J Korean Endocr Soc.
2008 Feb;23(1):18-26. 10.3803/jkes.2008.23.1.18.
Naloxone Increases the Anorexic Effect of MTII in OLETF Rats
- Affiliations
-
- 1Department of Neurosurgery, Joeun Hospital, Korea.
- 2Department of Physiology, College of Medicine, Yeungnam University, Korea.
- 3Department of Pediatrics, College of Medicine, Yeungnam University, Korea.
- 4Department of Neurosurgery, College of Medicine, Yeungnam University, Korea.
- 5Department of Internal Medicine, College of Medicine, Yeungnam University, Korea.
- KMID: 2063546
- DOI: http://doi.org/10.3803/jkes.2008.23.1.18
Abstract
-
BACKGROUND: Leptin, an adipocyte-derived hormone, inhibits obesity in lean subjects, but is not widely used because of leptin resistance. Thus, circumventing the arcuate nucleus of the hypothalamus, the site responsible for leptin resistance, has been evaluated for treatment of obesity. However, chronic treatment of melanotan II (MTII), a synthetic agonist of the melanocortin 3/4 receptor, induces tachyphylaxis. Here, we evaluated whether naloxone, a non-specific agouti-related peptide (AgRP) antagonist, increases the anorexic effect of MTII in Otsuka Long-Evans Tokushima Fatty (OLETF) rats.
METHODS
We measured food intake following intracerebroventricular (i.c.v.) infusion of MTII and/or naloxone in OLETF rats. Sprague-Dawley rats were used as a normal control group.
RESULTS
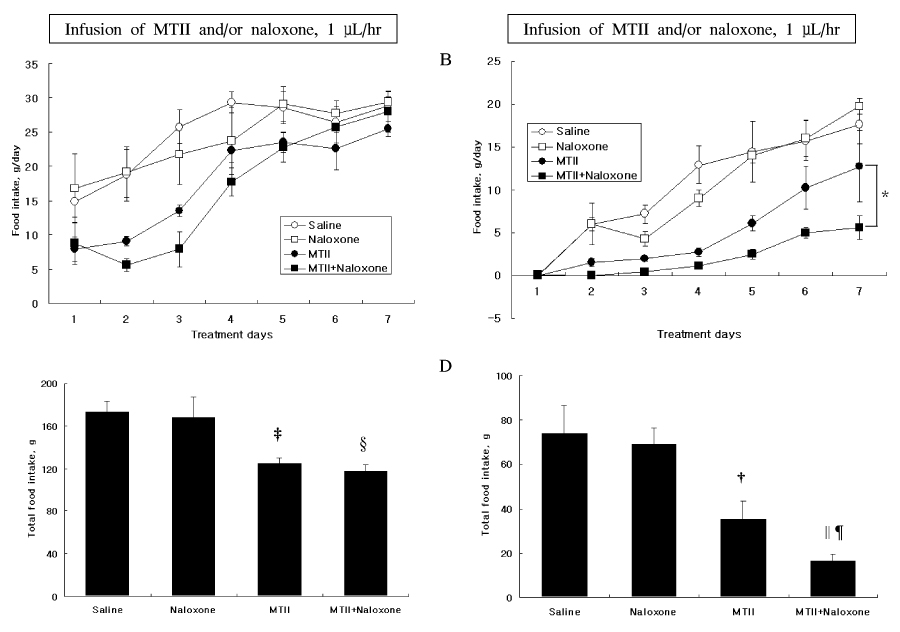
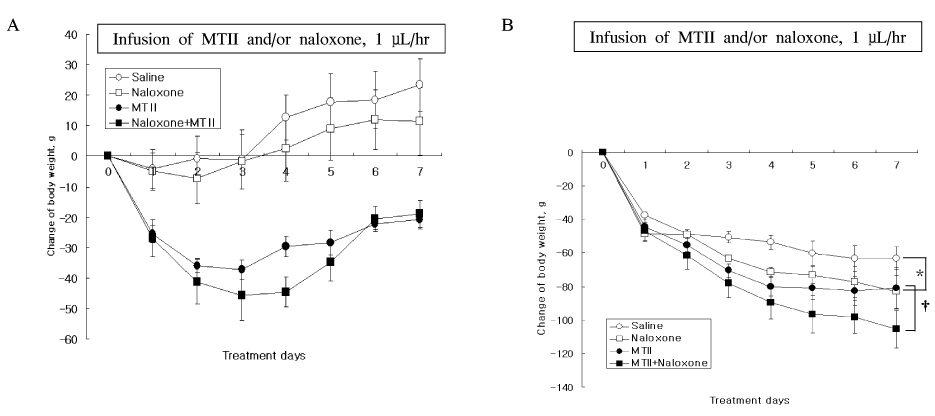
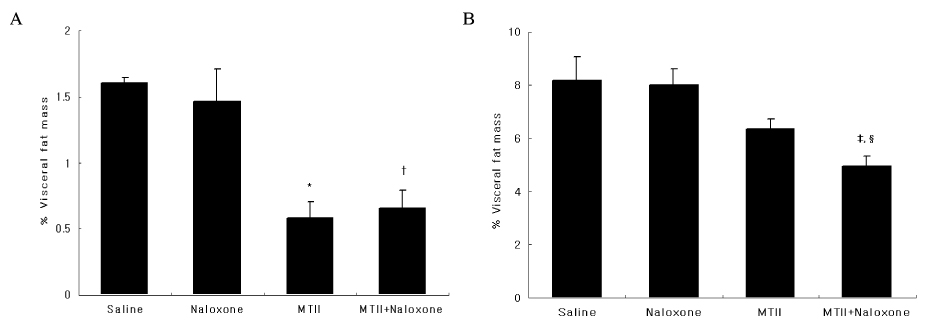
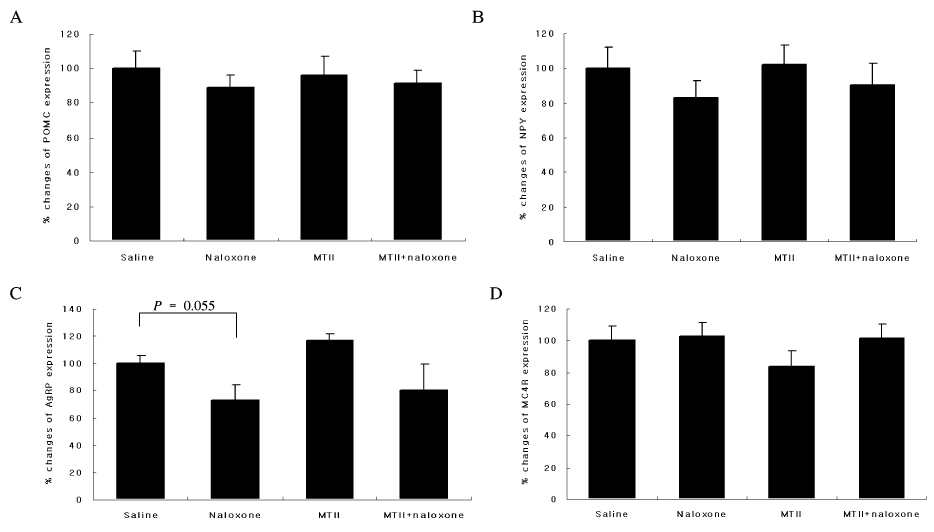
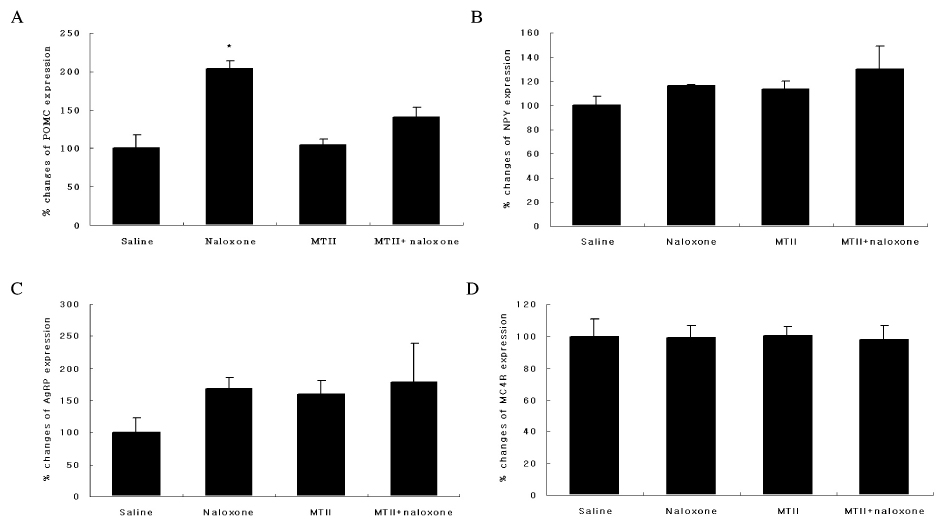
The anorexic effect of i.c.v. MTII infusion decreased with time in OLETF rats, indicating the development of tachyphylaxis. In normal control rats, naloxone alone decreased AgRP expression in the hypothalamus but failed to induce anorexia. Moreover, there was no additional anorexic effect with co-treatment of naloxone and MTII. In OLETF rats, naloxone alone did not show an anorexic effect despite increased POMC expression in the hypothalamus. However, naloxone sensitized the anorexic effect of MTII when treated together.
CONCLUSION
These results suggest that naloxone augmented the anorexic effect of MTII when treated together in OLETF rats, but had no effect alone. These results suggest that a combination therapy of naloxone and a melanocortin receptor activator would be an effective modality for treatment of obesity.
Keyword
MeSH Terms
Figure
Reference
-
1. Zhang Y, Proenca R, Maffei M, Barone M, Leopold L, Friedman JM. Positional cloning of the mouse obese gene and its human homologue. Nature. 1994. 372:425–432.2. Scarpace PJ, Tümer N. Peripheral and hypothalamic leptin resistance with age-related obesity. Physiol Behav. 2001. 74:721–727.3. Shek EW, Scarpace PJ. Resistance to the anorexic and thermogenic effects of centrally administrated leptin in obese aged rats. Regul Pept. 2000. 92(1-3):65–71.4. Van Heek M, Compton DS, France CF, Tedesco RP, Fawzi AB, Graziano MP, Sybertz EJ, Strader CD, Davis HR Jr. Diet-induced obese mice develop peripheral, but not central, resistance to leptin. J Clin Invest. 1997. 99:385–390.5. Widdowson PS, Upton R, Buckingham R, Arch J, Williams G. Inhibition of food response to intracerebroventricular injection of leptin is attenuated in rats with diet-induced obesity. Diabetes. 1997. 46:1782–1785.6. Caro JF, Kolaczynski JW, Nyce MR, Ohannesian JP, Opentanova I, Goldman WH, Lynn RB, Zhang PL, Sinha MK, Considine RV. Decraesed cerebrospinal-fluid/serum leptin ratio in obesity, a possible mechanism for leptin resistance. Lancet. 1996. 348:159–161.7. Halaas JL, Boozer C, Blair-West J, Fidahusein N, Denton DA, Friedman JM. Physiological response to long term peripheral and central leptin infusion in lean and obese mice. Proc Natl Acad Sci USA. 1997. 94:8878–8883.8. Jonsson L, Skarphedinsson JO, Skuladottir GV, Atlason PT, Eiriksdottir VH, Franzson L, Schiöth HB. Melanocortin receptor agonist transiently increases oxygen consumption in rats. Neuroreport. 2001. 12:3703–3708.9. Scarpace PJ, Matheny M, Zolotukhin S, Tümer N, Zhang Y. Leptin-induced leptin resistant rats exhibit enhanced responses to the melanocortin agonist MT II. Neuropharmacology. 2003. 45:211–219.10. Kim YW, Choi DW, Park YH, Huh JY, Won KC, Choi KH, Park SY, Kim JY, Lee SK. Leptin-like effect of MTII are augmented in MSG-obese rats. Regul Pept. 2005. 127:63–70.11. Brugman S, Clegg DJ, Woods SC, Seeley RJ. Combined blockade of both micro- and kappa-opioid receptors prevents the acute orexigenic action of Agouti-related protein. Endocrinology. 2002. 143:4265–4270.12. Dube MG, Horvath TL, Leranth C, Kalra PS, Kalra SP. Naloxone reduces the feeding evoked by intracerebroventricular galanin injection. Physiol Behav. 1994. 56:811–813.13. Kim YW, Kim JY, Park YH, Park YH, Park SY, Won KC, Choi KH, Huh JY, Moon KH. Metformin Restores Leptin Sensitivity in High Fat Fed Obese Rats with Leptin Resistance. Diabetes. 2006. 55:716–724.14. Covasa M, Ritter RC. Attenuated satiation response to intestinal nutrients in rats that do not express CCK-A receptors. Peptides. 2001. 22:1339–1348.15. Spiegelman BM, Flier JS. Obesity and the regulation of energy balance. Cell. 2001. 104:531–543.16. Li G, Zhang Y, Wilsey JT, Scarpace PJ. Hypothalamic pro-opiomelanocortin gene delivery ameliorates obesity and glucose intolerance in aged rats. Diabetologia. 2005. 48:2376–2385.17. Li G, Zhang Y, Cheng KY, Scarpace PJ. Lean rats with hypothalamic pro-opiomelanocortin overexpression exhibit greater diet-induced obesity and impaired central melanocortin responsiveness. Diabetologia. 2007. 50:1490–1499.18. Fong TM, Mao C, MacNeil T, Kalyani R, Smith T, Weinberg D, Tota MR, Van der Ploeg LH. ART (protein product of agouti-related transcript) as an antagonist of MC-3 and MC-4 receptors. Biochem Biophys Res Commun. 1997. 237:629–631.19. Ollmann MM, Wilson BD, Yang YK, Kerns JA, Chen Y, Gantz I, Barsh GS. Antagonism of central melanocortin receptors in vitro and in vivo by agouti-related protein. Science. 1997. 278:135–138.20. Wortley KE, Anderson KD, Yasenchak J, Murphy A, Valenzuela D, Diano S, Yancopoulos GD, Wiegand SJ, Sleeman MW. Agouti-related protein-deficient mice display an age-related lean phenotype. Cell Metab. 2005. 2:421–427.21. Rossi M, Kim MS, Morgan DG, Small CJ, Edwards CM, Sunter D, Abusnana S, Goldstone AP, Russell SH, Stanley SA, Smith DM, Yagaloff K, Ghatei MA, Bloom SR. A C-terminal fragment of agouti-related protein increases feeding and antagonizes the effect of α-melanocyte stimulating hormone in vivo. Endocrinology. 1998. 139:4428–4431.22. Hagan MM, Rushing PA, Pritchard LM, Schwartz MW, Strack AM, Van der Ploeg LH, Woods SC, Seeley RJ. Long-term orexigenic effects of AgRP-(83-132) involve mechanisms other than melanocortin receptor blockade. Am J Physiol. 2000. 279:R47–R52.23. Hagan MM, Rushing PA, Pritchard LM, Schwartz MW, Strack AM, Van der Ploeg HT, Woods SC, Seeley RJ. Long-term orexigenic effects of AgRP-(83-132) involve mechanisms other than melanocortin receptor blockade. Am J Physiol. 2000. 279:R47–R52.24. Kotz CM, Grace MK, Briggs J, Levine AS, Billington CJ. Effects of opioid antagonists naloxone and naltrexone on neuropeptide Y-induced feeding and brown fat thermogenesis in the rat. Neural site of action. J Clin Invest. 1995. 96:163–170.25. Tabarin A, Diz-Chaves Y, Carmona Mdel C, Catargi B, Zorrilla EP, Roberts AJ, Coscina DV, Rousset S, Redonnet A, Parker GC, Inoue K, Ricquier D, Pénicaud L, Kieffer BL, Koob GF. Resistance to diet-induced obesity in mu-opioid receptor-deficient mice: evidence for a "thrifty gene". Diabetes. 2005. 54(12):3510–3516.26. Schusdziarra V, Zimmermann JP, Schick RR. Importance of orexigenic counter-regulation for multiple targeted feeding inhibition. Obes Res. 2004. 12:627–632.27. MaLaughlin CL, Baile CA. Feeding behavior responses of Zucker rats to naloxone. Physiol Behav. 1984. 32:755–761.28. Jarosz PA, Metzger BL. The effect of opioid antagonism on food intake behavior and body weight in a biobehavioral model of obese binge eating. Biol Res Nurs. 2002. 3:198–209.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Naloxone Increases the Anorexic Effect of Melanocortin II
- The Effect of Leptin Level Fluctuations by a Repeated Fasting/Refeeding on the Leptin Sensitivity in OLETF Rats
- The Effect of Leptin Level Fluctuations by a Repeated Fasting/Refeeding on the Leptin Sensitivity in OLETF Rats
- Effects of Intracerebroventricular Infusion of MTII in Monosodium Glutamate(MSG)-obese Rats
- Reduction of Food Intake by Fenofibrate is Associated with Cholecystokinin Release in Long-Evans Tokushima Rats