 3Department of Neurology, Chungnam National University Hospital, Daejeon 35015, Korea.
- 4Division of Cardiology, Internal Medicine, School of Medicine, Chungnam National University, Chungnam National University Hospital, Daejeon 35015, Korea.
- 5Department of Endocrinology, Chungnam National University Hospital, Daejeon 35015, Korea.
- 6Department of Anatomy & BK21Plus CNU Integrative Biomedical Education Initiative, School of Medicine, Chungnam National University, Daejeon 35015, Korea. visnu528@cnu.ac.kr
- KMID: 2350511
- DOI: http://doi.org/10.4196/kjpp.2016.20.5.539
Abstract
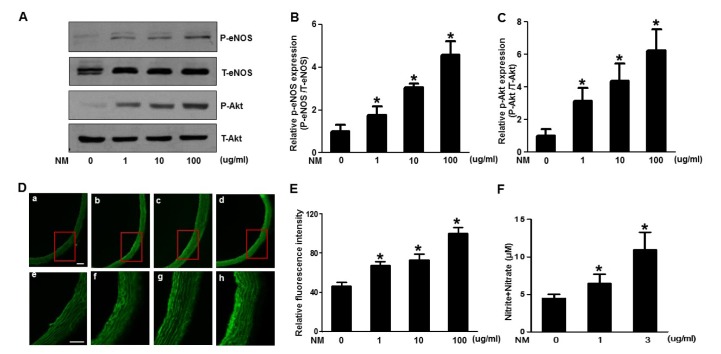
- Nafamostat mesilate (NM), a synthetic serine protease inhibitor, has anticoagulant and anti-inflammatory properties. The intracellular mediator and external anti-inflammatory external signal in the vascular wall have been reported to protect endothelial cells, in part due to nitric oxide (NO) production. This study was designed to examine whether NM exhibit endothelium dependent vascular relaxation through Akt/endothelial nitric oxide synthase (eNOS) activation and generation of NO. NM enhanced Akt/eNOS phosphorylation and NO production in a dose- and time-dependent manner in human umbilical vein endothelial cells (HUVECs) and aorta tissues obtained from rats treated with various concentrations of NM. NM concomitantly decreased arginase activity, which could increase the available arginine substrate for NO production. Moreover, we investigated whether NM increased NO bioavailability and decreased aortic relaxation response to an eNOS inhibitor in the aorta. These results suggest that NM increases NO generation via the Akt/eNOS signaling pathway, leading to endothelium-dependent vascular relaxation. Therefore, the vasorelaxing action of NM may contribute to the regulation of cardiovascular function.
MeSH Terms
-
Animals
Aorta
Arginase
Arginine
Biological Availability
Endothelial Cells
Endothelium
Human Umbilical Vein Endothelial Cells
Mesylates*
Nitric Oxide
Nitric Oxide Synthase
Nitric Oxide Synthase Type III
Phosphorylation
Rats
Relaxation
Serine Proteases
Vasodilation*
Arginase
Arginine
Mesylates
Nitric Oxide
Nitric Oxide Synthase
Nitric Oxide Synthase Type III
Serine Proteases
Figure
Reference
-
1. Ross R, Glomset JA. The pathogenesis of atherosclerosis (first of two parts). N Engl J Med. 1976; 295:369–377. PMID: 819830.2. Rees DD, Palmer RM, Moncada S. Role of endothelium-derived nitric oxide in the regulation of blood pressure. Proc Natl Acad Sci U S A. 1989; 86:3375–3378. PMID: 2497467.
Article3. Vallance P, Collier J, Moncada S. Effects of endothelium-derived nitric oxide on peripheral arteriolar tone in man. Lancet. 1989; 2:997–1000. PMID: 2572793.
Article4. Shesely EG, Maeda N, Kim HS, Desai KM, Krege JH, Laubach VE, Sherman PA, Sessa WC, Smithies O. Elevated blood pressures in mice lacking endothelial nitric oxide synthase. Proc Natl Acad Sci U S A. 1996; 93:13176–13181. PMID: 8917564.
Article5. Watson T, Goon PK, Lip GY. Endothelial progenitor cells, endothelial dysfunction, inflammation, and oxidative stress in hypertension. Antioxid Redox Signal. 2008; 10:1079–1088. PMID: 18315493.
Article6. Huang PL, Huang Z, Mashimo H, Bloch KD, Moskowitz MA, Bevan JA, Fishman MC. Hypertension in mice lacking the gene for endothelial nitric oxide synthase. Nature. 1995; 377:239–242. PMID: 7545787.
Article7. Dimmeler S, Fleming I, Fisslthaler B, Hermann C, Busse R, Zeiher AM. Activation of nitric oxide synthase in endothelial cells by Akt-dependent phosphorylation. Nature. 1999; 399:601–605. PMID: 10376603.
Article8. Palmer RM, Ashton DS, Moncada S. Vascular endothelial cells synthesize nitric oxide from L-arginine. Nature. 1988; 333:664–666. PMID: 3131684.
Article9. Palmer RM, Ferrige AG, Moncada S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature. 1987; 327:524–526. PMID: 3495737.
Article10. Bobadilla NA, Gamba G, Tapia E, García-Torres R, Bolio A, López-Zetina P, Herrera-Acosta J. Role of NO in cyclosporin nephrotoxicity: effects of chronic NO inhibition and NO synthases gene expression. Am J Physiol. 1998; 274:F791–F798. PMID: 9575905.11. Panza JA, Casino PR, Kilcoyne CM, Quyyumi AA. Role of endothelium-derived nitric oxide in the abnormal endothelium-dependent vascular relaxation of patients with essential hypertension. Circulation. 1993; 87:1468–1474. PMID: 8491001.
Article12. Akizawa T, Koshikawa S, Ota K, Kazama M, Mimura N, Hirasawa Y. Nafamostat mesilate: a regional anticoagulant for hemodialysis in patients at high risk for bleeding. Nephron. 1993; 64:376–381. PMID: 8341382.
Article13. Iwaki M, Ino Y, Motoyoshi A, Ozeki M, Sato T, Kurumi M, Aoyama T. Pharmacological studies of FUT-175, nafamostat mesilate. V. effects on the pancreatic enzymes and experimental acute pancreatitis in rats. Jpn J Pharmacol. 1986; 41:155–162. PMID: 2427760.
Article14. Kang MW, Song HJ, Kang SK, Kim Y, Jung SB, Jee S, Moon JY, Suh KS, Lee SD, Jeon BH, Kim CS. Nafamostat mesilate inhibits TNF-α-induced vascular endothelial cell dysfunction by inhibiting reactive oxygen species production. Korean J Physiol Pharmacol. 2015; 19:229–234. PMID: 25954127.
Article15. Kwon SK, Ahn M, Song HJ, Kang SK, Jung SB, Harsha N, Jee S, Moon JY, Suh KS, Lee SD, Jeon BH, Kim DW, Kim CS. Nafamostat mesilate attenuates transient focal ischemia/reperfusion-induced brain injury via the inhibition of endoplasmic reticulum stress. Brain Res. 2015; 1627:12–20. PMID: 26390938.
Article16. Gonzalez W, Fontaine V, Pueyo ME, Laquay N, Messika-Zeitoun D, Philippe M, Arnal JF, Jacob MP, Michel JB. Molecular plasticity of vascular wall during N(G)-nitro-L-arginine methyl ester-induced hypertension: modulation of proinflammatory signals. Hypertension. 2000; 36:103–109. PMID: 10904020.17. Tedgui A, Mallat Z. Anti-inflammatory mechanisms in the vascular wall. Circ Res. 2001; 88:877–887. PMID: 11348996.
Article18. Szabó G, Veres G, Radovits T, Haider H, Krieger N, Bährle S, Miesel-Gröschel C, Niklisch S, Karck M, van de Locht A. Effects of novel synthetic serine protease inhibitors on postoperative blood loss, coagulation parameters, and vascular relaxation after cardiac surgery. J Thorac Cardiovasc Surg. 2010; 139:181–188. discussion 188. PMID: 20106364.
Article19. Chao J, Stallone JN, Liang YM, Chen LM, Wang DZ, Chao L. Kallistatin is a potent new vasodilator. J Clin Invest. 1997; 100:11–17. PMID: 9202051.
Article20. García-Cardeña G, Fan R, Shah V, Sorrentino R, Cirino G, Papapetropoulos A, Sessa WC. Dynamic activation of endothelial nitric oxide synthase by Hsp90. Nature. 1998; 392:821–824. PMID: 9580552.
Article21. Fulton D, Gratton JP, McCabe TJ, Fontana J, Fujio Y, Walsh K, Franke TF, Papapetropoulos A, Sessa WC. Regulation of endothelium-derived nitric oxide production by the protein kinase Akt. Nature. 1999; 399:597–601. PMID: 10376602.
Article22. Mori M, Gotoh T. Regulation of nitric oxide production by arginine metabolic enzymes. Biochem Biophys Res Commun. 2000; 275:715–719. PMID: 10973788.
Article23. Berkowitz DE, White R, Li D, Minhas KM, Cernetich A, Kim S, Burke S, Shoukas AA, Nyhan D, Champion HC, Hare JM. Arginase reciprocally regulates nitric oxide synthase activity and contributes to endothelial dysfunction in aging blood vessels. Circulation. 2003; 108:2000–2006. PMID: 14517171.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Syringaresinol causes vasorelaxation by elevating nitric oxide production through the phosphorylation and dimerization of endothelial nitric oxide synthase
- Oxytocin-induced endothelial nitric oxide dependent vasorelaxation and ERK1/2-mediated vasoconstriction in the rat aorta
- Genistein activates endothelial nitric oxide synthase in broiler pulmonary arterial endothelial cells by an Akt-dependent mechanism
- Role of Endothelium -Derived Relaxing Factor in the Pathogenesis of Coronary Artery Spasm and Its Relationship with Ethanol
- Inhibition of eNOS/sGC/PKG Pathway Decreases Akt Phosphorylation Induced by Kainic Acid in Mouse Hippocampus