Korean J Physiol Pharmacol.
2015 Jul;19(4):327-334. 10.4196/kjpp.2015.19.4.327.
Involvement of Heme Oxygenase-1 in Orexin-A-induced Angiogenesis in Vascular Endothelial Cells
- Affiliations
-
- 1Department of Oral Physiology, School of Dentistry, Pusan National University, Yangsan 626-870, Korea. mkbae@pusan.ac.kr
- 2Department of Dental Pharmacology, School of Dentistry, Pusan National University, Yangsan 626-870, Korea.
- 3Neuroprotection Research Laboratory, Department of Radiology and Neurology, Massachusetts General Hospital and Harvard Medical School, Charlestown, MA 02129, USA.
- KMID: 2285575
- DOI: http://doi.org/10.4196/kjpp.2015.19.4.327
Abstract
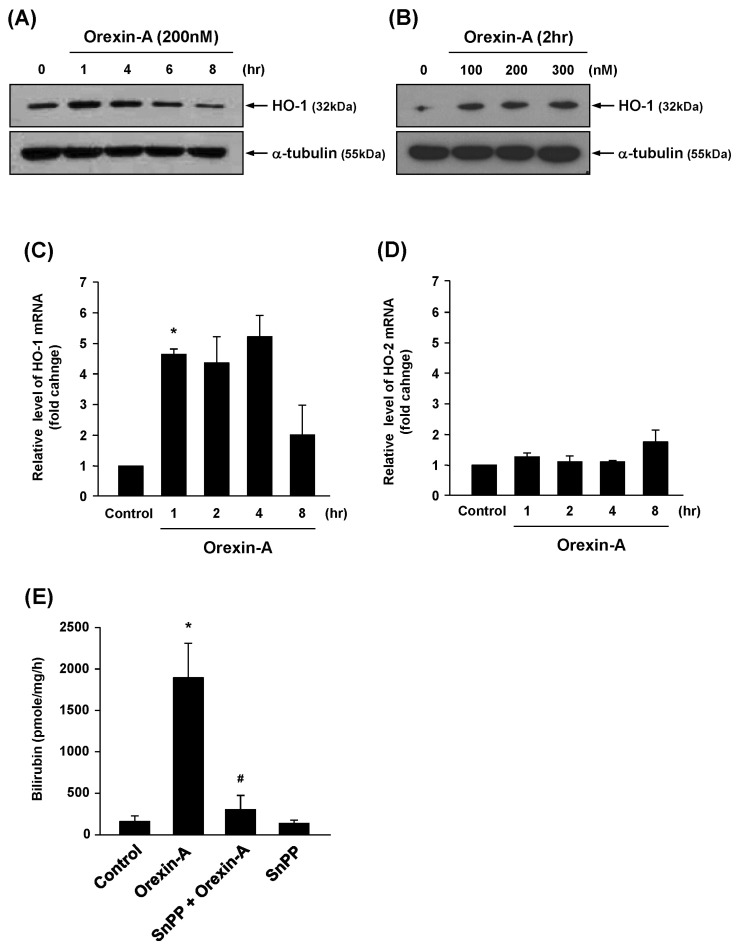
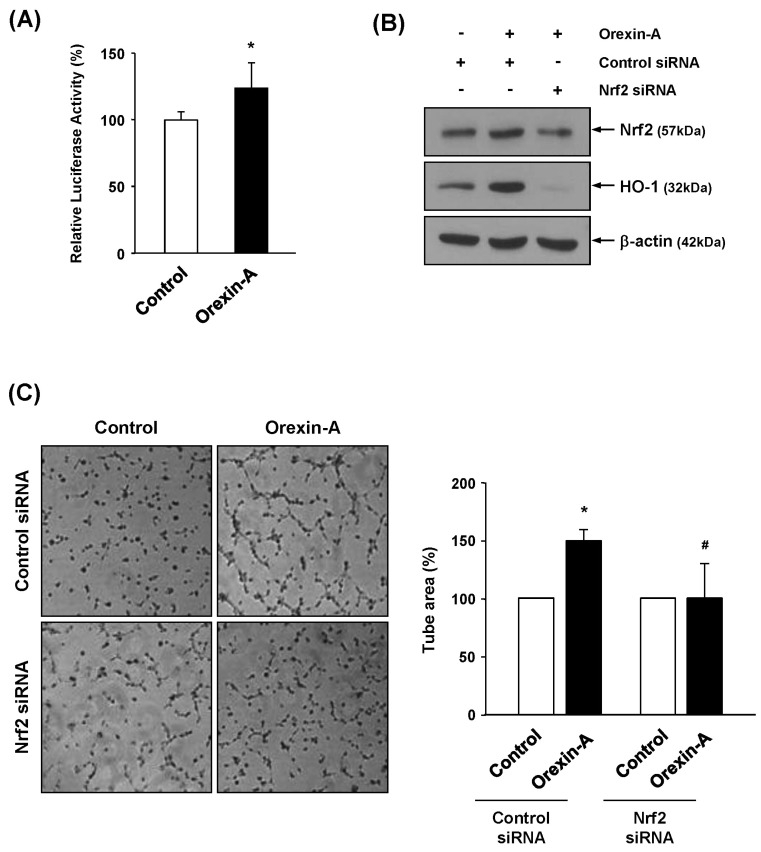
- The cytoprotective enzyme heme oxygenase-1 (HO-1) influences endothelial cell survival, proliferation, inflammatory response, and angiogenesis in response to various angiogenic stimuli. In this study, we investigate the involvement of HO-1 in the angiogenic activity of orexin-A. We showed that orexin-A stimulates expression and activity of HO-1 in human umbilical vein endothelial cells (HUVECs). Furthermore, we showed that inhibition of HO-1 by tin (Sn) protoporphryin-IX (SnPP) reduced orexin-A-induced angiogenesis in vivo and ex vivo. Orexin-A-stimulated endothelial tube formation and chemotactic activity were also blocked in SnPP-treated vascular endothelial cells. Orexin-A treatment increased the expression of nuclear factor erythroid-derived 2 related factor 2 (Nrf2), and antioxidant response element (ARE) luciferase activity, leading to induction of HO-1. Collectively, these findings indicate that HO-1 plays a role as an important mediator of orexin-A-induced angiogenesis, and provide new possibilities for therapeutic approaches in pathophysiological conditions associated with angiogenesis.
MeSH Terms
Figure
Cited by 1 articles
-
Advanced tube formation assay using human endothelial colony forming cells for
in vitro evaluation of angiogenesis
Hyunsook Lee, Kyu-Tae Kang
Korean J Physiol Pharmacol. 2018;22(6):705-712. doi: 10.4196/kjpp.2018.22.6.705.
Reference
-
1. de Lecea L, Kilduff TS, Peyron C, Gao X, Foye PE, Danielson PE, Fukuhara C, Battenberg EL, Gautvik VT, Bartlett FS 2nd, Frankel WN, van den Pol AN, Bloom FE, Gautvik KM, Sutcliffe JG. The hypocretins: hypothalamus-specific peptides with neuroexcitatory activity. Proc Natl Acad Sci U S A. 1998; 95:322–327. PMID: 9419374.
Article2. Sakurai T, Amemiya A, Ishii M, Matsuzaki I, Chemelli RM, Tanaka H, Williams SC, Richardson JA, Kozlowski GP, Wilson S, Arch JR, Buckingham RE, Haynes AC, Carr SA, Annan RS, McNulty DE, Liu WS, Terrett JA, Elshourbagy NA, Bergsma DJ, Yanagisawa M. Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell. 1998; 92:573–585. PMID: 9491897.
Article3. Sutcliffe JG, de Lecea L. The hypocretins: excitatory neuromodulatory peptides for multiple homeostatic systems, including sleep and feeding. J Neurosci Res. 2000; 62:161–168. PMID: 11020209.
Article4. Jochem J, Zwirska-Korczala K, Zabielski R, Kato I, Kuwahara A. Cardiovascular effects of centrally acting orexin A in haemorrhage-shocked rats. J Physiol Pharmacol. 2006; 57(Suppl 11):115–124.5. Tanida M, Niijima A, Shen J, Yamada S, Sawai H, Fukuda Y, Nagai K. Dose-different effects of orexin-A on the renal sympathetic nerve and blood pressure in urethane-anesthetized rats. Exp Biol Med (Maywood). 2006; 231:1616–1625. PMID: 17060682.
Article6. Kim MK, Park HJ, Kim SR, Choi YK, Shin HK, Jeon JH, Jang HO, Yun I, Bae SK, Bae MK. Angiogenic role of orexin-A via the activation of extracellular signal-regulated kinase in endothelial cells. Biochem Biophys Res Commun. 2010; 403:59–65. PMID: 21040705.
Article7. Carmeliet P. Angiogenesis in health and disease. Nat Med. 2003; 9:653–660. PMID: 12778163.
Article8. Tonini T, Rossi F, Claudio PP. Molecular basis of angiogenesis and cancer. Oncogene. 2003; 22:6549–6556. PMID: 14528279.
Article9. Ribatti D, Conconi MT, Nussdorfer GG. Nonclassic endogenous novel [corrected] regulators of angiogenesis. Pharmacol Rev. 2007; 59:185–205. PMID: 17540906.10. Maines MD. The heme oxygenase system: a regulator of second messenger gases. Annu Rev Pharmacol Toxicol. 1997; 37:517–554. PMID: 9131263.11. Agarwal A, Nick HS. Renal response to tissue injury: lessons from heme oxygenase-1 GeneAblation and expression. J Am Soc Nephrol. 2000; 11:965–973. PMID: 10770977.12. Soares MP, Brouard S, Smith RN, Bach FH. Heme oxygenase-1, a protective gene that prevents the rejection of transplanted organs. Immunol Rev. 2001; 184:275–285. PMID: 12086318.
Article13. Wagener FA, da Silva JL, Farley T, de Witte T, Kappas A, Abraham NG. Differential effects of heme oxygenase isoforms on heme mediation of endothelial intracellular adhesion molecule 1 expression. J Pharmacol Exp Ther. 1999; 291:416–423. PMID: 10490932.14. Duckers HJ, Boehm M, True AL, Yet SF, San H, Park JL, Clinton Webb R, Lee ME, Nabel GJ, Nabel EG. Heme oxygenase-1 protects against vascular constriction and proliferation. Nat Med. 2001; 7:693–698. PMID: 11385506.
Article15. Deramaudt BM, Braunstein S, Remy P, Abraham NG. Gene transfer of human heme oxygenase into coronary endothelial cells potentially promotes angiogenesis. J Cell Biochem. 1998; 68:121–127. PMID: 9407320.
Article16. Park SY, Bae JU, Hong KW, Kim CD. HO-1 induced by cilostazol protects against TNF-α-associated cytotoxicity via a PPAR-γ-dependent pathway in human endothelial cells. Korean J Physiol Pharmacol. 2011; 15:83–88. PMID: 21660147.
Article17. Deshane J, Chen S, Caballero S, Grochot-Przeczek A, Was H, Li Calzi S, Lach R, Hock TD, Chen B, Hill-Kapturczak N, Siegal GP, Dulak J, Jozkowicz A, Grant MB, Agarwal A. Stromal cell-derived factor 1 promotes angiogenesis via a heme oxygenase 1-dependent mechanism. J Exp Med. 2007; 204:605–618. PMID: 17339405.
Article18. Balla G, Jacob HS, Balla J, Rosenberg M, Nath K, Apple F, Eaton JW, Vercellotti GM. Ferritin: a cytoprotective antioxidant strategem of endothelium. J Biol Chem. 1992; 267:18148–18153. PMID: 1517245.
Article19. Sakurai T. Orexin: a link between energy homeostasis and adaptive behaviour. Curr Opin Clin Nutr Metab Care. 2003; 6:353–360. PMID: 12806206.20. Heinonen MV, Purhonen AK, Mäkelä KA, Herzig KH. Functions of orexins in peripheral tissues. Acta Physiol (Oxf). 2008; 192:471–485. PMID: 18294339.
Article21. Kastin AJ, Akerstrom V. Orexin A but not orexin B rapidly enters brain from blood by simple diffusion. J Pharmacol Exp Ther. 1999; 289:219–223. PMID: 10087007.22. Arihara Z, Takahashi K, Murakami O, Totsune K, Sone M, Satoh F, Ito S, Mouri T. Immunoreactive orexin-A in human plasma. Peptides. 2001; 22:139–142. PMID: 11179609.
Article23. Sun G, Tian Z, Yao Y, Li H, Higuchi T. Central and/or peripheral immunoreactivity of orexin-A in pregnant rats and women. J Mol Endocrinol. 2006; 36:131–138. PMID: 16461933.
Article24. Kitamura E, Hamada J, Kanazawa N, Yonekura J, Masuda R, Sakai F, Mochizuki H. The effect of orexin-A on the pathological mechanism in the rat focal cerebral ischemia. Neurosci Res. 2010; 68:154–157. PMID: 20600373.
Article25. Esmaeili-Mahani S, Vazifekhah S, Pasban-Aliabadi H, Abbasnejad M, Sheibani V. Protective effect of orexin-A on 6-hydroxydopamine-induced neurotoxicity in SH-SY5Y human dopaminergic neuroblastoma cells. Neurochem Int. 2013; 63:719–725. PMID: 24135219.
Article26. Brzozowski T, Konturek PC, Sliwowski Z, Drozdowicz D, Burnat G, Pajdo R, Pawlik M, Bielanski W, Kato I, Kuwahara A, Konturek SJ, Pawlik WW. Gastroprotective action of orexin-A against stress-induced gastric damage is mediated by endogenous prostaglandins, sensory afferent neuropeptides and nitric oxide. Regul Pept. 2008; 148:6–20. PMID: 18378017.
Article27. Dreger H, Westphal K, Weller A, Baumann G, Stangl V, Meiners S, Stangl K. Nrf2-dependent upregulation of antioxidative enzymes: a novel pathway for proteasome inhibitor-mediated cardioprotection. Cardiovasc Res. 2009; 83:354–361. PMID: 19351736.
Article28. Dulak J, Deshane J, Jozkowicz A, Agarwal A. Heme oxygenase-1 and carbon monoxide in vascular pathobiology: focus on angiogenesis. Circulation. 2008; 117:231–241. PMID: 18195184.29. Lakkisto P, Kytö V, Forsten H, Siren JM, Segersvärd H, Voipio-Pulkki LM, Laine M, Pulkki K, Tikkanen I. Heme oxygenase-1 and carbon monoxide promote neovascularization after myocardial infarction by modulating the expression of HIF-1alpha, SDF-1alpha and VEGF-B. Eur J Pharmacol. 2010; 635:156–164. PMID: 20303947.30. Choi YK, Kim CK, Lee H, Jeoung D, Ha KS, Kwon YG, Kim KW, Kim YM. Carbon monoxide promotes VEGF expression by increasing HIF-1alpha protein level via two distinct mechanisms, translational activation and stabilization of HIF-1alpha protein. J Biol Chem. 2010; 285:32116–32125. PMID: 20724477.31. Sikder D, Kodadek T. The neurohormone orexin stimulates hypoxia-inducible factor-1 activity. Genes Dev. 2007; 21:2995–3005. PMID: 18006690.
Article32. Wang Z, Liu S, Kakizaki M, Hirose Y, Ishikawa Y, Funato H, Yanagisawa M, Yu Y, Liu Q. Orexin/hypocretin activates mTOR complex 1 (mTORC1) via an Erk/Akt-independent and calcium-stimulated lysosome v-ATPase pathway. J Biol Chem. 2014; 289:31950–31959. PMID: 25278019.
Article33. Laplante M, Sabatini DM. mTOR signaling in growth control and disease. Cell. 2012; 149:274–293. PMID: 22500797.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Role of Carbon Monoxide in Neurovascular Repair Processing
- Heme Oxygenase-1: Its Therapeutic Roles in Inflammatory Diseases
- Heme Oxygenase-1 Induced by Aprotinin Inhibits Vascular Smooth Muscle Cell Proliferation Through Cell Cycle Arrest in Hypertensive Rats
- Effect of heme oxygenase induction by NO donor on the aortic contractility
- Mitochondrial Channel Opener Diazoxide Attenuates Hypoxia-Induced sFlt-1 Release in Human Choriocarcinoma Cells