Korean J Physiol Pharmacol.
2014 Feb;18(1):79-86. 10.4196/kjpp.2014.18.1.79.
Phosphorylation of Akt Mediates Anti-Inflammatory Activity of 1-p-Coumaroyl beta-D-Glucoside Against Lipopolysaccharide-Induced Inflammation in RAW264.7 Cells
- Affiliations
-
- 1Department of Pharmacology, College of Medicine, Kangwon National University, Chuncheon 200-701, Korea. wchun@kangwon.ac.kr
- 2College of Pharmacy, Kangwon National University, Chuncheon 200-701, Korea.
- KMID: 2285495
- DOI: http://doi.org/10.4196/kjpp.2014.18.1.79
Abstract
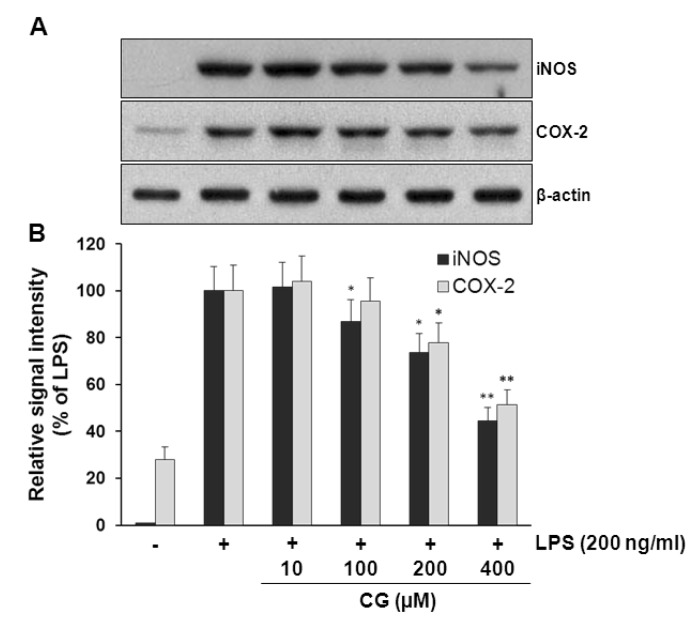
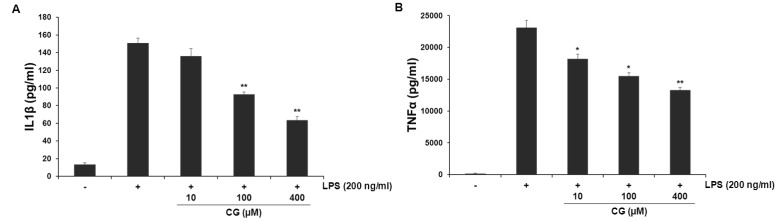
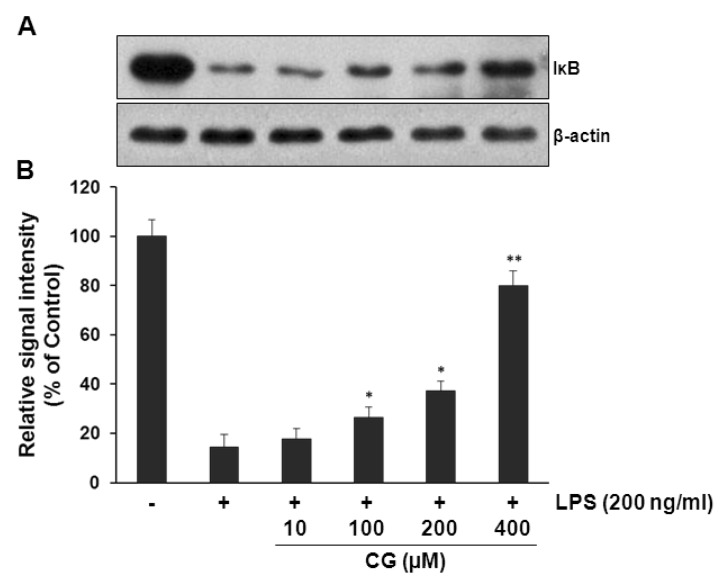
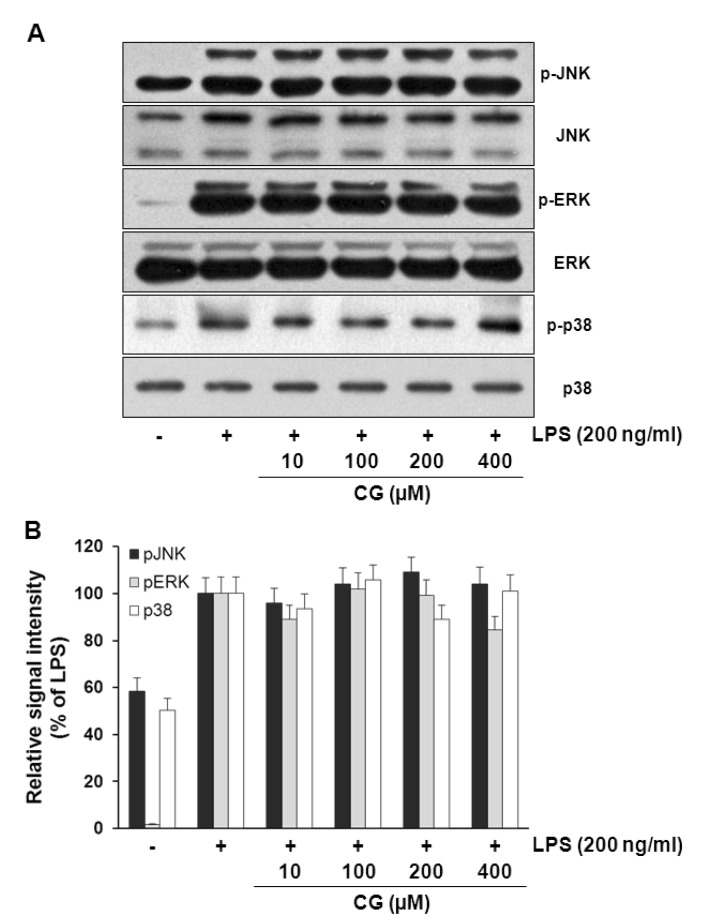
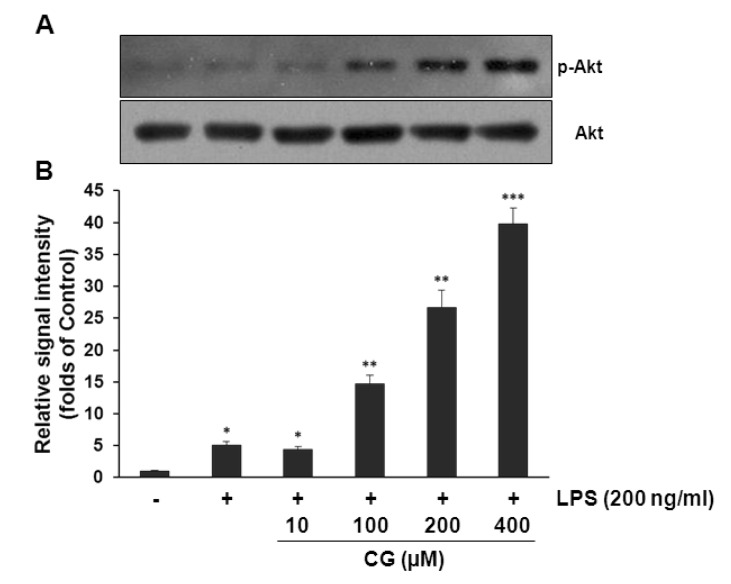
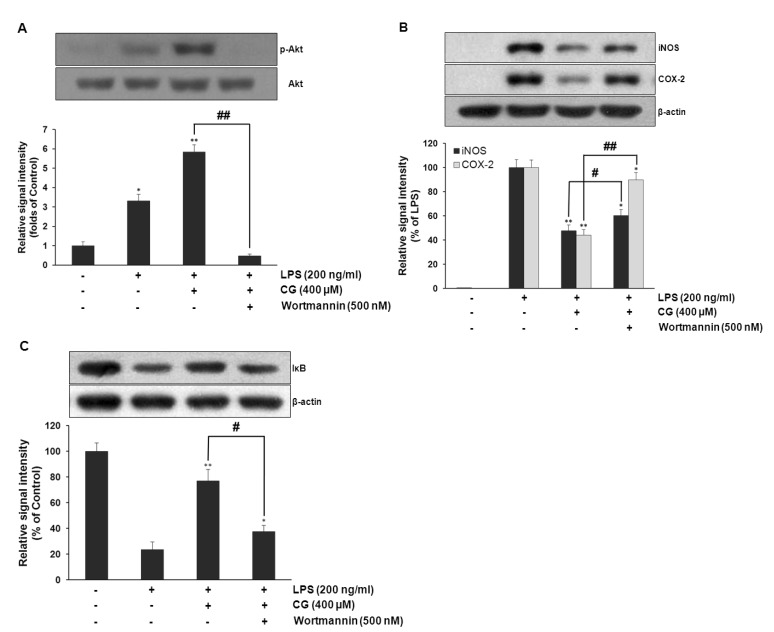
- Hydroxycinnamic acids have been reported to possess numerous pharmacological activities such as antioxidant, anti-inflammatory, and anti-tumor properties. However, the biological activity of 1-p-coumaroyl beta-D-glucoside (CG), a glucose ester derivative of p-coumaric acid, has not been clearly examined. The objective of this study is to elucidate the anti-inflammatory action of CG in lipopolysaccharide (LPS)-stimulated RAW264.7 macrophage cells. In the present study, CG significantly suppressed LPS-induced excessive production of pro-inflammatory mediators such as nitric oxide (NO) and PGE2 and the protein expression of iNOS and COX-2. CG also inhibited LPS-induced secretion of pro-inflammatory cytokines, IL-1beta and TNF-alpha. In addition, CG significantly suppressed LPS-induced degradation of IkappaB. To elucidate the underlying mechanism by which CG exerts its anti-inflammatory action, involvement of various signaling pathways were examined. CG exhibited significantly increased Akt phosphorylation in a concentration-dependent manner, although MAPKs such as Erk, JNK, and p38 appeared not to be involved. Furthermore, inhibition of Akt/PI3K signaling pathway with wortmannin significantly, albeit not completely, abolished CG-induced Akt phosphorylation and anti-inflammatory actions. Taken together, the present study demonstrates that Akt signaling pathway might play a major role in CG-mediated anti-inflammatory activity in LPS-stimulated RAW264.7 macrophage cells.
Keyword
MeSH Terms
Figure
Reference
-
1. Nagasaka R, Chotimarkorn C, Shafiqul IM, Hori M, Ozaki H, Ushio H. Anti-inflammatory effects of hydroxycinnamic acid derivatives. Biochem Biophys Res Commun. 2007; 358:615–619. PMID: 17499610.
Article2. Steinbrecher T, Hrenn A, Dormann KL, Merfort I, Labahn A. Bornyl (3,4,5-trihydroxy)-cinnamate--an optimized human neutrophil elastase inhibitor designed by free energy calculations. Bioorg Med Chem. 2008; 16:2385–2390. PMID: 18078761.3. Kim YC. Neuroprotective phenolics in medicinal plants. Arch Pharm Res. 2010; 33:1611–1632. PMID: 21052937.
Article4. Teixeira J, Gaspar A, Garrido EM, Garrido J, Borges F. Hydroxycinnamic acid antioxidants: an electrochemical overview. Biomed Res Int. 2013; 2013:251754. PMID: 23956973.
Article5. Lee JW, Bae CJ, Choi YJ, Kim SI, Kim NH, Lee HJ, Kim SS, Kwon YS, Chun W. 3,4,5-trihydroxycinnamic acid inhibits LPS-Induced iNOS expression by suppressing NF-κB activation in BV2 microglial cells. Korean J Physiol Pharmacol. 2012; 16:107–112. PMID: 22563255.
Article6. Lee JW, Choi YJ, Park JH, Sim JY, Kwon YS, Lee HJ, Kim SS, Chun W. 3,4,5-trihydroxycinnamic acid inhibits lipopolysaccharide-induced inflammatory response through the activation of Nrf2 pathway in BV2 microglial cells. Biomol Ther (Seoul). 2013; 21:60–65. PMID: 24009860.
Article7. Lee JW, Cheong IY, Kim HS, Lee JJ, Lee YS, Kwon YS, Kim MJ, Lee HJ, Kim SS, Chun W. Anti-inflammatory activity of 1-docosanoyl cafferate isolated from rhus verniciflua in LPS-stimulated BV2 microglial cells. Korean J Physiol Pharmacol. 2011; 15:9–15. PMID: 21461235.8. Lee Y, Shin DH, Kim JH, Hong S, Choi D, Kim YJ, Kwak MK, Jung Y. Caffeic acid phenethyl ester-mediated Nrf2 activation and IkappaB kinase inhibition are involved in NFkappaB inhibitory effect: structural analysis for NFkappaB inhibition. Eur J Pharmacol. 2010; 643:21–28. PMID: 20599928.9. Hooper SN, Jurgens T, Chandler RF, Stevens MFG. Methyl p-coumarate: A cytotoxic constituent from Comptonia peregrina. Phytochemistry. 1984; 23:2096–2209.
Article10. Kwon YS, Kim CM. Antioxidant constituents from the stem of Sorghum bicolor. Arch Pharm Res. 2003; 26:535–539. PMID: 12934645.11. Kubo I, Nihei K, Tsujimoto K. Methyl p-coumarate, a melanin formation inhibitor in B16 mouse melanoma cells. Bioorg Med Chem. 2004; 12:5349–5354. PMID: 15388162.
Article12. Lee M, Lee HH, Lee JK, Ye SK, Kim SH, Sung SH. Anti-adipogenic activity of compounds isolated from Idesia polycarpa on 3T3-L1 cells. Bioorg Med Chem Lett. 2013; 23:3170–3174. PMID: 23628332.
Article13. Zhang Y, Seeram NP, Lee R, Feng L, Heber D. Isolation and identification of strawberry phenolics with antioxidant and human cancer cell antiproliferative properties. J Agric Food Chem. 2008; 56:670–675. PMID: 18211028.
Article14. Jeon SH, Chun W, Choi YJ, Kwon YS. Cytotoxic constituents from the bark of Salix hulteni. Arch Pharm Res. 2008; 31:978–982. PMID: 18787784.
Article15. Rehman MU, Yoshihisa Y, Miyamoto Y, Shimizu T. The anti-inflammatory effects of platinum nanoparticles on the lipopolysaccharide-induced inflammatory response in RAW 264.7 macrophages. Inflamm Res. 2012; 61:1177–1185. PMID: 22752115.
Article16. Kim YJ, Shin Y, Lee KH, Kim TJ. Anethum graveloens flower extracts inhibited a lipopolysaccharide-induced inflammatory response by blocking iNOS expression and NF-κB activity in macrophages. Biosci Biotechnol Biochem. 2012; 76:1122–1127. PMID: 22790933.17. Itharat A, Hiransai P. Dioscoreanone suppresses LPS-induced nitric oxide production and inflammatory cytokine expression in RAW 264.7 macrophages by NF-κB and ERK1/2 signaling transduction. J Cell Biochem. 2012; 113:3427–3435. PMID: 22678775.
Article19. Sweet MJ, Hume DA. Endotoxin signal transduction in macrophages. J Leukoc Biol. 1996; 60:8–26. PMID: 8699127.
Article20. Ock J, Kim S, Suk K. Anti-inflammatory effects of a fluorovinyloxyacetamide compound KT-15087 in microglia cells. Pharmacol Res. 2009; 59:414–422. PMID: 19429474.
Article21. Zheng LT, Ryu GM, Kwon BM, Lee WH, Suk K. Anti-inflammatory effects of catechols in lipopolysaccharide-stimulated microglia cells: inhibition of microglial neurotoxicity. Eur J Pharmacol. 2008; 588:106–113. PMID: 18499097.
Article22. Fresco P, Borges F, Diniz C, Marques MP. New insights on the anticancer properties of dietary polyphenols. Med Res Rev. 2006; 26:747–766. PMID: 16710860.
Article23. Razzaghi-Asl N, Garrido J, Khazraei H, Borges F, Firuzi O. Antioxidant properties of hydroxycinnamic acids: a review of structure-activity relationships. Curr Med Chem. 2013; 20:4436–4450. PMID: 23834166.24. Navaneethan D, Rasool M. p-Coumaric acid, a common dietary polyphenol, protects cadmium chloride-induced nephrotoxicity in rats. Ren Fail. 2013; [Epub ahead of print].
Article25. Lee JW, Cheong IY, Kim HS, Lee JJ, Lee YS, Kwon YS, Kim MJ, Lee HJ, Kim SS, Chun W. Anti-inflammatory activity of 1-docosanoyl cafferate isolated from rhus verniciflua in LPS-stimulated BV2 microglial cells. Korean J Physiol Pharmacol. 2011; 15:9–15. PMID: 21461235.26. Fürer K, Raith M, Brenneisen R, Mennet M, Simões-Wüst AP, von Mandach U, Hamburger M, Potterat O. Two new flavonol glycosides and a metabolite profile of Bryophyllum pinnatum, a phytotherapeutic used in obstetrics and gynaecology. Planta Med. 2013; 79:1565–1571. PMID: 24072500.
Article27. Hou L, Zhou B, Yang L, Liu ZL. Inhibition of free radical initiated peroxidation of human erythrocyte ghosts by flavonols and their glycosides. Org Biomol Chem. 2004; 2:1419–1423. PMID: 15105935.
Article28. Vo VA, Lee JW, Chang JE, Kim JY, Kim NH, Lee HJ, Kim SS, Chun W, Kwon YS. Avicularin inhibits lipopolysaccharide-induced inflammatory response by suppressing ERK phosphorylation in RAW 264.7 macrophages. Biomol Ther (Seoul). 2012; 20:532–537. PMID: 24009846.
Article29. Akao T, Kobashi K, Aburada M. Enzymic studies on the animal and intestinal bacterial metabolism of geniposide. Biol Pharm Bull. 1994; 17:1573–1576. PMID: 7735197.
Article30. Arts IC, Sesink AL, Faassen-Peters M, Hollman PC. The type of sugar moiety is a major determinant of the small intestinal uptake and subsequent biliary excretion of dietary quercetin glycosides. Br J Nutr. 2004; 91:841–847. PMID: 15182387.
Article31. Ha YM, Ham SA, Kim YM, Lee YS, Kim HJ, Seo HG, Lee JH, Park MK, Chang KC. β1-adrenergic receptor-mediated HO-1 induction, via PI3K and p38 MAPK, by isoproterenol in RAW 264.7 cells leads to inhibition of HMGB1 release in LPS-activated RAW 264.7 cells and increases in survival rate of CLP-induced septic mice. Biochem Pharmacol. 2011; 82:769–777. PMID: 21763292.32. Jung WK, Lee DY, Park C, Choi YH, Choi I, Park SG, Seo SK, Lee SW, Yea SS, Ahn SC, Lee CM, Park WS, Ko JH, Choi IW. Cilostazol is anti-inflammatory in BV2 microglial cells by inactivating nuclear factor-kappaB and inhibiting mitogen-activated protein kinases. Br J Pharmacol. 2010; 159:1274–1285. PMID: 20128801.
Article33. O'Connell MA, Bennett BL, Mercurio F, Manning AM, Mackman N. Role of IKK1 and IKK2 in lipopolysaccharide signaling in human monocytic cells. J Biol Chem. 1998; 273:30410–30414. PMID: 9804806.34. Guha M, Mackman N. LPS induction of gene expression in human monocytes. Cell Signal. 2001; 13:85–94. PMID: 11257452.
Article35. Lee JW, Kim NH, Kim JY, Park JH, Shin SY, Kwon YS, Lee HJ, Kim SS, Chun W. Aromadendrin inhibits lipopolysaccharide-induced nuclear translocation of NF-κB and phosphorylation of JNK in RAW 264.7 macrophage cells. Biomol Ther (Seoul). 2013; 21:216–221. PMID: 24265867.
Article36. Kuprash DV, Udalova IA, Turetskaya RL, Rice NR, Nedospasov SA. Conserved kappa B element located downstream of the tumor necrosis factor alpha gene: distinct NF-kappa B binding pattern and enhancer activity in LPS activated murine macrophages. Oncogene. 1995; 11:97–106. PMID: 7624137.37. Siebenlist U, Franzoso G, Brown K. Structure, regulation and function of NF-kappa B. Annu Rev Cell Biol. 1994; 10:405–455. PMID: 7888182.38. Li Q, Verma IM. NF-kappaB regulation in the immune system. Nat Rev Immunol. 2002; 2:725–734. PMID: 12360211.39. Karin M, Takahashi T, Kapahi P, Delhase M, Chen Y, Makris C, Rothwarf D, Baud V, Natoli G, Guido F, Li N. Oxidative stress and gene expression: the AP-1 and NF-kappaB connections. Biofactors. 2001; 15:87–89. PMID: 12016332.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- N-(p-Coumaryol)-Tryptamine Suppresses the Activation of JNK/c-Jun Signaling Pathway in LPS-Challenged RAW264.7 Cells
- Luteolin and luteolin-7-O-glucoside inhibit lipopolysaccharide-induced inflammatory responses through modulation of NF-kappaB/AP-1/PI3K-Akt signaling cascades in RAW 264.7 cells
- Methyl p-Hydroxycinnamate Suppresses Lipopolysaccharide-Induced Inflammatory Responses through Akt Phosphorylation in RAW264.7 Cells
- Quercetin-3-O-β-D-Glucuronide Suppresses Lipopolysaccharide-Induced JNK and ERK Phosphorylation in LPS-Challenged RAW264.7 Cells
- Hydroquinone suppresses IFN-β expression by targeting AKT/IRF3 pathway