Allergy Asthma Immunol Res.
2016 Jul;8(4):338-345. 10.4168/aair.2016.8.4.338.
Age-Related Changes in Immunological Factors and Their Relevance in Allergic Disease Development During Childhood
- Affiliations
-
- 1Division of Allergy and Chronic Respiratory Disease, Center for Biomedical Sciences, Korea National Institute of Health, Korea Center for Disease Control and Prevention, Cheongju, Korea. jooshil@korea.kr
- 2Department of Internal Medicine and Institute of Allergy, Yonsei University College of Medicine, Seoul, Korea.
- 3Department of Pediatrics, University of Ulsan College of Medicine, Seoul, Korea.
- 4Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea.
- KMID: 2165917
- DOI: http://doi.org/10.4168/aair.2016.8.4.338
Abstract
- PURPOSE
Allergic diseases are triggered by Th2-mediated immune reactions to allergens and orchestrated by various immunological factors, including immune cells and cytokines. Although many reports have suggested that childhood is the critical period in the onset of allergic diseases and aging leads to alter the susceptibility of an individual to allergic diseases, age-related changes in various immunological factors in healthy individuals as well as their difference between healthy and allergic children have not yet been established.
METHODS
We investigated the ratio of Th1/Th2 cells and the levels of 22 allergy-related cytokines across all age groups in individuals who were classified as clinically non-atopic and healthy. We also examined their differences between healthy and allergic children to evaluate immunological changes induced by the development of allergic diseases during childhood.
RESULTS
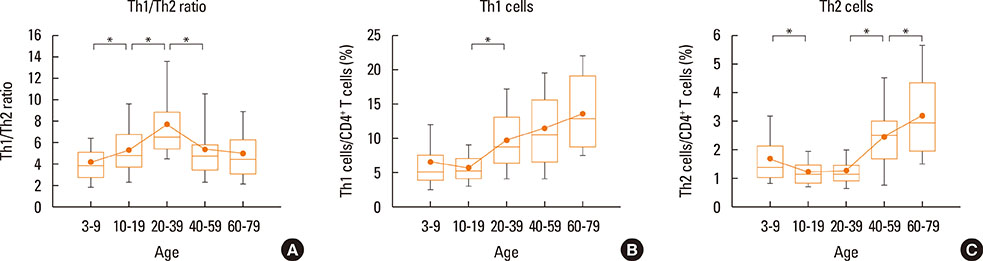
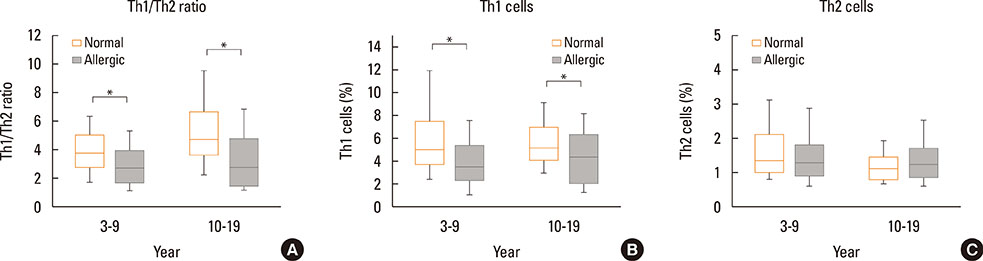
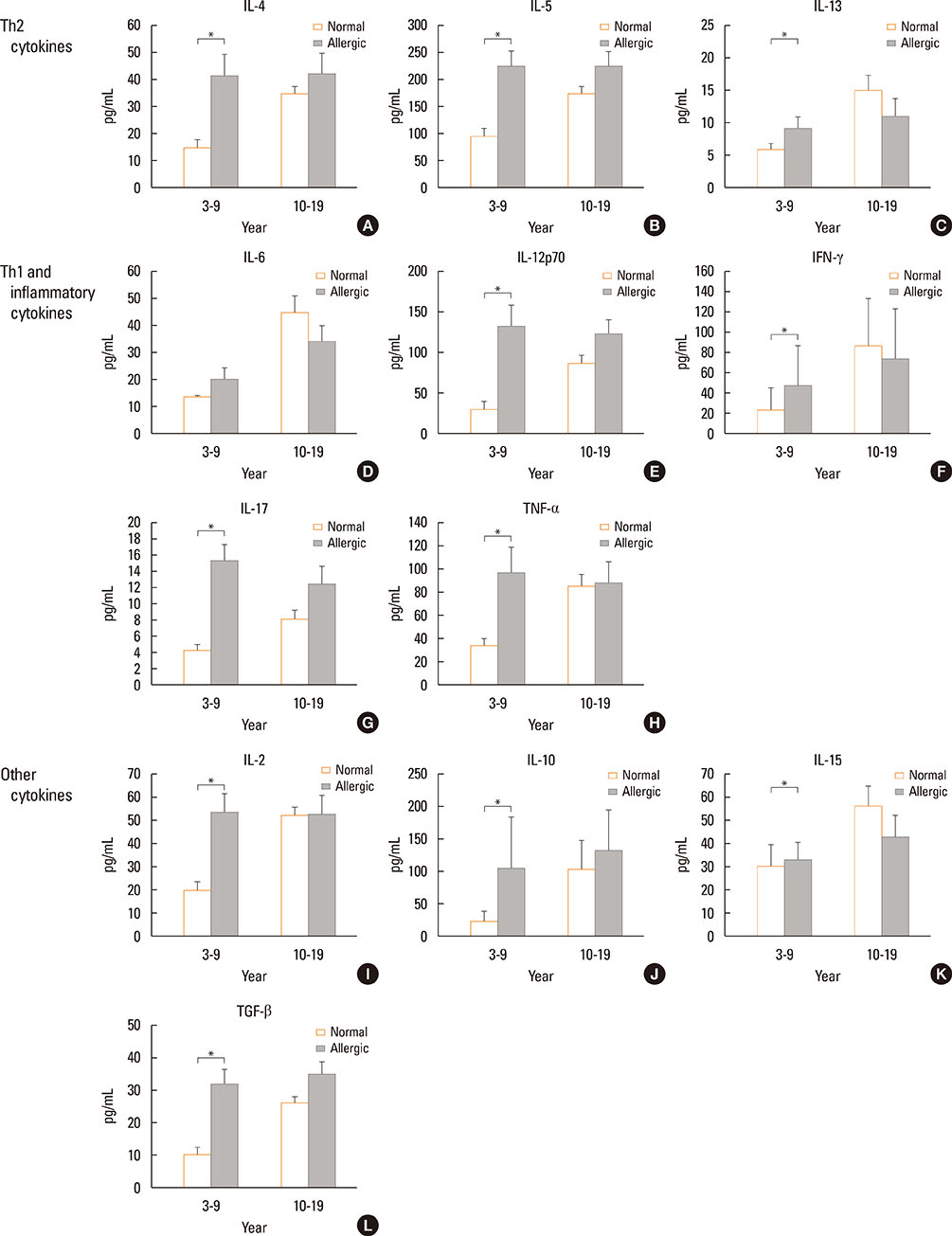
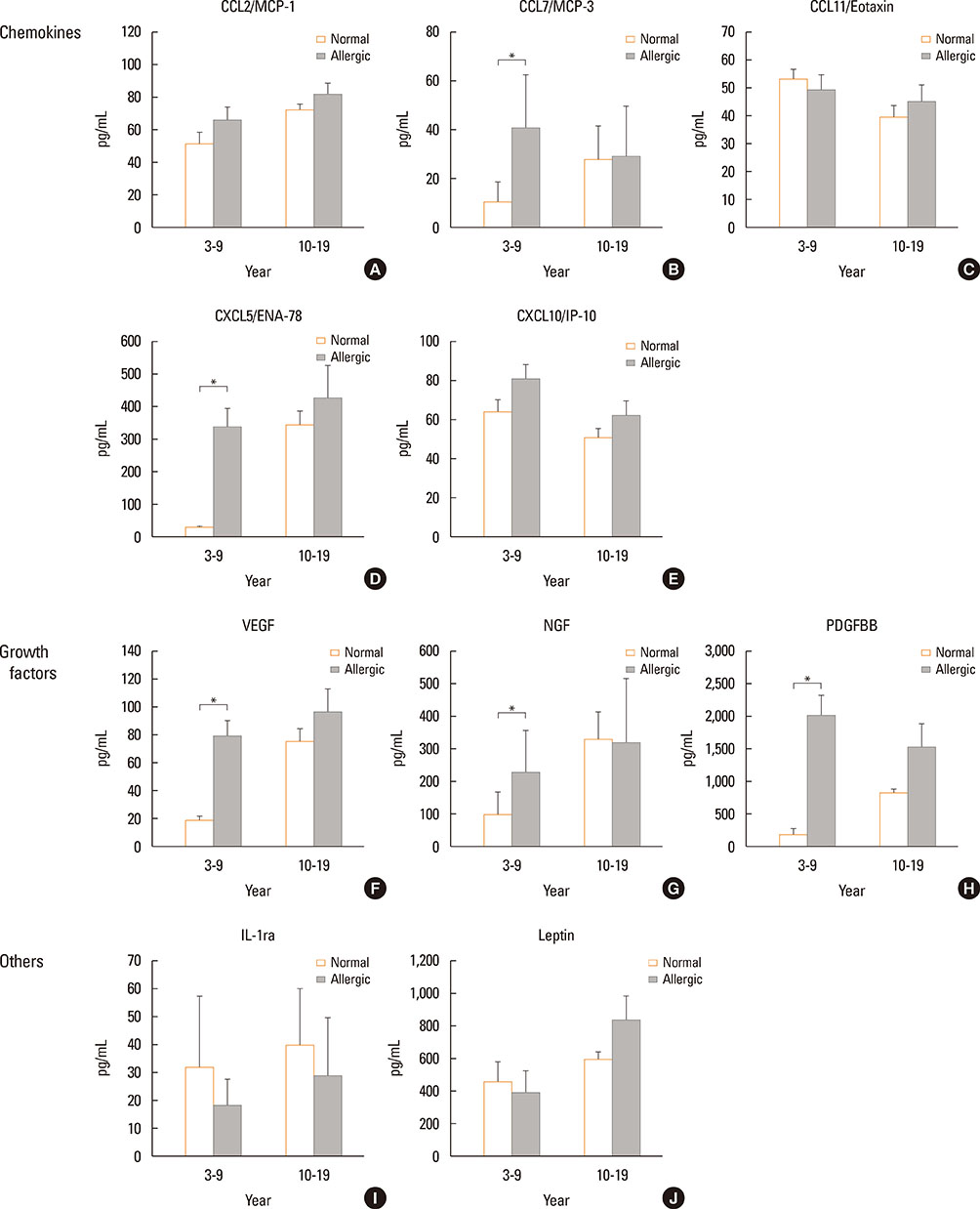
The Th1/Th2 ratio rose gradually during the growth period including childhood, reaching peak values in the twenties-thirties age group. Th1/Th2 ratios were significantly lower in allergic children than in healthy controls, whereas 14 of 22 cytokines were significantly higher in allergic children than in healthy controls. On the other hand, there were no differences in Th1/Th2 ratios and cytokines between healthy and allergic adolescents.
CONCLUSIONS
In this study, age-related changes in Th1/Th2 ratios were found in normal controls across all age groups, and decreases in Th1/Th2 ratio were observed with increasing of 14 cytokines in allergic children. The results of this study may be helpful as reference values for both monitoring immunological changes according to aging in healthy individuals and distinguishing between normal and allergic subjects in terms of immune cells and soluble factors.
Keyword
MeSH Terms
Figure
Reference
-
1. Eder W, Ege MJ, von Mutius E. The asthma epidemic. N Engl J Med. 2006; 355:2226–2235.2. Bousquet J, Khaltaev N, Cruz AA, Denburg J, Fokkens WJ, Togias A, et al. Allergic Rhinitis and its Impact on Asthma (ARIA) 2008 update (in collaboration with the World Health Organization, GA(2)LEN and AllerGen). Allergy. 2008; 63:Suppl 86. 8–160.3. Zhang Y, Zhang L. Prevalence of allergic rhinitis in china. Allergy Asthma Immunol Res. 2014; 6:105–113.4. Galli SJ, Tsai M, Piliponsky AM. The development of allergic inflammation. Nature. 2008; 454:445–454.5. Holgate ST. A brief history of asthma and its mechanisms to modern concepts of disease pathogenesis. Allergy Asthma Immunol Res. 2010; 2:165–171.6. Chung KF, Barnes PJ. Cytokines in asthma. Thorax. 1999; 54:825–857.7. Barnes PJ. The cytokine network in asthma and chronic obstructive pulmonary disease. J Clin Invest. 2008; 118:3546–3556.8. Locksley RM. Asthma and allergic inflammation. Cell. 2010; 140:777–783.9. Spergel JM, Paller AS. Atopic dermatitis and the atopic march. J Allergy Clin Immunol. 2003; 112:6 Suppl. S118–S127.10. Bacharier LB, Guilbert TW. Diagnosis and management of early asthma in preschool-aged children. J Allergy Clin Immunol. 2012; 130:287–296.11. Weng NP. Aging of the immune system: how much can the adaptive immune system adapt? Immunity. 2006; 24:495–499.12. Czesnikiewicz-Guzik M, Lee WW, Cui D, Hiruma Y, Lamar DL, Yang ZZ, et al. T cell subset-specific susceptibility to aging. Clin Immunol. 2008; 127:107–118.13. Zheng T, Yu J, Oh MH, Zhu Z. The atopic march: progression from atopic dermatitis to allergic rhinitis and asthma. Allergy Asthma Immunol Res. 2011; 3:67–73.14. Kleiner G, Marcuzzi A, Zanin V, Monasta L, Zauli G. Cytokine levels in the serum of healthy subjects. Mediators Inflamm. 2013; 2013:434010.15. Duramad P, Tager IB, Holland NT. Cytokines and other immunological biomarkers in children's environmental health studies. Toxicol Lett. 2007; 172:48–59.16. Stevens DA, Moss RB, Kurup VP, Knutsen AP, Greenberger P, Judson MA, et al. Allergic bronchopulmonary aspergillosis in cystic fibrosis--state of the art: Cystic Fibrosis Foundation Consensus Conference. Clin Infect Dis. 2003; 37:Suppl 3. S225–S264.17. Lee YC, Lee KH, Lee HB, Rhee YK. Serum levels of interleukins (IL)-4, IL-5, IL-13, and interferon-gamma in acute asthma. J Asthma. 2001; 38:665–671.18. Huang CS, Chen SJ, Chung RL, Tang RB. Serum interleukin-5 measurements for monitoring acute asthma in children. J Asthma. 2005; 42:297–300.19. Ciprandi G, De Amici M, Murdaca G, Fenoglio D, Ricciardolo F, Marseglia G, et al. Serum interleukin-17 levels are related to clinical severity in allergic rhinitis. Allergy. 2009; 64:1375–1378.20. Wong CK, Ho CY, Ko FW, Chan CH, Ho AS, Hui DS, et al. Proinflammatory cytokines (IL-17, IL-6, IL-18 and IL-12) and Th cytokines (IFN-gamma, IL-4, IL-10 and IL-13) in patients with allergic asthma. Clin Exp Immunol. 2001; 125:177–183.21. Jerzyńska J, Stelmach W, Majak P, Brzozowska A, Sobocińska A, Stelmach I. Effect of specific immunotherapy on serum levels of tumor necrosis factor alpha in asthmatic children. Allergy Asthma Proc. 2008; 29:274–279.22. Lewis CC, Chu HW, Westcott JY, Tucker A, Langmack EL, Sutherland ER, et al. Airway fibroblasts exhibit a synthetic phenotype in severe asthma. J Allergy Clin Immunol. 2005; 115:534–540.23. Matías V, San Feliciano L, Fernández JE, Lapeña S, Garrido E, Ardura J, et al. Host and environmental factors influencing respiratory secretion of pro-wheezing biomarkers in preterm children. Pediatr Allergy Immunol. 2012; 23:441–447.24. Wahn U. What drives the allergic march? Allergy. 2000; 55:591–599.25. Stern DA, Morgan WJ, Halonen M, Wright AL, Martinez FD. Wheezing and bronchial hyper-responsiveness in early childhood as predictors of newly diagnosed asthma in early adulthood: a longitudinal birth-cohort study. Lancet. 2008; 372:1058–1064.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Particulate matter and childhood allergic diseases
- Impact of perinatal environmental tobacco smoke on the development of childhood allergic diseases
- Effects of wheezing in early childhood in the development of allergic rhinitis in later years
- Genetic studies of allergic disease and their clinical implications
- Asthma in childhood: a complex, heterogeneous disease