J Clin Neurol.
2015 Oct;11(4):297-304. 10.3988/jcn.2015.11.4.297.
The Role of the PI3K Pathway in the Regeneration of the Damaged Brain by Neural Stem Cells after Cerebral Infarction
- Affiliations
-
- 1Neuroprotection Research Laboratory, Massachusetts General Hospital, Harvard Medical School, Boston, MA, USA. ksh213@hanyang.ac.kr
- 2Department of Neurology, Hanyang University College of Medicine, Seoul, Korea.
- KMID: 2179748
- DOI: http://doi.org/10.3988/jcn.2015.11.4.297
Abstract
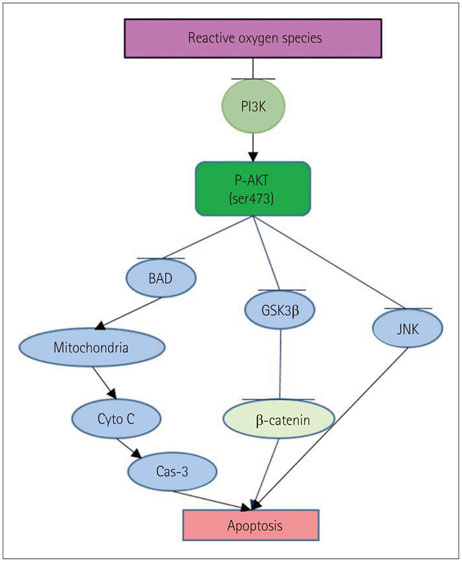
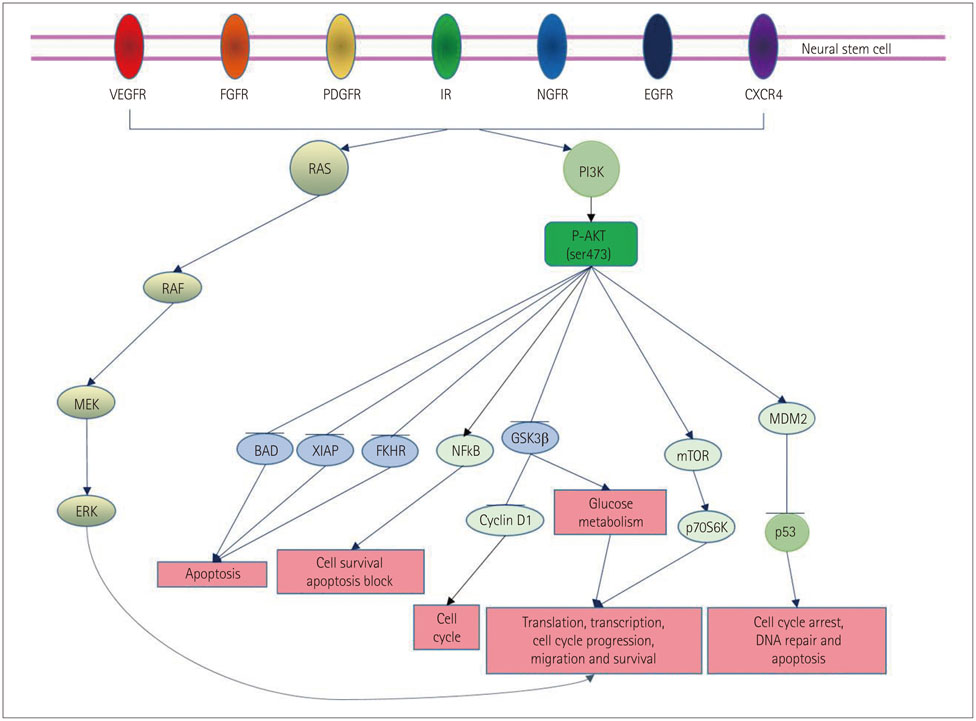
- Neurologic deficits resulting from stroke remain largely intractable, which has prompted thousands of studies aimed at developing methods for treating these neurologic sequelae. Endogenous neurogenesis is also known to occur after brain damage, including that due to cerebral infarction. Focusing on this process may provide a solution for treating neurologic deficits caused by cerebral infarction. The phosphatidylinositol-3-kinase (PI3K) pathway is known to play important roles in cell survival, and many studies have focused on use of the PI3K pathway to treat brain injury after stroke. Furthermore, since the PI3K pathway may also play key roles in the physiology of neural stem cells (NSCs), eliciting the appropriate activation of the PI3K pathway in NSCs may help to improve the sequelae of cerebral infarction. This review describes the PI3K pathway, its roles in the brain and NSCs after cerebral infarction, and the therapeutic possibility of activating the pathway to improve neurologic deficits after cerebral infarction.
MeSH Terms
Figure
Cited by 2 articles
-
The role of PI3K/AKT pathway and its therapeutic possibility in Alzheimer's disease
Hyun-Jung Yu, Seong-Ho Koh
Hanyang Med Rev. 2017;37(1):18-24. doi: 10.7599/hmr.2017.37.1.18.Postischemic Inflammation in Acute Stroke
Simone Vidale, Arturo Consoli, Marco Arnaboldi, Domenico Consoli
J Clin Neurol. 2017;13(1):1-9. doi: 10.3988/jcn.2017.13.1.1.
Reference
-
1. Del Zoppo GJ, Saver JL, Jauch EC, Adams HP Jr. American Heart Association Stroke Council. Expansion of the time window for treatment of acute ischemic stroke with intravenous tissue plasminogen activator: a science advisory from the American Heart Association/American Stroke Association. Stroke. 2009; 40:2945–2948.
Article2. Zhao H. Ischemic postconditioning as a novel avenue to protect against brain injury after stroke. J Cereb Blood Flow Metab. 2009; 29:873–885.
Article3. Chalecka-Franaszek E, Chuang DM. Lithium activates the serine/threonine kinase Akt-1 and suppresses glutamate-induced inhibition of Akt-1 activity in neurons. Proc Natl Acad Sci U S A. 1999; 96:8745–8750.
Article4. Miyawaki T, Ofengeim D, Noh KM, Latuszek-Barrantes A, Hemmings BA, Follenzi A, et al. The endogenous inhibitor of Akt, CTMP, is critical to ischemia-induced neuronal death. Nat Neurosci. 2009; 12:618–626.
Article5. Temple S. Division and differentiation of isolated CNS blast cells in microculture. Nature. 1989; 340:471–473.
Article6. Doetsch F. A niche for adult neural stem cells. Curr Opin Genet Dev. 2003; 13:543–550.
Article7. Yagita Y, Kitagawa K, Ohtsuki T, Takasawa Ki, Miyata T, Okano H, et al. Neurogenesis by progenitor cells in the ischemic adult rat hippocampus. Stroke. 2001; 32:1890–1896.
Article8. Parent JM, Vexler ZS, Gong C, Derugin N, Ferriero DM. Rat forebrain neurogenesis and striatal neuron replacement after focal stroke. Ann Neurol. 2002; 52:802–813.
Article9. Thored P, Arvidsson A, Cacci E, Ahlenius H, Kallur T, Darsalia V, et al. Persistent production of neurons from adult brain stem cells during recovery after stroke. Stem Cells. 2006; 24:739–747.
Article10. Crowder RJ, Freeman RS. Phosphatidylinositol 3-kinase and Akt protein kinase are necessary and sufficient for the survival of nerve growth factor-dependent sympathetic neurons. J Neurosci. 1998; 18:2933–2943.
Article11. Li BS, Ma W, Zhang L, Barker JL, Stenger DA, Pant HC. Activation of phosphatidylinositol-3 kinase (PI-3K) and extracellular regulated kinases (Erk1/2) is involved in muscarinic receptor-mediated DNA synthesis in neural progenitor cells. J Neurosci. 2001; 21:1569–1579.
Article12. Leevers SJ, Vanhaesebroeck B, Waterfield MD. Signalling through phosphoinositide 3-kinases: the lipids take centre stage. Curr Opin Cell Biol. 1999; 11:219–225.
Article13. Vanhaesebroeck B, Guillermet-Guibert J, Graupera M, Bilanges B. The emerging mechanisms of isoform-specific PI3K signalling. Nat Rev Mol Cell Biol. 2010; 11:329–341.
Article14. Knight ZA, Gonzalez B, Feldman ME, Zunder ER, Goldenberg DD, Williams O, et al. A pharmacological map of the PI3-K family defines a role for p110alpha in insulin signaling. Cell. 2006; 125:733–747.
Article15. Auger KR, Serunian LA, Soltoff SP, Libby P, Cantley LC. PDGF-dependent tyrosine phosphorylation stimulates production of novel polyphosphoinositides in intact cells. Cell. 1989; 57:167–175.
Article16. Liu P, Cheng H, Roberts TM, Zhao JJ. Targeting the phosphoinositide 3-kinase pathway in cancer. Nat Rev Drug Discov. 2009; 8:627–644.
Article17. Vivanco I, Sawyers CL. The phosphatidylinositol 3-Kinase AKT pathway in human cancer. Nat Rev Cancer. 2002; 2:489–501.
Article18. Mahadevan D, Chiorean EG, Harris WB, Von Hoff DD, Stejskal-Barnett A, Qi W, et al. Phase I pharmacokinetic and pharmacodynamic study of the pan-PI3K/mTORC vascular targeted pro-drug SF1126 in patients with advanced solid tumours and B-cell malignancies. Eur J Cancer. 2012; 48:3319–3327.
Article19. Noh MY, Kim YS, Lee KY, Lee YJ, Kim SH, Yu HJ, et al. The early activation of PI3K strongly enhances the resistance of cortical neurons to hypoxic injury via the activation of downstream targets of the PI3K pathway and the normalization of the levels of PARP activity, ATP, and NAD+. Mol Neurobiol. 2013; 47:757–769.
Article20. Laurino L, Wang XX, de la Houssaye BA, Sosa L, Dupraz S, Cáceres A, et al. PI3K activation by IGF-1 is essential for the regulation of membrane expansion at the nerve growth cone. J Cell Sci. 2005; 118(Pt 16):3653–3662.
Article21. Cuesto G, Enriquez-Barreto L, Caramés C, Cantarero M, Gasull X, Sandi C, et al. Phosphoinositide-3-kinase activation controls synaptogenesis and spinogenesis in hippocampal neurons. J Neurosci. 2011; 31:2721–2733.
Article22. Hasegawa Y, Suzuki H, Altay O, Zhang JH. Preservation of tropomyosin-related kinase B (TrkB) signaling by sodium orthovanadate attenuates early brain injury after subarachnoid hemorrhage in rats. Stroke. 2011; 42:477–483.
Article23. Noshita N, Sugawara T, Lewén A, Hayashi T, Chan PH. Copper-zinc superoxide dismutase affects Akt activation after transient focal cerebral ischemia in mice. Stroke. 2003; 34:1513–1518.
Article24. Kilic E, Kilic U, Wang Y, Bassetti CL, Marti HH, Hermann DM. The phosphatidylinositol-3 kinase/Akt pathway mediates VEGF's neuroprotective activity and induces blood brain barrier permeability after focal cerebral ischemia. FASEB J. 2006; 20:1185–1187.
Article25. Bai Y, Meng Z, Cui M, Zhang X, Chen F, Xiao J, et al. An Ang1-Tie2-PI3K axis in neural progenitor cells initiates survival responses against oxygen and glucose deprivation. Neuroscience. 2009; 160:371–381.
Article26. Xu X, Chua CC, Gao J, Chua KW, Wang H, Hamdy RC, et al. Neuroprotective effect of humanin on cerebral ischemia/reperfusion injury is mediated by a PI3K/Akt pathway. Brain Res. 2008; 1227:12–18.
Article27. Clarke DL, Johansson CB, Wilbertz J, Veress B, Nilsson E, Karlström H, et al. Generalized potential of adult neural stem cells. Science. 2000; 288:1660–1663.
Article28. Peltier J, O'Neill A, Schaffer DV. PI3K/Akt and CREB regulate adult neural hippocampal progenitor proliferation and differentiation. Dev Neurobiol. 2007; 67:1348–1361.
Article29. Nguyen N, Lee SB, Lee YS, Lee KH, Ahn JY. Neuroprotection by NGF and BDNF against neurotoxin-exerted apoptotic death in neural stem cells are mediated through Trk receptors, activating PI3-kinase and MAPK pathways. Neurochem Res. 2009; 34:942–951.
Article30. Park SM, Jung JS, Jang MS, Kang KS, Kang SK. Transforming growth factor-beta1 regulates the fate of cultured spinal cord-derived neural progenitor cells. Cell Prolif. 2008; 41:248–264.
Article31. Wu Y, Peng H, Cui M, Whitney NP, Huang Y, Zheng JC. CXCL12 increases human neural progenitor cell proliferation through Akt-1/FOXO3a signaling pathway. J Neurochem. 2009; 109:1157–1167.
Article32. Sato A, Sunayama J, Matsuda K, Tachibana K, Sakurada K, Tomiyama A, et al. Regulation of neural stem/progenitor cell maintenance by PI3K and mTOR. Neurosci Lett. 2010; 470:115–120.
Article33. Kendall SE, Najbauer J, Johnston HF, Metz MZ, Li S, Bowers M. Neural stem cell targeting of glioma is dependent on phosphoinositide 3-kinase signaling. Stem Cells. 2008; 26:1575–1586.
Article34. Leong SY, Faux CH, Turbic A, Dixon KJ, Turnley AM. The Rho kinase pathway regulates mouse adult neural precursor cell migration. Stem Cells. 2011; 29:332–343.
Article35. Shioda N, Han F, Fukunaga K. Role of Akt and ERK signaling in the neurogenesis following brain ischemia. Int Rev Neurobiol. 2009; 85:375–387.36. Wang L, Zhang ZG, Zhang RL, Jiao ZX, Wang Y, Pourabdollah-Nejad DS, et al. Neurogenin 1 mediates erythropoietin enhanced differentiation of adult neural progenitor cells. J Cereb Blood Flow Metab. 2006; 26:556–564.
Article37. Zhang X, Zhang L, Cheng X, Guo Y, Sun X, Chen G, et al. IGF-1 promotes Brn-4 expression and neuronal differentiation of neural stem cells via the PI3K/Akt pathway. PLoS One. 2014; 9:e113801.
Article38. Jin L, Hu X, Feng L. NT3 inhibits FGF2-induced neural progenitor cell proliferation via the PI3K/GSK3 pathway. J Neurochem. 2005; 93:1251–1261.
Article39. Chan WS, Sideris A, Sutachan JJ, Montoya G JV, Blanck TJ, Recio-Pinto E. Differential regulation of proliferation and neuronal differentiation in adult rat spinal cord neural stem/progenitors by ERK1/2, Akt, and PLCγ. Front Mol Neurosci. 2013; 6:23.
Article40. Gambarotta G, Garzotto D, Destro E, Mautino B, Giampietro C, Cutrupi S, et al. ErbB4 expression in neural progenitor cells (ST14A) is necessary to mediate neuregulin-1beta1-induced migration. J Biol Chem. 2004; 279:48808–48816.
Article41. Li S, Deng L, Gong L, Bian H, Dai Y, Wang Y. Upregulation of CXCR4 favoring neural-like cells migration via AKT activation. Neurosci Res. 2010; 67:293–299.
Article42. Groszer M, Erickson R, Scripture-Adams DD, Lesche R, Trumpp A, Zack JA, et al. Negative regulation of neural stem/progenitor cell proliferation by the Pten tumor suppressor gene in vivo. Science. 2001; 294:2186–2189.
Article43. Garcia JH, Lossinsky AS, Kauffman FC, Conger KA. Neuronal ischemic injury: light microscopy, ultrastructure and biochemistry. Acta Neuropathol. 1978; 43:85–95.
Article44. Rothman S. Synaptic release of excitatory amino acid neurotransmitter mediates anoxic neuronal death. J Neurosci. 1984; 4:1884–1891.
Article45. Deshpande JK, Siesjö BK, Wieloch T. Calcium accumulation and neuronal damage in the rat hippocampus following cerebral ischemia. J Cereb Blood Flow Metab. 1987; 7:89–95.
Article46. Chen YH, Chiang YH, Ma HI. Analysis of spatial and temporal protein expression in the cerebral cortex after ischemia-reperfusion injury. J Clin Neurol. 2014; 10:84–93.
Article47. Endoh M, Pulsinelli WA, Wagner JA. Transient global ischemia induces dynamic changes in the expression of bFGF and the FGF receptor. Brain Res Mol Brain Res. 1994; 22:76–88.
Article48. Zhang RL, Chopp M, Roberts C, Liu X, Wei M, Nejad-Davarani SP, et al. Stroke increases neural stem cells and angiogenesis in the neurogenic niche of the adult mouse. PLoS One. 2014; 9:e113972.
Article49. Lindvall O, Ernfors P, Bengzon J, Kokaia Z, Smith ML, Siesjö BK, et al. Differential regulation of mRNAs for nerve growth factor, brain-derived neurotrophic factor, and neurotrophin 3 in the adult rat brain following cerebral ischemia and hypoglycemic coma. Proc Natl Acad Sci U S A. 1992; 89:648–652.
Article50. Craig CG, Tropepe V, Morshead CM, Reynolds BA, Weiss S, van der Kooy D. In vivo growth factor expansion of endogenous subependymal neural precursor cell populations in the adult mouse brain. J Neurosci. 1996; 16:2649–2658.
Article51. Kuhn HG, Winkler J, Kempermann G, Thal LJ, Gage FH. Epidermal growth factor and fibroblast growth factor-2 have different effects on neural progenitors in the adult rat brain. J Neurosci. 1997; 17:5820–5829.
Article52. Pencea V, Bingaman KD, Wiegand SJ, Luskin MB. Infusion of brain-derived neurotrophic factor into the lateral ventricle of the adult rat leads to new neurons in the parenchyma of the striatum, septum, thalamus, and hypothalamus. J Neurosci. 2001; 21:6706–6717.
Article53. Nakatomi H, Kuriu T, Okabe S, Yamamoto S, Hatano O, Kawahara N, et al. Regeneration of hippocampal pyramidal neurons after ischemic brain injury by recruitment of endogenous neural progenitors. Cell. 2002; 110:429–441.
Article54. Teramoto T, Qiu J, Plumier JC, Moskowitz MA. EGF amplifies the replacement of parvalbumin-expressing striatal interneurons after ischemia. J Clin Invest. 2003; 111:1125–1132.
Article55. Jin K, Mao XO, Del Rio, Jin L, Greenberg DA. Heparin-binding epidermal growth factor-like growth factor stimulates cell proliferation in cerebral cortical cultures through phosphatidylinositol 3'-kinase and mitogen-activated protein kinase. J Neurosci Res. 2005; 81:497–505.
Article56. Wang L, Zhang ZG, Zhang RL, Gregg SR, Hozeska-Solgot A, LeTourneau Y, et al. Matrix metalloproteinase 2 (MMP2) and MMP9 secreted by erythropoietin-activated endothelial cells promote neural progenitor cell migration. J Neurosci. 2006; 26:5996–6003.
Article57. Lee SR, Kim HY, Rogowska J, Zhao BQ, Bhide P, Parent JM, et al. Involvement of matrix metalloproteinase in neuroblast cell migration from the subventricular zone after stroke. J Neurosci. 2006; 26:3491–3495.
Article58. Lee JS, Hong JM, Moon GJ, Lee PH, Ahn YH, Bang OY. STARTING collaborators. A long-term follow-up study of intravenous autologous mesenchymal stem cell transplantation in patients with ischemic stroke. Stem Cells. 2010; 28:1099–1106.
Article59. Wahane SD, Hellbach N, Prentzell MT, Weise SC, Vezzali R, Kreutz C, et al. PI3K-p110-alpha-subtype signalling mediates survival, proliferation and neurogenesis of cortical progenitor cells via activation of mTORC2. J Neurochem. 2014; 130:255–267.
Article60. Zhang BY, Saijilafu , Liu CM, Wang RY, Zhu Q, Jiao Z, et al. Akt-independent GSK3 inactivation downstream of PI3K signaling regulates mammalian axon regeneration. Biochem Biophys Res Commun. 2014; 443:743–748.
Article61. Saijilafu , Hur EM, Liu CM, Jiao Z, Xu WL, Zhou FQ. PI3K-GSK3 signalling regulates mammalian axon regeneration by inducing the expression of Smad1. Nat Commun. 2013; 4:2690.
Article62. Choi H, Park HH, Lee KY, Choi NY, Yu HJ, Lee YJ, et al. Coenzyme Q10 restores amyloid beta-inhibited proliferation of neural stem cells by activating the PI3K pathway. Stem Cells Dev. 2013; 22:2112–2120.
Article63. Song J, Kang SM, Lee KM, Lee JE. The protective effect of melatonin on neural stem cell against LPS-induced inflammation. Biomed Res Int. 2015; 2015:854359.
Article64. Sütterlin P, Williams EJ, Chambers D, Saraf K, von Schack D, Reisenberg M, et al. The molecular basis of the cooperation between EGF, FGF and eCB receptors in the regulation of neural stem cell function. Mol Cell Neurosci. 2013; 52:20–30.
Article65. Dolcet X, Egea J, Soler RM, Martin-Zanca D, Comella JX. Activation of phosphatidylinositol 3-kinase, but not extracellular-regulated kinases, is necessary to mediate brain-derived neurotrophic factor-induced motoneuron survival. J Neurochem. 1999; 73:521–531.
Article66. Tsai YW, Yang YR, Sun SH, Liang KC, Wang RY. Post ischemia intermittent hypoxia induces hippocampal neurogenesis and synaptic alterations and alleviates long-term memory impairment. J Cereb Blood Flow Metab. 2013; 33:764–773.
Article67. Wang H, Wang G, Wang C, Wei Y, Wen Z, Wang C, et al. The early stage formation of PI3K-AMPAR GluR2 subunit complex facilitates the long term neuroprotection induced by propofol post-conditioning in rats. PLoS One. 2013; 8:e65187.
Article68. Zhu Y, Jin K, Mao XO, Greenberg DA. Vascular endothelial growth factor promotes proliferation of cortical neuron precursors by regulating E2F expression. FASEB J. 2003; 17:186–193.
Article69. Tu XK, Yang WZ, Chen JP, Chen Y, Chen Q, Chen PP, et al. Repetitive ischemic preconditioning attenuates inflammatory reaction and brain damage after focal cerebral ischemia in rats: involvement of PI3K/Akt and ERK1/2 signaling pathway. J Mol Neurosci. 2015; 55:912–922.
Article70. Qi D, Ouyang C, Wang Y, Zhang S, Ma X, Song Y, et al. HO-1 attenuates hippocampal neurons injury via the activation of BDNF-TrkB-PI3K/Akt signaling pathway in stroke. Brain Res. 2014; 1577:69–76.
Article71. Liang K, Ye Y, Wang Y, Zhang J, Li C. Formononetin mediates neuroprotection against cerebral ischemia/reperfusion in rats via downregulation of the Bax/Bcl-2 ratio and upregulation PI3K/Akt signaling pathway. J Neurol Sci. 2014; 344:100–104.
Article