Korean J Physiol Pharmacol.
2015 Jan;19(1):29-34. 10.4196/kjpp.2015.19.1.29.
ERK Activation by Fucoidan Leads to Inhibition of Melanogenesis in Mel-Ab Cells
- Affiliations
-
- 1Department of Biochemistry, Chung-Ang University College of Medicine, Seoul 156-756, Korea. ds_kim@cau.ac.kr
- 2Department of Convergence Medicine and Pharmaceutical Biosciences, Chung-Ang University College of Medicine, Seoul 156-756, Korea.
- 3Thomas J. Long School of Pharmacy, University of the Pacific, Stockton, CA 95211, USA.
- KMID: 2071816
- DOI: http://doi.org/10.4196/kjpp.2015.19.1.29
Abstract
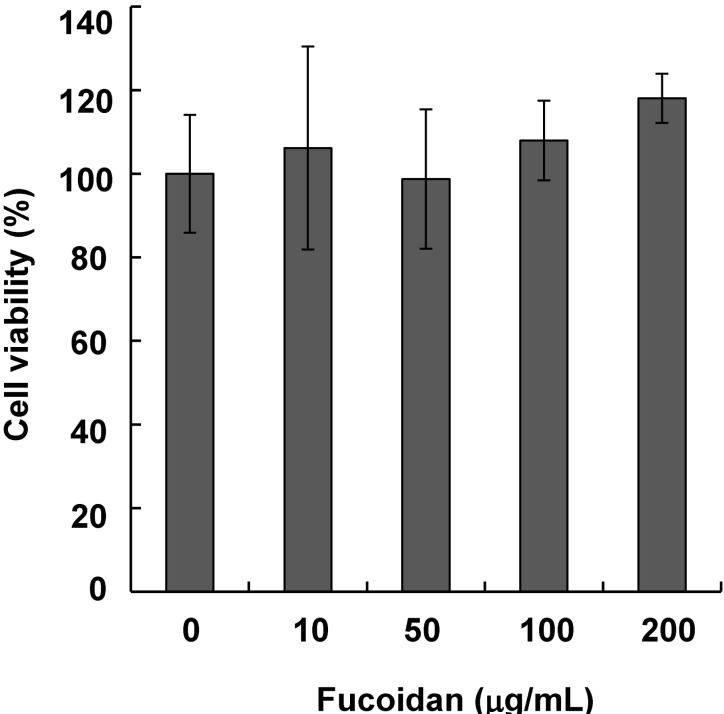
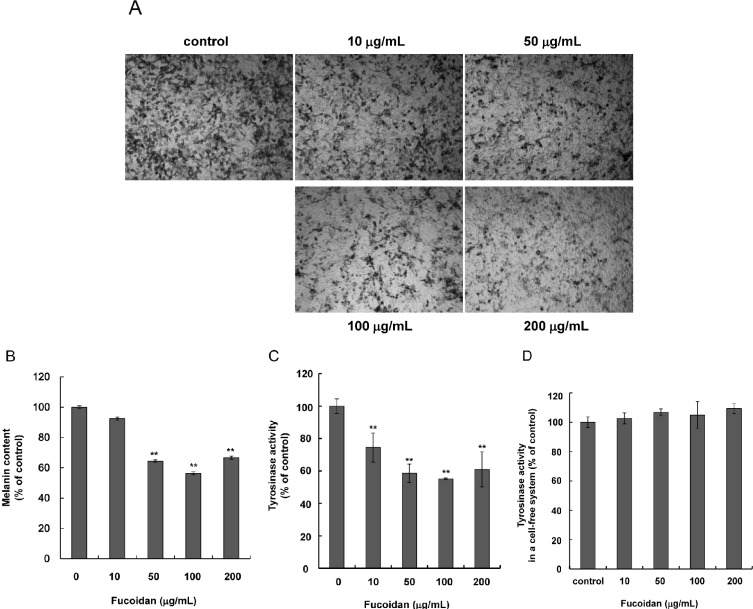
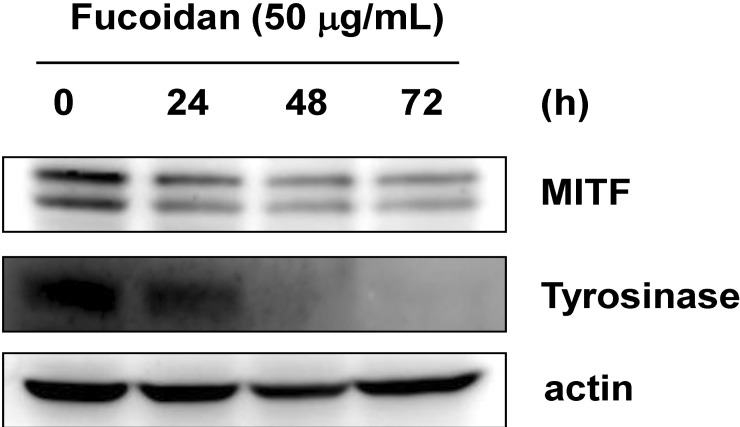
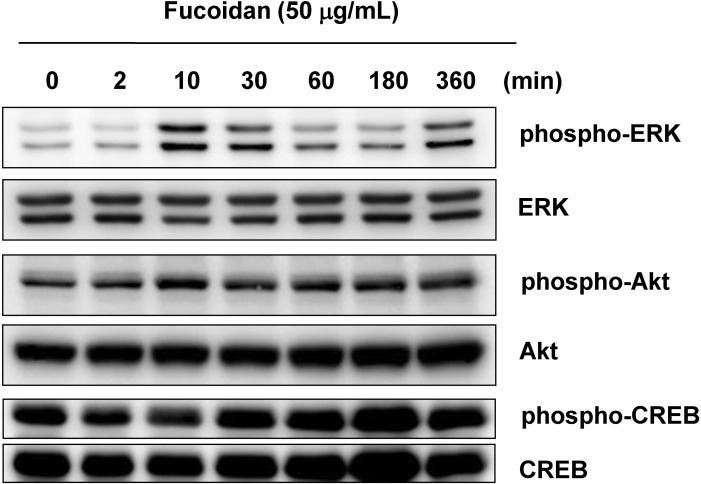
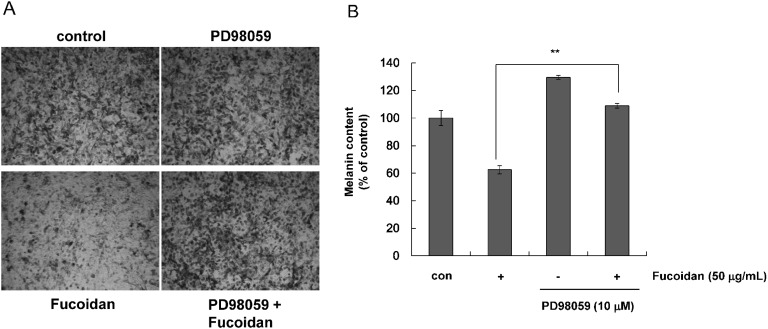
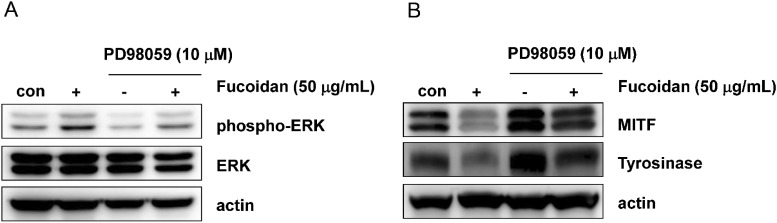
- Fucoidan, a fucose-rich sulfated polysaccharide derived from brown seaweed in the class Phaeophyceae, has been widely studied for its possible health benefits. However, the potential of fucoidan as a possible treatment for hyperpigmentation is not fully understood. This study investigated the effects of fucoidan on melanogenesis and related signaling pathways using Mel-Ab cells. Fucoidan significantly decreased melanin content. While fucoidan treatment decreased tyrosinase activity, it did not do so directly. Western blot analysis indicated that fucoidan downregulated microphthalmia-associated transcription factor and reduced tyrosinase protein expression. Further investigation showed that fucoidan activated the extracellular signal-regulated kinase (ERK) pathway, suggesting a possible mechanism for the inhibition of melanin synthesis. Treatment with PD98059, a specific ERK inhibitor, resulted in the recovery of melanin production. Taken together, these findings suggest that fucoidan inhibits melanogenesis via ERK phosphorylation.
Keyword
MeSH Terms
Figure
Reference
-
1. Ale MT, Maruyama H, Tamauchi H, Mikkelsen JD, Meyer AS. Fucoidan from Sargassum sp. and Fucus vesiculosus reduces cell viability of lung carcinoma and melanoma cells in vitro and activates natural killer cells in mice in vivo. Int J Biol Macromol. 2011; 49:331–336. PMID: 21624396.
Article2. Ale MT, Mikkelsen JD, Meyer AS. Important determinants for fucoidan bioactivity: a critical review of structure-function relations and extraction methods for fucose-containing sulfated polysaccharides from brown seaweeds. Mar Drugs. 2011; 9:2106–2130. PMID: 22073012.
Article3. Song YS, Li H, Balcos MC, Yun HY, Baek KJ, Kwon NS, Choi HR, Park KC, Kim DS. Fucoidan promotes the reconstruction of skin equivalents. Korean J Physiol Pharmacol. 2014; 18:327–331. PMID: 25177165.
Article4. Kondo T, Hearing VJ. Update on the regulation of mammalian melanocyte function and skin pigmentation. Expert Rev Dermatol. 2011; 6:97–108. PMID: 21572549.
Article5. Schiaffino MV. Signaling pathways in melanosome biogenesis and pathology. Int J Biochem Cell Biol. 2010; 42:1094–1104. PMID: 20381640.
Article6. Speeckaert R, Van Gele M, Speeckaert MM, Lambert J, van Geel N. The biology of hyperpigmentation syndromes. Pigment Cell Melanoma Res. 2014; 27:512–524. PMID: 24612852.
Article7. Gilchrest BA, Park HY, Eller MS, Yaar M. Mechanisms of ultraviolet light-induced pigmentation. Photochem Photobiol. 1996; 63:1–10. PMID: 8577860.
Article8. Vachtenheim J, Borovanský J. "Transcription physiology" of pigment formation in melanocytes: central role of MITF. Exp Dermatol. 2010; 19:617–627. PMID: 20201954.
Article9. Ando H, Kondoh H, Ichihashi M, Hearing VJ. Approaches to identify inhibitors of melanin biosynthesis via the quality control of tyrosinase. J Invest Dermatol. 2007; 127:751–761. PMID: 17218941.
Article10. Hearing VJ, Tsukamoto K. Enzymatic control of pigmentation in mammals. FASEB J. 1991; 5:2902–2909. PMID: 1752358.
Article11. Bertolotto C, Bille K, Ortonne JP, Ballotti R. In B16 melanoma cells, the inhibition of melanogenesis by TPA results from PKC activation and diminution of microphthalmia binding to the M-box of the tyrosinase promoter. Oncogene. 1998; 16:1665–1670. PMID: 9582014.
Article12. Kim EH, Kim MK, Yun HY, Baek KJ, Kwon NS, Park KC, Kim DS. Menadione (Vitamin K3) decreases melanin synthesis through ERK activation in Mel-Ab cells. Eur J Pharmacol. 2013; 718:299–304. PMID: 24012927.
Article13. Ahn MJ, Hur SJ, Kim EH, Lee SH, Shin JS, Kim MK, Uchizono JA, Whang WK, Kim DS. Scopoletin from Cirsium setidens Increases Melanin Synthesis via CREB Phosphorylation in B16F10 Cells. Korean J Physiol Pharmacol. 2014; 18:307–311. PMID: 25177162.14. Marshall CJ. Specificity of receptor tyrosine kinase signaling: transient versus sustained extracellular signal-regulated kinase activation. Cell. 1995; 80:179–185. PMID: 7834738.
Article15. Cowley S, Paterson H, Kemp P, Marshall CJ. Activation of MAP kinase kinase is necessary and sufficient for PC12 differentiation and for transformation of NIH 3T3 cells. Cell. 1994; 77:841–852. PMID: 7911739.
Article16. Sale EM, Atkinson PG, Sale GJ. Requirement of MAP kinase for differentiation of fibroblasts to adipocytes, for insulin activation of p90 S6 kinase and for insulin or serum stimulation of DNA synthesis. EMBO J. 1995; 14:674–684. PMID: 7882971.
Article17. Alesiani D, Cicconi R, Mattei M, Bei R, Canini A. Inhibition of Mek 1/2 kinase activity and stimulation of melanogenesis by 5,7-dimethoxycoumarin treatment of melanoma cells. Int J Oncol. 2009; 34:1727–1735. PMID: 19424591.
Article18. Li H, Kim J, Hahn HG, Yun J, Jeong HS, Yun HY, Baek KJ, Kwon NS, Min YS, Park KC, Kim DS. KHG26792 Inhibits Melanin Synthesis in Mel-Ab Cells and a Skin Equivalent Model. Korean J Physiol Pharmacol. 2014; 18:249–254. PMID: 24976765.
Article19. Kim DS, Park SH, Kwon SB, Park ES, Huh CH, Youn SW, Park KC. Sphingosylphosphorylcholine-induced ERK activation inhibits melanin synthesis in human melanocytes. Pigment Cell Res. 2006; 19:146–153. PMID: 16524430.
Article20. Kim DS, Hwang ES, Lee JE, Kim SY, Kwon SB, Park KC. Sphingosine-1-phosphate decreases melanin synthesis via sustained ERK activation and subsequent MITF degradation. J Cell Sci. 2003; 116:1699–1706. PMID: 12665551.
Article21. Oka M, Nagai H, Ando H, Fukunaga M, Matsumura M, Araki K, Ogawa W, Miki T, Sakaue M, Tsukamoto K, Konishi H, Kikkawa U, Ichihashi M. Regulation of melanogenesis through phosphatidylinositol 3-kinase-Akt pathway in human G361 melanoma cells. J Invest Dermatol. 2000; 115:699–703. PMID: 10998146.
Article22. Khaled M, Larribere L, Bille K, Ortonne JP, Ballotti R, Bertolotto C. Microphthalmia associated transcription factor is a target of the phosphatidylinositol-3-kinase pathway. J Invest Dermatol. 2003; 121:831–836. PMID: 14632202.
Article23. Kim BS, Park JY, Kang HJ, Kim HJ, Lee J. Fucoidan/FGF-2 induces angiogenesis through JNK- and p38-mediated activation of AKT/MMP-2 signalling. Biochem Biophys Res Commun. 2014; 450:1333–1338. PMID: 25003321.
Article24. Dooley TP, Gadwood RC, Kilgore K, Thomasco LM. Development of an in vitro primary screen for skin depigmentation and antimelanoma agents. Skin Pharmacol. 1994; 7:188–200. PMID: 8024800.
Article25. Ishiyama M, Tominaga H, Shiga M, Sasamoto K, Ohkura Y, Ueno K. A combined assay of cell viability and in vitro cytotoxicity with a highly water-soluble tetrazolium salt, neutral red and crystal violet. Biol Pharm Bull. 1996; 19:1518–1520. PMID: 8951178.
Article26. Buscà R, Bertolotto C, Ortonne JP, Ballotti R. Inhibition of the phosphatidylinositol 3-kinase/p70(S6)-kinase pathway induces B16 melanoma cell differentiation. J Biol Chem. 1996; 271:31824–31830. PMID: 8943224.
Article27. Wang N, Hebert DN. Tyrosinase maturation through the mammalian secretory pathway: bringing color to life. Pigment Cell Res. 2006; 19:3–18. PMID: 16420243.
Article28. Kim EH, Jeong HS, Yun HY, Baek KJ, Kwon NS, Park KC, Kim DS. Geranylgeranylacetone inhibits melanin synthesis via ERK activation in Mel-Ab cells. Life Sci. 2013; 93:226–232. PMID: 23792203.
Article29. Raghavendran HB, Sathivel A, Devaki T. Defensive nature of Sargassum polycystum (Brown alga) against acetaminopheninduced toxic hepatitis in rats: role of drug metabolizing microsomal enzyme system, tumor necrosis factor-alpha and fate of liver cell structural integrity. World J Gastroenterol. 2006; 12:3829–3834. PMID: 16804966.30. Chan YY, Kim KH, Cheah SH. Inhibitory effects of Sargassum polycystum on tyrosinase activity and melanin formation in B16F10 murine melanoma cells. J Ethnopharmacol. 2011; 137:1183–1188. PMID: 21810462.
Article31. Wang ZJ, Si YX, Oh S, Yang JM, Yin SJ, Park YD, Lee J, Qian GY. The effect of fucoidan on tyrosinase: computational molecular dynamics integrating inhibition kinetics. J Biomol Struct Dyn. 2012; 30:460–473. PMID: 22694253.
Article32. Bertolotto C, Abbe P, Hemesath TJ, Bille K, Fisher DE, Ortonne JP, Ballotti R. Microphthalmia gene product as a signal transducer in cAMP-induced differentiation of melanocytes. J Cell Biol. 1998; 142:827–835. PMID: 9700169.
Article33. Lee HE, Kim EH, Choi HR, Sohn UD, Yun HY, Baek KJ, Kwon NS, Park KC, Kim DS. Dipeptides Inhibit Melanin Synthesis in Mel-Ab Cells through Down-Regulation of Tyrosinase. Korean J Physiol Pharmacol. 2012; 16:287–291. PMID: 22915995.
Article34. Buscà R, Ballotti R. Cyclic AMP a key messenger in the regulation of skin pigmentation. Pigment Cell Res. 2000; 13:60–69. PMID: 10841026.35. Dent P. Crosstalk between ERK, AKT, and cell survival. Cancer Biol Ther. 2014; 15:245–246. PMID: 24424114.
Article36. Ocana A, Vera-Badillo F, Al-Mubarak M, Templeton AJ, Corrales-Sanchez V, Diez-Gonzalez L, Cuenca-Lopez MD, Seruga B, Pandiella A, Amir E. Activation of the PI3K/mTOR/AKT pathway and survival in solid tumors: systematic review and meta-analysis. PLoS One. 2014; 9:e95219. PMID: 24777052.
Article37. Khaled M, Larribere L, Bille K, Aberdam E, Ortonne JP, Ballotti R, Bertolotto C. Glycogen synthase kinase 3beta is activated by cAMP and plays an active role in the regulation of melanogenesis. J Biol Chem. 2002; 277:33690–33697. PMID: 12093801.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Dipeptides Inhibit Melanin Synthesis in Mel-Ab Cells through Down-Regulation of Tyrosinase
- Involvement of Transglutaminase-2 in alpha-MSH-Induced Melanogenesis in SK-MEL-2 Human Melanoma Cells
- Antitumor Effects of Fucoidan on Human Colon Cancer Cells via Activation of Akt Signaling
- Fucoidan Induces Apoptosis in A2058 Cells through ROS-exposed Activation of MAPKs Signaling Pathway
- Anti-inflammatory Activity of Fucoidan with Blocking NF-kappaB and STAT1 in Human Keratinocytes Cells