Korean J Gynecol Oncol.
2008 Mar;19(1):26-39. 10.3802/kjgo.2008.19.1.26.
Induction of apoptosis by the kinase inhibitor flavopiridol in human ovarian cancer cell lines
- Affiliations
-
- 1Department of Obstetrics and Gynecology, College of Medicine, The Catholic University of Korea, Seoul, Korea. leejm@catholic.ac.kr
- KMID: 1979499
- DOI: http://doi.org/10.3802/kjgo.2008.19.1.26
Abstract
-
OBJECTIVE
Flavopiridol that inhibits cyclin-dependent kinase, can cause cell cycle arrest, induce apoptosis in human tumor cell lines. In the present study, we investigated apoptotic effects of flavopiridol and the underlying molecular mechanisms in human ovarian cancer cell lines.
METHODS
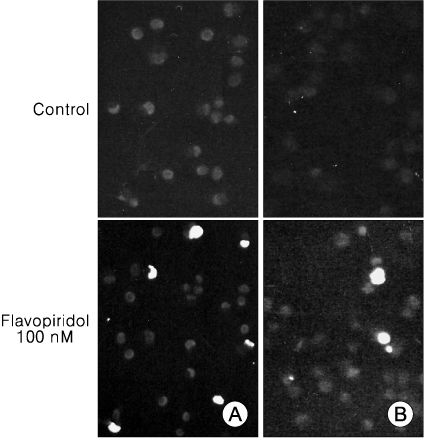
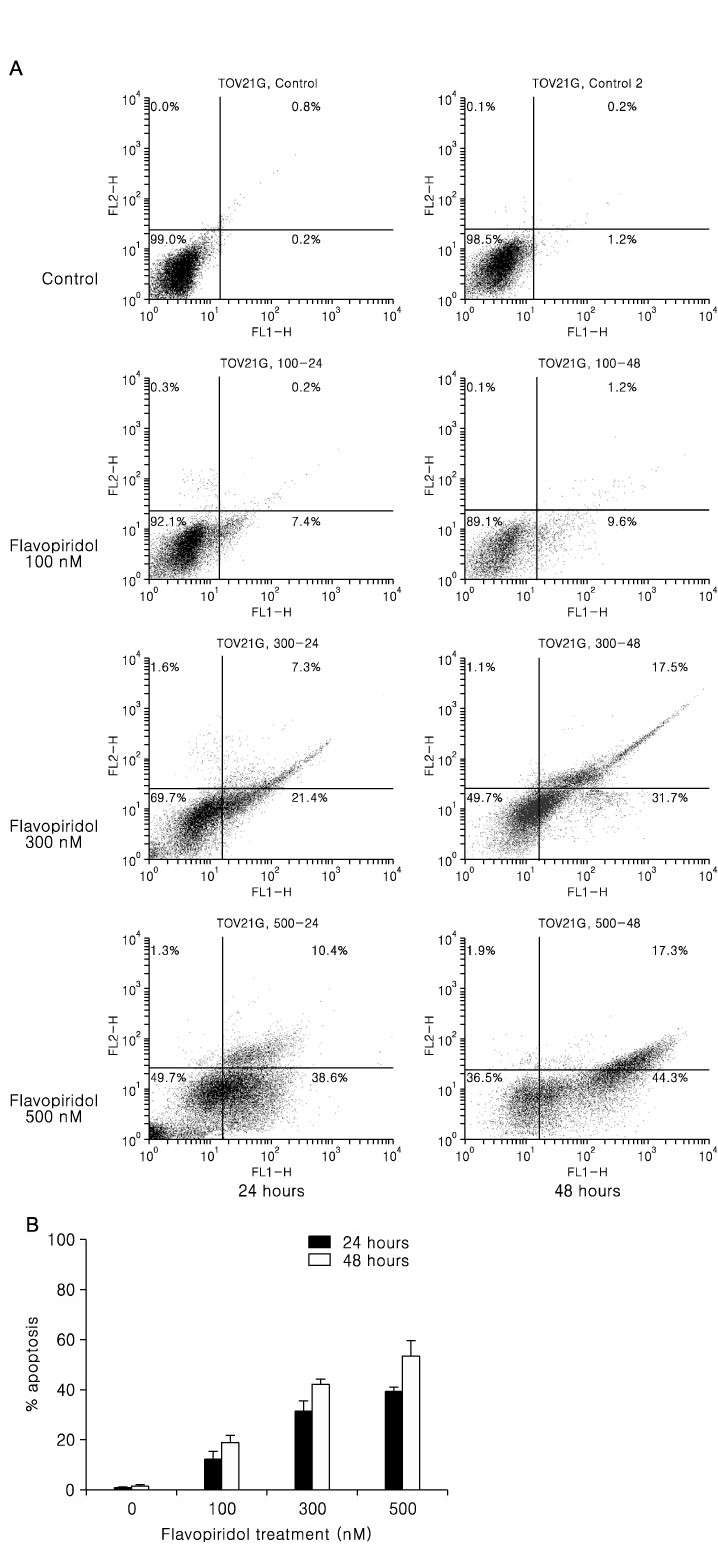
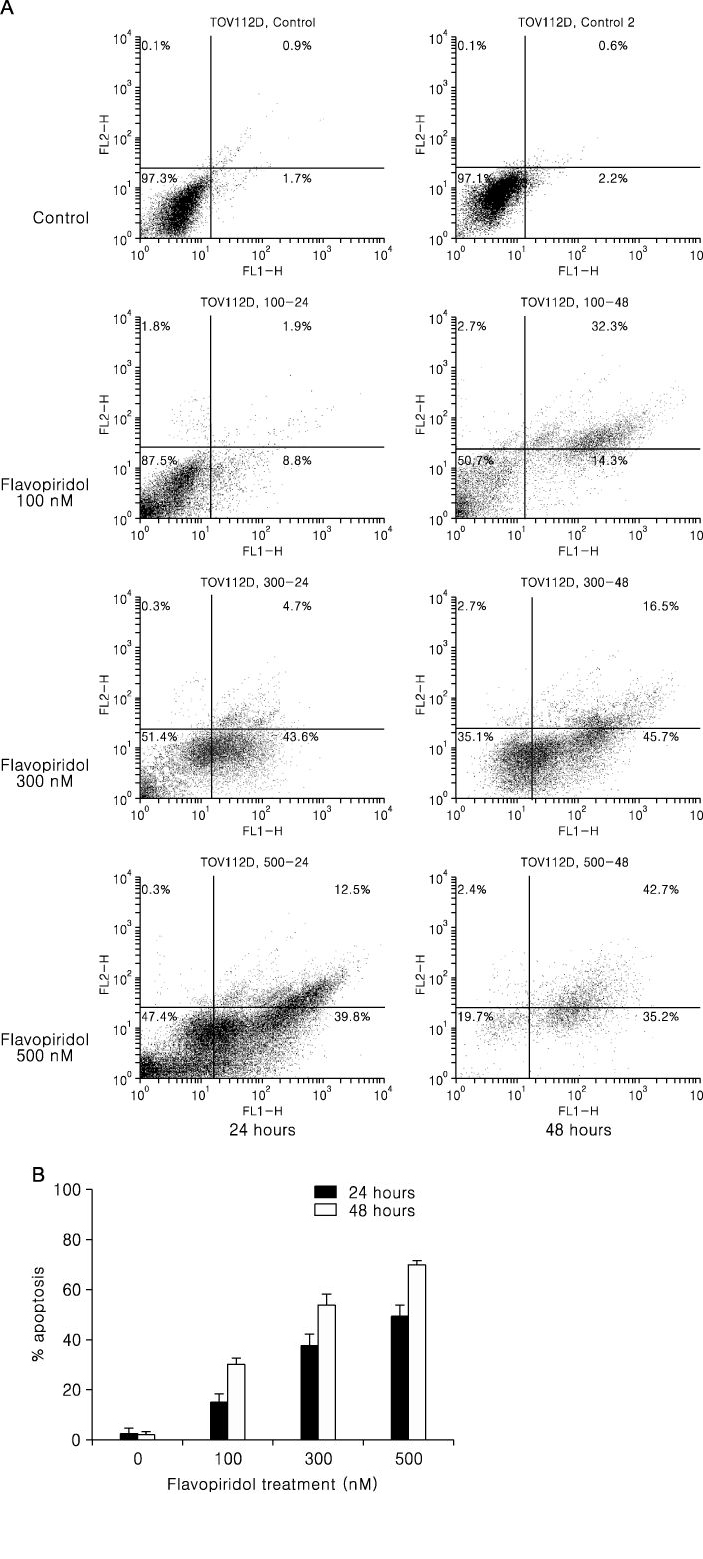
We used TOV-21G and TOV-112D cell lines. The cell viability was tested by MTT assay and apoptosis was assessed by TUNEL assay and annexin-V binding. Western blot was used to examine apoptosis related protein levels. MAP kinase activity was analyzed by non-radioactive MAP kinase assay kit.
RESULTS
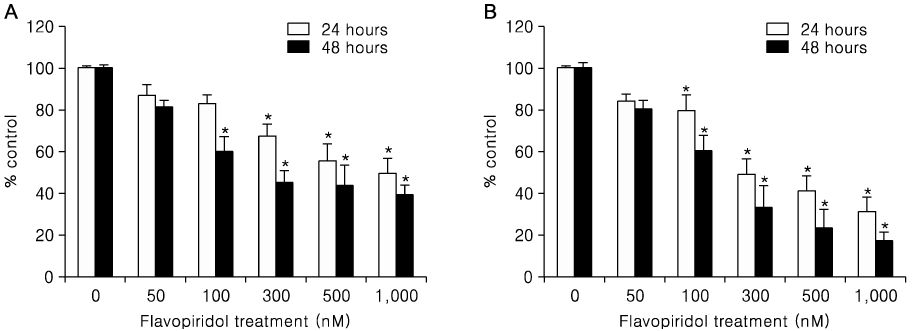
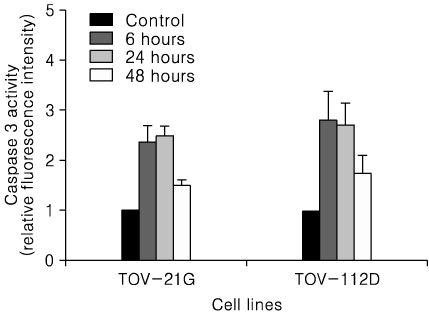
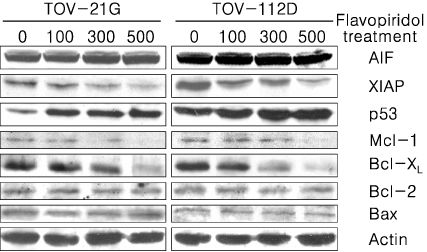
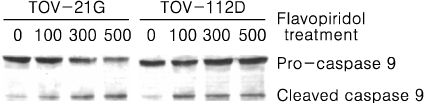
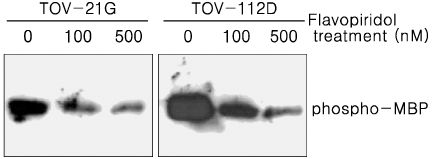
Treatment of TOV-21G and TOV-112D cells with flavopiridol (50 nM to 1000 nM) led to a dose- and time-dependent inhibition of cell growth and survival. Dose-related induction of apoptosis was also observed in these cell lines. Flavopiridol (500 nM) induced striking decreases in the levels of the antiapoptic proteins Mcl-1, Bcl-X(L), and XIAP in both cell lines. In contrast, expression of Bax, Bcl-2, and AIF was not significantly influenced by flavopiridol. Although flavopiridol resulted in accumulation of p53 in both cells, flavopiridol mediated apoptosis was p53 independent because it occurred to the same degree in TOV-112D cells in which p53 was inactivated by mutation. Flavopiridol treatment resulted in enhanced cleavage of pro-caspase 9 and activation of caspase 3. Apoptosis was associated with suppression of ERK activity.
CONCLUSION
Although the precise mechanisms of flavopiridol mediated cytotoxicity have not been fully defined, these data suggest that flavopiridol has activity against ovarian cancers in vitro and is worthy of continued clinical development in the treatment of ovarian cancer.
Keyword
MeSH Terms
Figure
Reference
-
1. Kaur G, Stetler-Stevenson M, Sebers S, Worland P, Sedlacek H, Myers C, et al. Growth inhibition with reversible cell cycle arrest of carcinoma cells by flavone L86-8275. J Natl Cancer Inst. 1992. 84:1736–1740.
Article2. Zhai S, Senderowicz AM, Sausville EA, Figg WD. Flavopiridol, a novel cyclin-dependent kinase inhibitor, in clinical development. Ann Pharmacother. 2002. 36:905–911.
Article3. Worland PJ, Kaur G, Stetler-Stevenson M, Sebers S, Sartor O, Sausville EA. Alteration of the phosphorylation state of p34cdc2 kinase by the flavone L86-8275 in breast carcinoma cells. Correlation with decreased H1 kinase activity. Biochem Pharmacol. 1993. 46:1831–1840.4. De Azevedo WF Jr, Mueller-Dieckmann HJ, Schulze-Gahmen U, Worland PJ, Sausville E, Kim SH. Structural basis for specificity and potency of a flavonoid inhibitor of human CDK2, a cell cycle kinase. Proc Natl Acad Sci USA. 1996. 93:2735–2740.
Article5. Carlson B, Lahusen T, Singh S, Loaiza-Perez A, Worland PJ, Pestell R, et al. Down-regulation of cyclin D1 by transcriptional repression in MCF-7 human breast carcinoma cells induced by flavopiridol. Cancer Res. 1999. 59:4634–4641.6. Sedlacek HH. Mechanisms of action of flavopiridol. Crit Rev Oncol Hematol. 2001. 38:139–170.
Article7. Melillo G, Sausville EA, Cloud K, Lahusen T, Varesio L, Senderow AM. Flavopiridol, a protein kinase inhibitor, down-regulates hypoxic induction of vascular endothelial growth factor expression in human monocytes. Cancer Res. 1999. 59:5433–5437.8. Arguello F, Alexander M, Sterry JA, Tudor G, Smith EM, Kalavar NT, et al. Flavopiridol induces apoptosis of normal lymphoid cells, causes immunosuppression, and has potent antitumor activity In vivo against human leukemia and lymphoma xenografts. Blood. 1998. 91:2482–2490.9. Patel V, Senderowicz AM, Pinto D Jr, Igishi T, Raffeld M, Quintanilla-Martinez L, et al. Flavopiridol, a novel cyclin-dependent kinase inhibitor, suppresses the growth of head and neck squamous cell carcinomas by inducing apoptosis. J Clin Invest. 1998. 102:1674–1681.
Article10. Parker BW, Kaur G, Nieves-Neira W, Taimi M, Kohlhagen G, Shimizu T, et al. Early induction of apoptosis in hematopoietic cell lines after exposure to flavopiridol. Blood. 1998. 91:458–465.
Article11. Achenbach TV, Muller R, Slater EP. Bcl-2 independence of flavopiridol-induced apoptosis. Mitochondrial depolarization in the absence of cytochrome c release. J Biol Chem. 2000. 275:32089–32097.12. Kitada S, Zapata JM, Andreeff M, Reed JC. Protein kinase inhibitors flavopiridol and 7-hydroxy-staurosporine down-regulate antiapoptosis proteins in B-cell chronic lymphocytic leukemia. Blood. 2000. 96:393–397.
Article13. Raju U, Nakata E, Mason KA, Ang KK, Milas L. Flavopiridol, a cyclin-dependent kinase inhibitor, enhances radiosensitivity of ovarian carcinoma cells. Cancer Res. 2003. 63:3263–3267.14. Motwani M, Delohery TM, Schwartz GK. Sequential dependent enhancement of caspase activation and apoptosis by flavopiridol on paclitaxel-treated human gastric and breast cancer cells. Clin Cancer Res. 1999. 5:1876–1883.15. Shapiro GI. Preclinical and clinical development of the cyclin-dependent kinase inhibitor flavopiridol. Clin Cancer Res. 2004. 10:4270s–4275s.
Article16. Provencher DM, Lounis H, Champoux L, Tetrault M, Manderson EN, Wang JC, et al. Characterization of four novel epithelial ovarian cancer cell lines. In Vitro Cell Dev Biol Anim. 2000. 36:357–361.
Article17. Shapiro GI, Koestner DA, Matranga CB, Rollins BJ. Flavopiridol induces cell cycle arrest and p53-independent apoptosis in non-small cell lung cancer cell lines. Clin Cancer Res. 1999. 5:2925–2938.18. Samouelian V, Maugard CM, Jolicoeur M, Bertrand R, Arcand SL, Tonin PN, et al. Chemosensitivity and radiosensitivity profiles of four new human epithelial ovarian cancer cell lines exhibiting genetic alterations in BRCA2, TGFbeta-RII, KRAS2, TP53 and/or CDNK2A. Cancer Chemother Pharmacol. 2004. 54:377–383.19. Wittmann S, Bali P, Donapaty S, Nimmanapalli R, Guo F, Yamaguchi H, et al. Flavopiridol down-regulates antiapoptotic proteins and sensitizes human breast cancer cells to epothilone B-induced apoptosis. Cancer Res. 2003. 63:93–99.20. Gojo I, Zhang B, Fenton RG. The cyclin-dependent kinase inhibitor flavopiridol induces apoptosis in multiple myeloma cells through transcriptional repression and down-regulation of Mcl-1. Clin Cancer Res. 2002. 8:3527–3538.21. Ma Y, Cress WD, Haura EB. Flavopiridol-induced apoptosis is mediated through up-regulation of E2F1 and repression of Mcl-1. Mol Cancer Ther. 2003. 2:73–81.22. Susin SA, Lorenzo HK, Zamzami N, Marzo I, Snow BE, Brothers GM, et al. Molecular characterization of mitochondrial apoptosis-inducing factor. Nature. 1999. 397:441–446.
Article23. Cande C, Cecconi F, Dessen P, Kroemer G. Apoptosis-inducing factor (AIF): Key to the conserved caspase-independent pathways of cell death? J Cell Sci. 2002. 115:4727–4734.24. Deveraux QL, Takahashi R, Salvesen GS, Reed JC. X-linked IAP is a direct inhibitor of cell-death proteases. Nature. 1997. 388:300–304.
Article25. Woo M, Hakem R, Soengas MS, Duncan GS, Shahinian A, Kagi D, et al. Essential contribution of caspase 3/CPP32 to apoptosis and its associated nuclear changes. Genes Dev. 1998. 12:806–819.26. Laframboise S, Chapman W, McLaughlin J, Andrulis IL. p53 mutations in epithelial ovarian cancers: possible role in predicting chemoresistance. Cancer J. 2000. 6:302–308.27. Schuijer M, Berns EM. TP53 and ovarian cancer. Hum Mutat. 2003. 21:285–291.
Article28. Robinson MJ, Cobb MH. Mitogen-activated protein kinase pathways. Curr Opin Cell Biol. 1997. 9:180–186.
Article29. Xia Z, Dickens M, Raingeaud J, Davis RJ, Greenberg ME. Opposing effects of ERK and JNK-p38 MAP kinases on apoptosis. Science. 1995. 270:1326–1331.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Flavopiridol on Radiation-induced Apoptosis of Human Laryngeal and Lung Cancer Cells
- Induction of Apoptosis and Inhibition of Cellular Proliferation in Aspirin-treated SNU-668 Human Gastric Adenocarcinoma Cell Lines
- Effects of p27 Overexpression on Head and Neck Squamous Cell Carcinoma Cell Lines
- Synergistic Induction of Apoptosis by the Combination of an Axl Inhibitor and Auranofin in Human Breast Cancer Cells
- Enhancement of Radiation Effects by Flavopiridol in Uterine Cervix Cancer Cells