Ann Surg Treat Res.
2015 Jan;88(1):1-7. 10.4174/astr.2015.88.1.1.
The differentiation of human multipotent adult progenitor cells into hepatocyte-like cells induced by coculture with human hepatocyte line L02
- Affiliations
-
- 1Department of Hepatobiliary Surgery, Fuzhou General Hospital, Fuzhou, China. xjsnmn@163.com
- 2Department of Nephrology, Xijing Hospital, Fourth Military Medical University, Xi'an, China.
- 3Institute of Materia Medica, School of Pharmacy, The Fourth Military Medical University, Xi'an, China.
- 4Department of Nephrology, Tangdu Hospital, The Fourth Military Medical University, Xi'an, China.
- KMID: 2167052
- DOI: http://doi.org/10.4174/astr.2015.88.1.1
Abstract
- PURPOSE
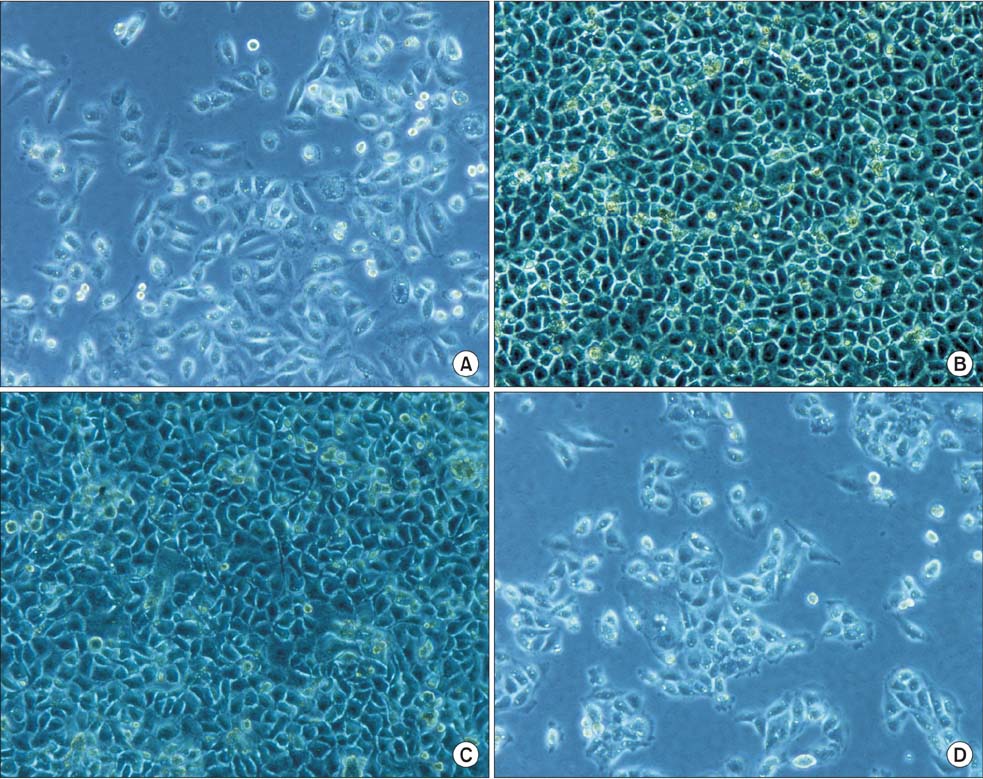
The aim of this study was to establish an in vitro method to purify human multipotent adult progenitor cells (hMAPCs) and assess their possible differentiation into hepatocytes by coculture with human hepatocyte line L02.
METHODS
hMAPCs were isolated by magnetic activated cell sorting (MACS) depletion selection using CD45 and GlyA microbeads. After indirect or direct coculture of hMAPCs and human hepatocyte line L02, the expression of albumin (ALB), alpha-fetoprotein (AFP), cytokeratin (CK) 18, and CK19 by hMAPCs was detected by immunocytochemistry.
RESULTS
With the MACS method, (5-10) x 10(4)/mL hMAPCs could be separated from 1 x 10(6)/mL bone marrow mononuclear cells. The purity of CD45-/GlyA- cells separated from bone marrow adherent cells was more than 98%, as determined by flow cytometry. In the coculture without cell-to-cell contact, hMAPCs expressed high AFP on day 1, and then tapered daily to low expression on day 7; ALB expression reached its peak on day 5, and remained high on day 7; CK18 was initially expressed on day 5 and was higher on day 7; CK19 was negative in all assays. In the coculture with cell-to-cell contact, ALB and CK18 were expressed by most cells while AFP appeared in only a few on day 5.
CONCLUSION
hMAPCs were induced to differentiate into mature hepatocyte-like cells by coculture with a hepatocyte cell line, either with or without cell-to-cell contact, but the former seemed more effective.
Keyword
MeSH Terms
Figure
Reference
-
1. Li M, Ikehara S. Bone-marrow-derived mesenchymal stem cells for organ repair. Stem Cells Int. 2013; 2013:132642.2. Prasajak P, Leeanansaksiri W. Developing a new two-step protocol to generate functional hepatocytes from Whartons Jelly-derived mesenchymal stem cells under hypoxic condition. Stem Cells Int. 2013; 2013:762196.3. Ayatollahi M, Soleimani M, Tabei SZ, Kabir Salmani M. Hepatogenic differentiation of mesenchymal stem cells induced by insulin like growth factor-I. World J Stem Cells. 2011; 3:113–121.4. Chen XW, Zhu DJ, Ju YL, Zhou SF. Therapeutic effect of transplanting magnetically labeled bone marrow stromal stem cells in a liver injury rat model with 70%-hepatectomy. Med Sci Monit. 2012; 18:BR375–BR382.5. Jiang Y, Henderson D, Blackstad M, Chen A, Miller RF, Verfaillie CM. Neuroectodermal differentiation from mouse multipotent adult progenitor cells. Proc Natl Acad Sci U S A. 2003; 100:Suppl 1. 11854–11860.6. Mizuguchi T, Hui T, Palm K, Sugiyama N, Mitaka T, Demetriou AA, et al. Enhanced proliferation and differentiation of rat hepatocytes cultured with bone marrow stromal cells. J Cell Physiol. 2001; 189:106–119.7. Reyes M, Lund T, Lenvik T, Aguiar D, Koodie L, Verfaillie CM. Purification and ex vivo expansion of postnatal human marrow mesodermal progenitor cells. Blood. 2001; 98:2615–2625.8. Zhang L, Ye JS, Decot V, Stoltz JF, de Isla N. Research on stem cells as candidates to be differentiated into hepatocytes. Biomed Mater Eng. 2012; 22:105–111.9. Petersen BE, Bowen WC, Patrene KD, Mars WM, Sullivan AK, Murase N, et al. Bone marrow as a potential source of hepatic oval cells. Science. 1999; 284:1168–1170.10. Monga SP, Tang Y, Candotti F, Rashid A, Wildner O, Mishra B, et al. Expansion of hepatic and hematopoietic stem cells utilizing mouse embryonic liver explants. Cell Transplant. 2001; 10:81–89.11. Ouyang J, Shao J, Zou H, Lou Y, Yu Y. Hepatic differentiation of rat mesenchymal stem cells by a small molecule. ChemMedChem. 2012; 7:1447–1452.12. Tan Y, Xiao EH, Xiao LZ, Yuan YH, Ma C, Shang QL, et al. VEGF(165) expressing bone marrow mesenchymal stem cells differentiate into hepatocytes under HGF and EGF induction in vitro. Cytotechnology. 2012; 64:635–647.13. Magner NL, Jung Y, Wu J, Nolta JA, Zern MA, Zhou P. Insulin and IGFs enhance hepatocyte differentiation from human embryonic stem cells via the PI3K/AKT pathway. Stem Cells. 2013; 31:2095–2103.14. Jang YY, Collector MI, Baylin SB, Diehl AM, Sharkis SJ. Hematopoietic stem cells convert into liver cells within days without fusion. Nat Cell Biol. 2004; 6:532–539.15. Yamazaki S, Miki K, Hasegawa K, Sata M, Takayama T, Makuuchi M. Sera from liver failure patients and a demethylating agent stimulate transdifferentiation of murine bone marrow cells into hepatocytes in coculture with nonparenchymal liver cells. J Hepatol. 2003; 39:17–23.16. Lange C, Bassler P, Lioznov MV, Bruns H, Kluth D, Zander AR, et al. Hepatocytic gene expression in cultured rat mesenchymal stem cells. Transplant Proc. 2005; 37:276–279.17. Moore RN, Dasgupta A, Rajaei N, Yarmush ML, Toner M, Larue L, et al. Enhanced differentiation of embryonic stem cells using co-cultivation with hepatocytes. Biotechnol Bioeng. 2008; 101:1332–1343.18. Choi HJ, Choi D. Successful mouse hepatocyte culture with sandwich collagen gel formation. J Korean Surg Soc. 2013; 84:202–208.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Augmentation of Butyrate-induced Differentiation of Human Hepatocyte for the Development of an Efficient Bioartificial Liver
- Concise Review: Differentiation of Human Adult Stem Cells Into Hepatocyte-like Cells In vitro
- Clinical application of stem cells in liver diseases
- Functional Comparison of Human Embryonic Stem Cells and Induced Pluripotent Stem Cells as Sources of Hepatocyte-Like Cells
- Ectopic Over-expression of Oncogene Pim-2 Induce Malignant Transformation of Nontumorous Human Liver Cell Line L02