Korean Diabetes J.
2008 Feb;32(1):10-20. 10.4093/kdj.2008.32.1.10.
Protective Effects of Lithospermic Acid B on Diabetic Nephropathy in OLETF Rats Comparing with Amlodipine and Losartan
- Affiliations
-
- 1Division of Endocrinology and Metabolism, Department of Internal Medicine, Yonsei University College of Medicine, Korea.
- 2Brain Korea 21 Project for Medical Science, Yonsei University College of Medicine, Korea.
- 3Institute of Endocrine Research, Yonsei University College of Medicine, Korea.
- 4Division of Nephrology, Department of Internal Medicine, Yonsei University College of Medicine, Korea.
- 5Department of Pharmacology, Yonsei University College of Medicine, Korea.
- 6Ewha Womans University College of Pharmacy and Center for Cell Signaling and Drug Discovery Research, Korea.
- 7Department of Chemistry, Yonsei University College of Science, Korea.
- KMID: 2298105
- DOI: http://doi.org/10.4093/kdj.2008.32.1.10
Abstract
-
BACKGROUND: Lithospermic acid B (LAB), an active component isolated from Salvia miltiorrhizae, has been reported to have renoprotective effects in type 1 and type 2 diabetic animal models. We examined the effects of LAB on the prevention of diabetic nephropathy compared with amlodipine, a calcium channel blocker, and losartan, an angiotensin receptor blocker, in Otsuka Long-Evans-Tokushima Fatty (OLETF) rats, an animal model of type 2 diabetes.
METHODS
LAB (20 mg/kg), amlodipine (10 mg/kg), or losartan (10 mg/kg) was given orally once daily to 10-week-old male OLETF rats for 28 weeks.
RESULTS
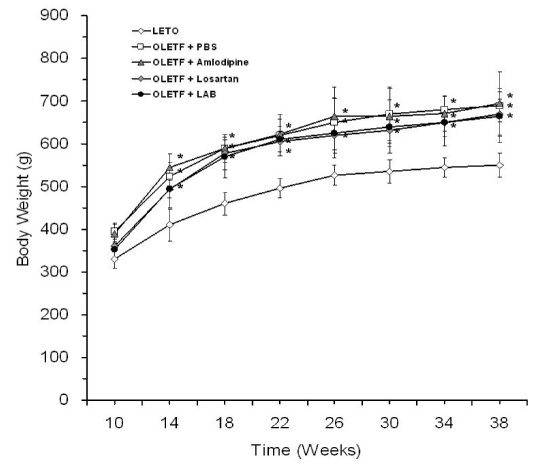
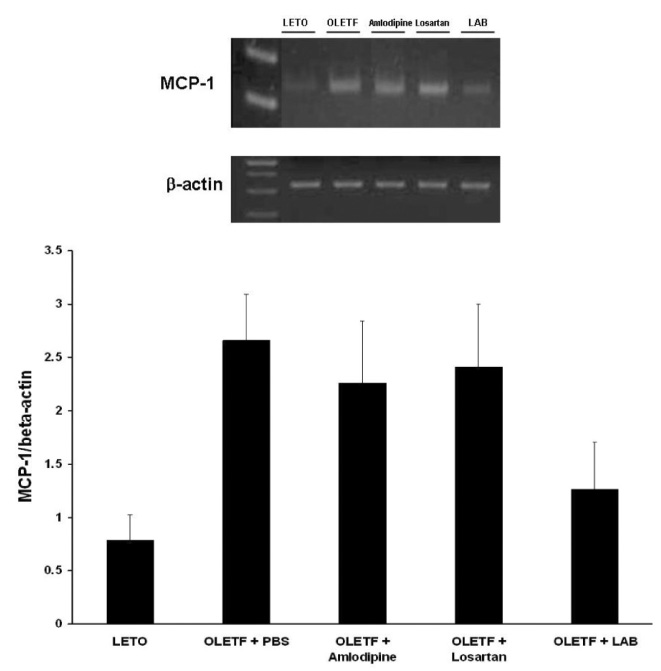
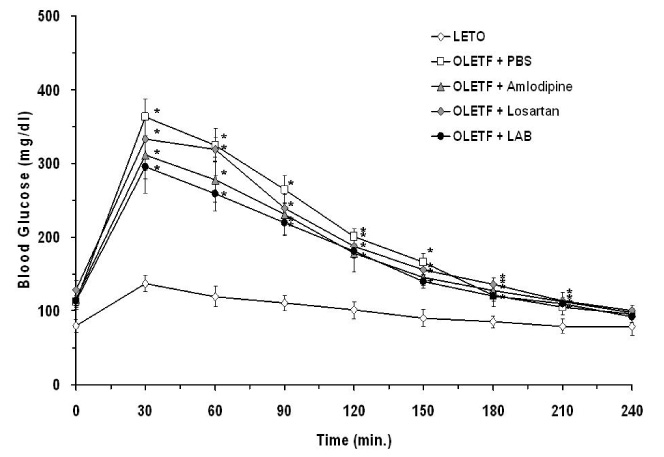
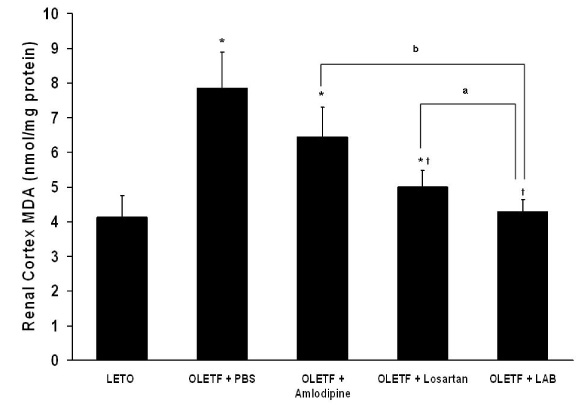
None of LAB, losartan, and amlodipine exhibited effects on blood glucose levels. Treatment with amlodipine or losartan resulted in similar reductions in blood pressure; however, LAB was less effective in lowering blood pressure. Albuminuria was markedly suppressed by losartan and LAB, but not by amlodipine. LAB treatment decreased levels of renal lipid peroxidation, monocyte chemoattractant protein-1 (MCP-1), and transforming growth factor-beta1 (TGF-beta1).
CONCLUSION
These results suggest that LAB has beneficial effects on the diabetic nephropathy in OLETF rats by decreasing oxidative stress and inflammation as potent as losartan.
MeSH Terms
-
Albuminuria
Amlodipine
Angiotensins
Animals
Benzofurans
Blood Glucose
Blood Pressure
Calcium Channels
Chemokine CCL2
Depsides
Diabetic Nephropathies
Humans
Inflammation
Lipid Peroxidation
Losartan
Male
Models, Animal
Oxidative Stress
Pyridines
Rats
Rats, Inbred OLETF
Salvia miltiorrhiza
Thiazoles
Amlodipine
Angiotensins
Benzofurans
Blood Glucose
Calcium Channels
Chemokine CCL2
Depsides
Losartan
Pyridines
Thiazoles
Figure
Reference
-
1. Mauer SM, Steffes MW, Ellis EN, Sutherland DE, Brown DM, Goetz FC. Structural-functional relationships in diabetic nephropathy. J Clin Invest. 1984. 74:1143–1155.2. Ziyadeh FN. Mediators of diabetic renal disease: the case for TGF-β as the major mediator. J Am Soc Nephrol. 2004. 15:Suppl 1. S55–S57.3. Brosius FC 3rd. Trophic factors and cytokines in early diabetic glomerulopathy. Exp Diabesity Res. 2003. 4:225–233.4. Whiteside CI, Thompson J. The role of angiotensin-II in progressive diabetic glomerulopathy in the rat. Endocrinology. 1989. 125:1932–1940.5. Ha H, Yu MR, Choi YJ, Kitamura M, Lee HB. Role of high glucose-induced nuclear factor-kappaB activation in monocyte chemoattractant protein-1 expression by mesangial cells. J Am Soc Nephrol. 2002. 13:894–902.6. Ha H, Lee HB. Reactive oxygen species as glucose signaling molecules in mesangial cells cultured under high glucose. Kidney Int Suppl. 2000. 77:S19–S25.7. Ziyadeh FN, Sharma K, Ericksen M, Wolf G. Stimulation of collagen gene expression and protein synthesis in murine mesangial cells by high glucose is mediated by autocrine activation of transforming growth factor-beta. J Clin Invest. 1994. 93:536–542.8. Oh JH, Ha H, Yu MR, Lee HB. Sequential effects of high glucose on mesangial cell transforming growth factor-beta 1 and fibronectin synthesis. Kidney Int. 1998. 54:1872–1878.9. Ha H, Lee SH, Kim KH. Effects of rebamipide in a model of experimental diabetes and on the synthesis of transforming growth factor-beta and fibronectin, and lipid peroxidation induced by high glucose in cultured mesangial cells. J Pharmacol Exp Ther. 1997. 281:1457–1462.10. Yokozawa T, Dong E, Oura H, Kashiwagi H, Nonaka G, Nishioka I. Magnesium lithospermate B suppresses the increase of active oxygen in rats after subtotal nephrectomy. Nephron. 1997. 75:88–93.11. Kamata K, Iizuka T, Nagai M, Kasuya Y. Endothelium-dependent vasodilator effects of the extract from Salviae Miltiorrhizae radix. A study on the identification of lithospermic acid B in the extracts. Gen Pharmacol. 1993. 24:977–981.12. Uehara Y, Hirawa N, Numabe A, Kawabata Y, Nagoshi H, Negoro H, Fujiwara S, Gomi T, Ikeda T, Goto A, Omata M. Angiotensin-Converting Enzyme Inhibition Delays Onset of Glucosuria With Regression of Renal Injuries in Genetic Rat Model of Non-Insulin-Dependent Diabetes Mellitus. J Cardiovasc Pharmacol Ther. 1998. 3:327–336.13. Koga K, Yamagishi S, Takeuchi M, Inagaki Y, Amano S, Okamoto T, Saga T, Makita Z, Yoshizuka M. CS-886, a new angiotensin II type 1 receptor antagonist, ameliorates glomerular anionic site loss and prevents progression of diabetic nephropathy in Otsuka Long-Evans Tokushima fatty rats. Mol Med. 2002. 8:591–599.14. Okada M, Takemura T, Yanagida H, Yoshioka K. Response of mesangial cells to low-density lipoprotein and angiotensin II in diabetic (OLETF) rats. Kidney Int. 2002. 61:113–124.15. Lee GT, Ha H, Jung M, Li H, Hong SW, Cha BS, Lee HC, Cho YD. Delayed treatment with lithospermate B attenuates experimental diabetic renal injury. J Am Soc Nephrol. 2003. 14:709–720.16. Kang ES, Lee GT, Kim BS, Kim CH, Seo GH, Han SJ, Hur KY, Ahn CW, Ha H, Jung M, Ahn YS, Cha BS, Lee HC. Lithospermic acid B ameliorates the development of diabetic nephropathy in OLETF rats. Eur J Pharmacol. 2008. 579:418–425.17. Khattab M, Ahmad M, Al-Shabanah OA, Raza M. Effects of losartan on blood pressure, oxidative stress, and nitrate/nitrite levels in the nitric oxide deficient hypertensive rats. Receptors Channels. 2004. 10:147–157.18. Naelten G, Liu KL, Chapuis B, Lo M. Persistent effects on blood pressure and renal function of perindopril alone or combined with losartan in Lyon hypertensive rats. Am J Hypertens. 2005. 18:699–706.19. Zhou MS, Jaimes EA, Raij L. Inhibition of oxidative stress and improvement of endothelial function by amlodipine in angiotensin II-infused rats. Am J Hypertens. 2004. 17:167–171.20. Yu G, Liang X, Xie X, Su M, Zhao S. Diverse effects of chronic treatment with losartan, fosinopril, and amlodipine on apoptosis, angiotensin II in the left ventricle of hypertensive rats. Int J Cardiol. 2001. 81:123–129. discussion 129-30.21. Jung M, Lee HC, Ahn CW, Park W, Choi S, Kim H, Cho D, Lee GT, Li HR. Effective isolation of magnesium lithospermate B and its inhibition of aldose reductase and fibronectin on mesangial cell line. Chem Pharm Bull (Tokyo). 2002. 50:1135–1136.22. Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 1979. 95:351–358.23. Evans JL, Goldfine ID, Maddux BA, Grodsky GM. Oxidative stress and stress-activated signaling pathways: a unifying hypothesis of type 2 diabetes. Endocr Rev. 2002. 23:599–622.24. Kawano K, Hirashima T, Mori S, Saitoh Y, Kurosumi M, Natori T. Spontaneous long-term hyperglycemic rat with diabetic complications. Otsuka Long-Evans Tokushima Fatty (OLETF) strain. Diabetes. 1992. 41:1422–1428.25. Kamata K, Noguchi M, Nagai M. Hypotensive effects of lithospermic acid B isolated from the extract of Salviae miltiorrhizae Radix in the rat. Gen Pharmacol. 1994. 25:69–73.26. Chander PN, Gealekman O, Brodsky SV, Elitok S, Tojo A, Crabtree M, Gross SS, Goligorsky MS. Nephropathy in Zucker diabetic fat rat is associated with oxidative and nitrosative stress: prevention by chronic therapy with a peroxynitrite scavenger ebselen. J Am Soc Nephrol. 2004. 15:2391–2403.27. Ha H, Yoon SJ, Kim KH. High glucose can induce lipid peroxidation in the isolated rat glomeruli. Kidney Int. 1994. 46:1620–1626.28. Ha H, Lee HB. Oxidative stress in diabetic nephropathy: basic and clinical information. Curr Diab Rep. 2001. 1:282–287.29. Piconi L, Quagliaro L, Ceriello A. Oxidative stress in diabetes. Clin Chem Lab Med. 2003. 41:1144–1149.30. Lykkesfeldt J. Malondialdehyde as biomarker of oxidative damage to lipids caused by smoking. Clin Chim Acta. 2007. 380:50–58.31. Del Rio D, Stewart AJ, Pellegrini N. A review of recent studies on malondialdehyde as toxic molecule and biological marker of oxidative stress. Nutr Metab Cardiovasc Dis. 2005. 15:316–328.32. Takaishi H, Taniguchi T, Takahashi A, Ishikawa Y, Yokoyama M. High glucose accelerates MCP-1 production via p38 MAPK in vascular endothelial cells. Biochem Biophys Res Commun. 2003. 305:122–128.33. Tanifuji C, Suzuki Y, Geot WM, Horikoshi S, Sugaya T, Ruiz-Ortega M, Egido J, Tomino Y. Reactive oxygen species-mediated signaling pathways in angiotensin II-induced MCP-1 expression of proximal tubular cells. Antioxid Redox Signal. 2005. 7:1261–1268.34. Tejera N, Gomez-Garre D, Lazaro A, Gallego-Delgado J, Alonso C, Blanco J, Ortiz A, Egido J. Persistent proteinuria up-regulates angiotensin II type 2 receptor and induces apoptosis in proximal tubular cells. Am J Pathol. 2004. 164:1817–1826.35. Viedt C, Orth SR. Monocyte chemoattractant protein-1 (MCP-1) in the kidney: does it more than simply attract monocytes? Nephrol Dial Transplant. 2002. 17:2043–2047.36. Chow FY, Nikolic-Paterson DJ, Ozols E, Atkins RC, Rollin BJ, Tesch GH. Monocyte chemoattractant protein-1 promotes the development of diabetic renal injury in streptozotocin-treated mice. Kidney Int. 2006. 69:73–80.37. Di Mario U, Pugliese G. 15th Golgi lecture: from hyperglycaemia to the dysregulation of vascular remodelling in diabetes. Diabetologia. 2001. 44:674–692.38. Ikezumi Y, Hurst LA, Masaki T, Atkins RC, Nikolic-Paterson DJ. Adoptive transfer studies demonstrate that macrophages can induce proteinuria and mesangial cell proliferation. Kidney Int. 2003. 63:83–95.39. Ceol M, Vianello D, Schleicher E, Anglani F, Barbanti M, Bonfante L, Bertaglia G, Graziotto R, D'Angelo A, Del Prete D, Gambaro G. Heparin reduces glomerular infiltration and TGF-beta protein expression by macrophages in puromycin glomerulosclerosis. J Nephrol. 2003. 16:210–218.40. Tesch GH, Maifert S, Schwarting A, Rollins BJ, Kelley VR. Monocyte chemoattractant protein 1-dependent leukocytic infiltrates are responsible for autoimmune disease in MRL-Fas(lpr) mice. J Exp Med. 1999. 190:1813–1824.41. Tesch GH, Schwarting A, Kinoshita K, Lan HY, Rollins BJ, Kelley VR. Monocyte chemoattractant protein-1 promotes macrophage-mediated tubular injury, but not glomerular injury, in nephrotoxic serum nephritis. J Clin Invest. 1999. 103:73–80.42. Wolf G, Schneider A, Helmchen U, Stahl RA. AT1-receptor antagonists abolish glomerular MCP-1 expression in a model of mesangial proliferative glomerulonephritis. Exp Nephrol. 1998. 6:112–120.43. Cheng J, Diaz Encarnacion MM, Warner GM, Gray CE, Nath KA, Grande JP. TGF-beta1 stimulates monocyte chemoattractant protein-1 expression in mesangial cells through a phosphodiesterase isoenzyme 4-dependent process. Am J Physiol Cell Physiol. 2005. 289:C959–C970.44. Schneider A, Panzer U, Zahner G, Wenzel U, Wolf G, Thaiss F, Helmchen U, Stahl RA. Monocyte chemoattractant protein-1 mediates collagen deposition in experimental glomerulonephritis by transforming growth factor-beta. Kidney Int. 1999. 56:135–144.45. Duncan MR, Frazier KS, Abramson S, Williams S, Klapper H, Huang X, Grotendorst GR. Connective tissue growth factor mediates transforming growth factor beta-induced collagen synthesis: down-regulation by cAMP. Faseb J. 1999. 13:1774–1786.46. Riser BL, Denichilo M, Cortes P, Baker C, Grondin JM, Yee J, Narins RG. Regulation of connective tissue growth factor activity in cultured rat mesangial cells and its expression in experimental diabetic glomerulosclerosis. J Am Soc Nephrol. 2000. 11:25–38.47. Zhou L, Zuo Z, Chow MS. Danshen: an overview of its chemistry, pharmacology, pharmacokinetics, and clinical use. J Clin Pharmacol. 2005. 45:1345–1359.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Spironolactone and Losartan on Diabetic Nephropathy in a Type 2 Diabetic Rat Model
- Protective Effects of Lithospermate B on Diabetic Nephropathy in OLETF Rat
- Diabetic Nephropathy - Preventive effects of lithospermic acid B (LAB)
- Effects of ferulic acid on diabetic nephropathy in a rat model of type 2 diabetes
- Beneficial Effects of Thiazolidinediones on Diabetic Nephropathy in OLETF Rats