Korean J Physiol Pharmacol.
2016 Jan;20(1):15-23. 10.4196/kjpp.2016.20.1.15.
Carnosic acid protects against acetaminophen-induced hepatotoxicity by potentiating Nrf2-mediated antioxidant capacity in mice
- Affiliations
-
- 1Department of Ultrasound, The Second Hospital Affiliated of Zhengzhou University, Zhengzhou, Henan 450014, P. R. China.
- 2Department of Gastroenterology, The Second Hospital Affiliated of Zhengzhou University, Zhengzhou, Henan 450014, P. R. China. zhengpeng-yuandr@126.com
- KMID: 2150469
- DOI: http://doi.org/10.4196/kjpp.2016.20.1.15
Abstract
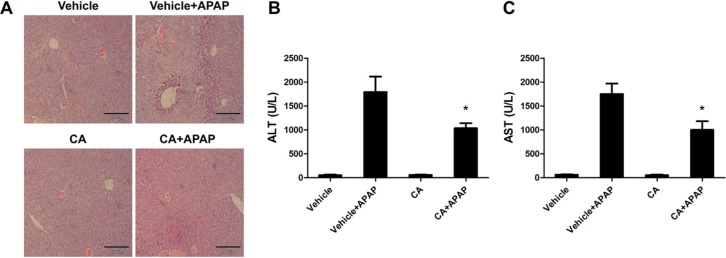
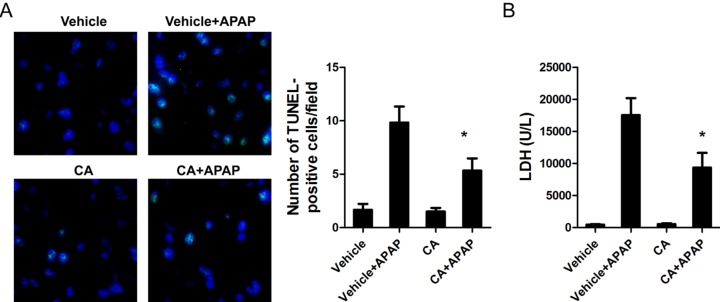
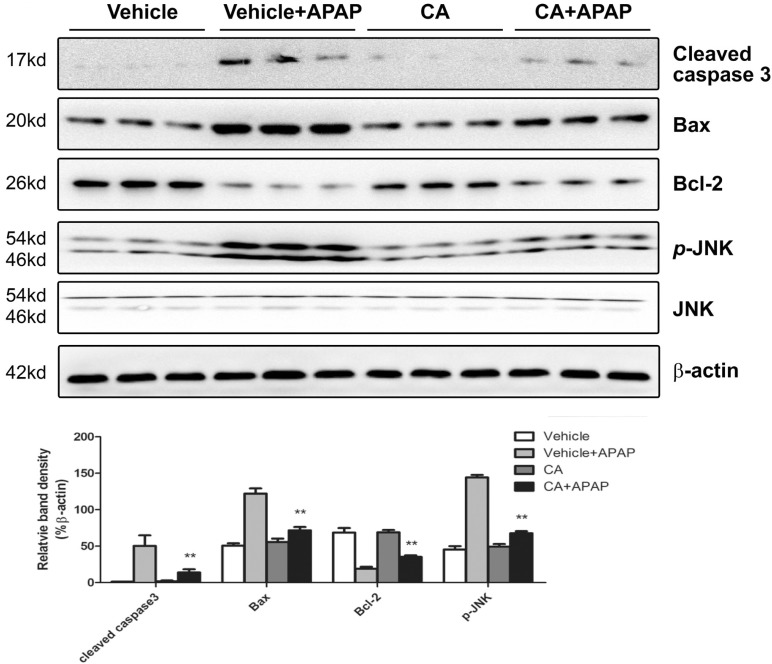
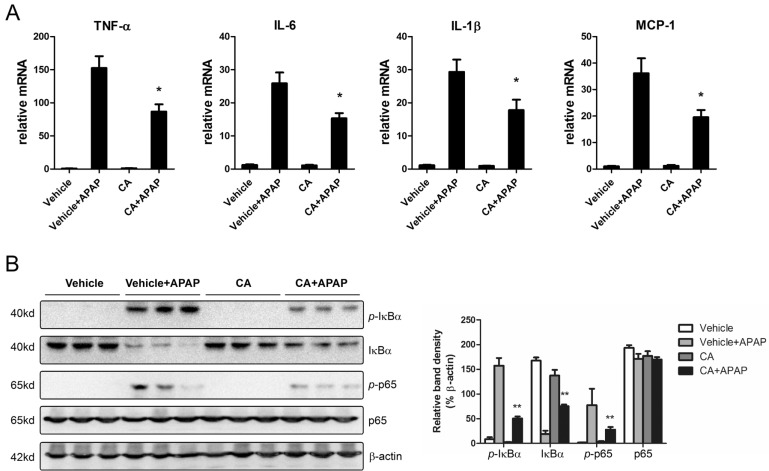
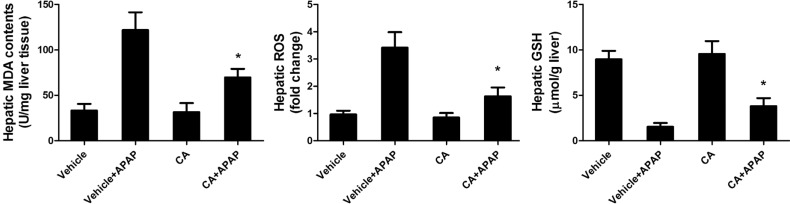
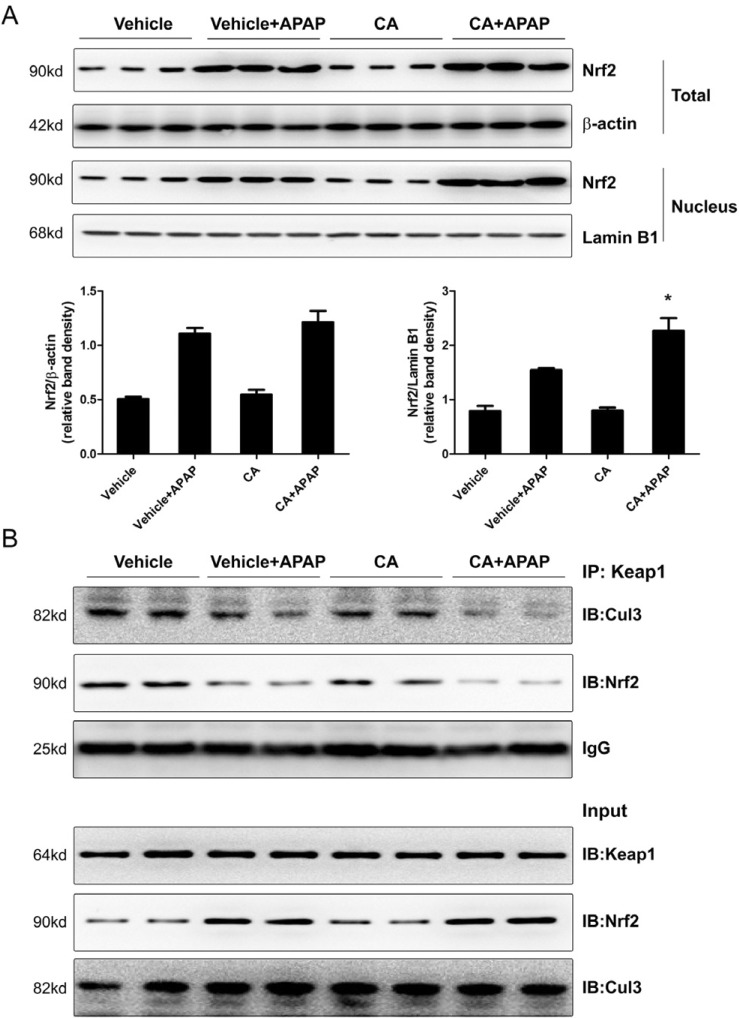
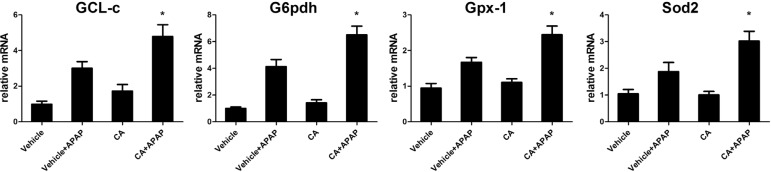
- Acetaminophen (APAP) overdose is one of the most common causes of acute liver failure. The study aimed to investigate the protective effect of carnosic acid (CA) on APAP-induced acute hepatotoxicity and its underlying mechanism in mice. To induce hepatotoxicity, APAP solution (400 mg/kg) was administered into mice by intraperitoneal injection. Histological analysis revealed that CA treatment significantly ameliorated APAP-induced hepatic necrosis. The levels of both alanine aminotransferase (ALT) and aspartate transaminase (AST) in serum were reduced by CA treatment. Moreover, CA treatment significantly inhibited APAP-induced hepatocytes necrosis and lactate dehydrogenase (LDH) releasing. Western blot analysis showed that CA abrogated APAP-induced cleaved caspase-3, Bax and phosphorylated JNK protein expression. Further results showed that CA treatment markedly inhibited APAP-induced pro-inflammatory cytokines TNF-alpha, IL-1beta, IL-6 and MCP-1 mRNA expression and the levels of phosphorylated IkappaBalpha and p65 protein in the liver. In addition, CA treatment reduced APAP- induced hepatic malondialdehyde (MDA) contents and reactive oxygen species (ROS) accumulation. Conversely, hepatic glutathione (GSH) level was increased by administration of CA in APAP-treated mice. Mechanistically, CA facilitated Nrf2 translocation into nuclear through blocking the interaction between Nrf2 and Keap1, which, in turn, upregulated anti-oxidant genes mRNA expression. Taken together, our results indicate that CA facilitates Nrf2 nuclear translocation, causing induction of Nrf2-dependent genes, which contributes to protection from acetaminophen hepatotoxicity.
Keyword
MeSH Terms
-
Acetaminophen
Alanine Transaminase
Animals
Aspartate Aminotransferases
Blotting, Western
Caspase 3
Cytokines
Glutathione
Hepatocytes
Injections, Intraperitoneal
Interleukin-6
L-Lactate Dehydrogenase
Liver
Liver Failure, Acute
Malondialdehyde
Mice*
Necrosis
Reactive Oxygen Species
RNA, Messenger
Tumor Necrosis Factor-alpha
Acetaminophen
Alanine Transaminase
Aspartate Aminotransferases
Caspase 3
Cytokines
Glutathione
Interleukin-6
L-Lactate Dehydrogenase
Malondialdehyde
RNA, Messenger
Reactive Oxygen Species
Tumor Necrosis Factor-alpha
Figure
Cited by 1 articles
-
Loss of RAR-α and RXR-α and enhanced caspase-3-dependent apoptosis in N-acetyl-p-aminophenol-induced liver injury in mice is tissue factor dependent
Mohamed Sadek Abdel-Bakky, Gouda Kamel Helal, El-Sayed Mohamed El-Sayed, Elham Amin, Abdulmajeed Alqasoumi, Ahmad Alhowail, Eman Sayed Said Abdelmoti, Ahmed Saad Saad
Korean J Physiol Pharmacol. 2021;25(5):385-393. doi: 10.4196/kjpp.2021.25.5.385.
Reference
-
1. Bernal W, Auzinger G, Dhawan A, Wendon J. Acute liver failure. Lancet. 2010; 376:190–201. PMID: 20638564.
Article2. Blieden M, Paramore LC, Shah D, Ben-Joseph R. A perspective on the epidemiology of acetaminophen exposure and toxicity in the United States. Expert Rev Clin Pharmacol. 2014; 7:341–348. PMID: 24678654.
Article3. Mitka M. FDA asks physicians to stop prescribing high-dose acetaminophen products. JAMA. 2014; 311:563. PMID: 24519283.
Article4. Holubek WJ, Kalman S, Hoffman RS. Acetaminophen-induced acute liver failure: results of a United States multicenter, prospective study. Hepatology. 2006; 43:880. author reply 882. PMID: 16557558.
Article5. Jaeschke H, McGill MR, Williams CD, Ramachandran A. Current issues with acetaminophen hepatotoxicity--a clinically relevant model to test the efficacy of natural products. Life Sci. 2011; 88:737–745. PMID: 21296090.
Article6. McGill MR, Sharpe MR, Williams CD, Taha M, Curry SC, Jaeschke H. The mechanism underlying acetaminophen-induced hepatotoxicity in humans and mice involves mitochondrial damage and nuclear DNA fragmentation. J Clin Invest. 2012; 122:1574–1583. PMID: 22378043.
Article7. Kensler TW, Wakabayashi N, Biswal S. Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annu Rev Pharmacol Toxicol. 2007; 47:89–116. PMID: 16968214.
Article8. Cullinan SB, Gordan JD, Jin J, Harper JW, Diehl JA. The Keap1-BTB protein is an adaptor that bridges Nrf2 to a Cul3- based E3 ligase: oxidative stress sensing by a Cul3-Keap1 ligase. Mol Cell Biol. 2004; 24:8477–8486. PMID: 15367669.9. Motohashi H, Yamamoto M. Nrf2-Keap1 defines a physiologically important stress response mechanism. Trends Mol Med. 2004; 10:549–557. PMID: 15519281.
Article10. Enomoto A, Itoh K, Nagayoshi E, Haruta J, Kimura T, O'Connor T, Harada T, Yamamoto M. High sensitivity of Nrf2 knockout mice to acetaminophen hepatotoxicity associated with decreased expression of ARE-regulated drug metabolizing enzymes and antioxidant genes. Toxicol Sci. 2001; 59:169–177. PMID: 11134556.
Article11. Chan K, Han XD, Kan YW. An important function of Nrf2 in combating oxidative stress: detoxification of acetaminophen. Proc Natl Acad Sci U S A. 2001; 98:4611–4616. PMID: 11287661.
Article12. Hadad N, Levy R. The synergistic anti-inflammatory effects of lycopene, lutein, β-carotene, and carnosic acid combinations via redox-based inhibition of NF-κB signaling. Free Radic Biol Med. 2012; 53:1381–1391. PMID: 22889596.
Article13. Sahu BD, Rentam KK, Putcha UK, Kuncha M, Vegi GM, Sistla R. Carnosic acid attenuates renal injury in an experimental model of rat cisplatin-induced nephrotoxicity. Food Chem Toxicol. 2011; 49:3090–3097. PMID: 21930180.
Article14. Hou CW, Lin YT, Chen YL, Wang YH, Chou JL, Ping LY, Jeng KC. Neuroprotective effects of carnosic acid on neuronal cells under ischemic and hypoxic stress. Nutr Neurosci. 2012; 15:257–263. PMID: 22687582.
Article15. Miller DM, Singh IN, Wang JA, Hall ED. Administration of the Nrf2-ARE activators sulforaphane and carnosic acid attenuates 4-hydroxy-2-nonenal-induced mitochondrial dysfunction ex vivo. Free Radic Biol Med. 2013; 57:1–9. PMID: 23275005.
Article16. Wang T, Takikawa Y, Tabuchi T, Satoh T, Kosaka K, Suzuki K. Carnosic acid (CA) prevents lipid accumulation in hepatocytes through the EGFR/MAPK pathway. J Gastroenterol. 2012; 47:805–813. PMID: 22350699.
Article17. Xiang Q, Liu Z, Wang Y, Xiao H, Wu W, Xiao C, Liu X. Carnosic acid attenuates lipopolysaccharide-induced liver injury in rats via fortifying cellular antioxidant defense system. Food Chem Toxicol. 2013; 53:1–9. PMID: 23200889.
Article18. Satoh T, Kosaka K, Itoh K, Kobayashi A, Yamamoto M, Shimojo Y, Kitajima C, Cui J, Kamins J, Okamoto S, Izumi M, Shirasawa T, Lipton SA. Carnosic acid, a catechol-type electrophilic compound, protects neurons both in vitro and in vivo through activation of the Keap1/Nrf2 pathway via S-alkylation of targeted cysteines on Keap1. J Neurochem. 2008; 104:1116–1131. PMID: 17995931.19. Sahu BD, Putcha UK, Kuncha M, Rachamalla SS, Sistla R. Carnosic acid promotes myocardial antioxidant response and prevents isoproterenol-induced myocardial oxidative stress and apoptosis in mice. Mol Cell Biochem. 2014; 394:163–176. PMID: 24903830.
Article20. Kim EJ, Lee MY, Jeon YJ. Silymarin Inhibits Morphological Changes in LPS-Stimulated Macrophages by Blocking NF-κB Pathway. Korean J Physiol Pharmacol. 2015; 19:211–218. PMID: 25954125.
Article21. Jaeschke H, Knight TR, Bajt ML. The role of oxidant stress and reactive nitrogen species in acetaminophen hepatotoxicity. Toxicol Lett. 2003; 144:279–288. PMID: 12927346.
Article22. Reid AB, Kurten RC, McCullough SS, Brock RW, Hinson JA. Mechanisms of acetaminophen-induced hepatotoxicity: role of oxidative stress and mitochondrial permeability transition in freshly isolated mouse hepatocytes. J Pharmacol Exp Ther. 2005; 312:509–516. PMID: 15466245.
Article23. Jaeschke H, McGill MR, Ramachandran A. Oxidant stress, mitochondria, and cell death mechanisms in drug-induced liver injury: lessons learned from acetaminophen hepatotoxicity. Drug Metab Rev. 2012; 44:88–106. PMID: 22229890.
Article24. Wang H, Liu F, Yang L, Zu Y, Wang H, Qu S, Zhang Y. Oxidative stability of fish oil supplemented with carnosic acid compared with synthetic antioxidants during long-term storage. Food Chem. 2011; 128:93–99. PMID: 25214334.
Article25. Tamaki Y, Tabuchi T, Takahashi T, Kosaka K, Satoh T. Activated glutathione metabolism participates in protective effects of carnosic acid against oxidative stress in neuronal HT22 cells. Planta Med. 2010; 76:683–688. PMID: 19941258.
Article26. Ni HM, Bockus A, Boggess N, Jaeschke H, Ding WX. Activation of autophagy protects against acetaminophen-induced hepatotoxicity. Hepatology. 2012; 55:222–232. PMID: 21932416.
Article27. Ma Q, He X. Molecular basis of electrophilic and oxidative defense: promises and perils of Nrf2. Pharmacol Rev. 2012; 64:1055–1081. PMID: 22966037.
Article28. Ye D, Wang Y, Li H, Jia W, Man K, Lo CM, Wang Y, Lam KS, Xu A. Fibroblast growth factor 21 protects against acetaminopheninduced hepatotoxicity by potentiating peroxisome proliferatoractivated receptor coactivator protein-1α-mediated antioxidant capacity in mice. Hepatology. 2014; 60:977–989. PMID: 24590984.
Article29. Baird L, Dinkova-Kostova AT. The cytoprotective role of the Keap1-Nrf2 pathway. Arch Toxicol. 2011; 85:241–272. PMID: 21365312.
Article30. Lee KK, Imaizumi N, Chamberland SR, Alder NN, Boelsterli UA. Targeting mitochondria with methylene blue protects mice against acetaminophen-induced liver injury. Hepatology. 2015; 61:326–336. PMID: 25142022.
Article31. Hanawa N, Shinohara M, Saberi B, Gaarde WA, Han D, Kaplowitz N. Role of JNK translocation to mitochondria leading to inhibition of mitochondria bioenergetics in acetaminophen- induced liver injury. J Biol Chem. 2008; 283:13565–13577. PMID: 18337250.32. Dambach DM, Durham SK, Laskin JD, Laskin DL. Distinct roles of NF-kappaB p50 in the regulation of acetaminophen- induced inflammatory mediator production and hepatotoxicity. Toxicol Appl Pharmacol. 2006; 211:157–165. PMID: 16081117.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Recent Updates on Acetaminophen Hepatotoxicity: The Role of Nrf2 in Hepatoprotection
- The Combination of Gefitinib and Acetaminophen Exacerbates Hepatotoxicity via ROS-Mediated Apoptosis
- Four active monomers from Moutan Cortex exert inhibitoryeffects against oxidative stress by activating Nrf2/Keap1signaling pathway
- Diets with corn oil and/or low protein increase acute acetaminophen hepatotoxicity compared to diets with beef tallow in a rat model
- Rosmarinic Acid Inhibits Ultraviolet B-Mediated Oxidative Damage via the AKT/ERK-NRF2-GSH Pathway In Vitro and In Vivo