J Korean Med Sci.
2013 Oct;28(10):1435-1442. 10.3346/jkms.2013.28.10.1435.
Expression of Semaphorin 3A and Neuropilin 1 in Asthma
- Affiliations
-
- 1Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea. guinea71@snu.ac.kr
- 2Institute of Allergy and Clinical Immunology, Seoul National University College of Medicine, Seoul, Korea.
- KMID: 1777678
- DOI: http://doi.org/10.3346/jkms.2013.28.10.1435
Abstract
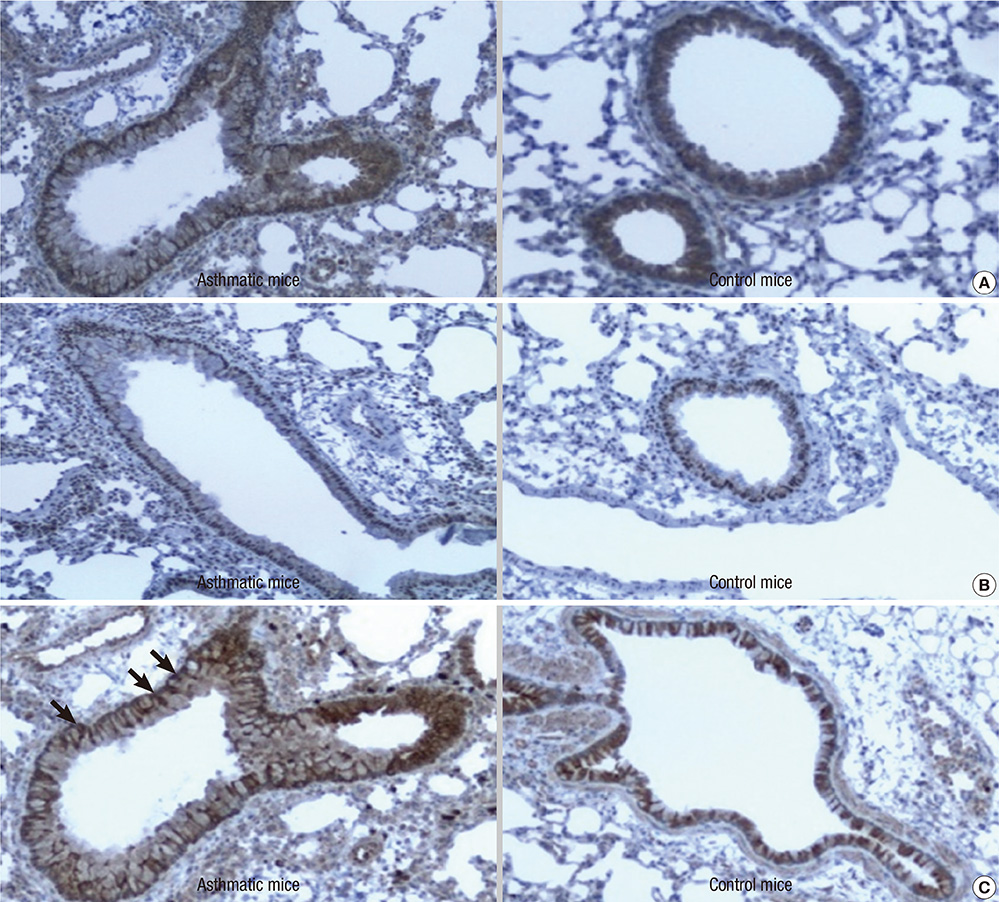
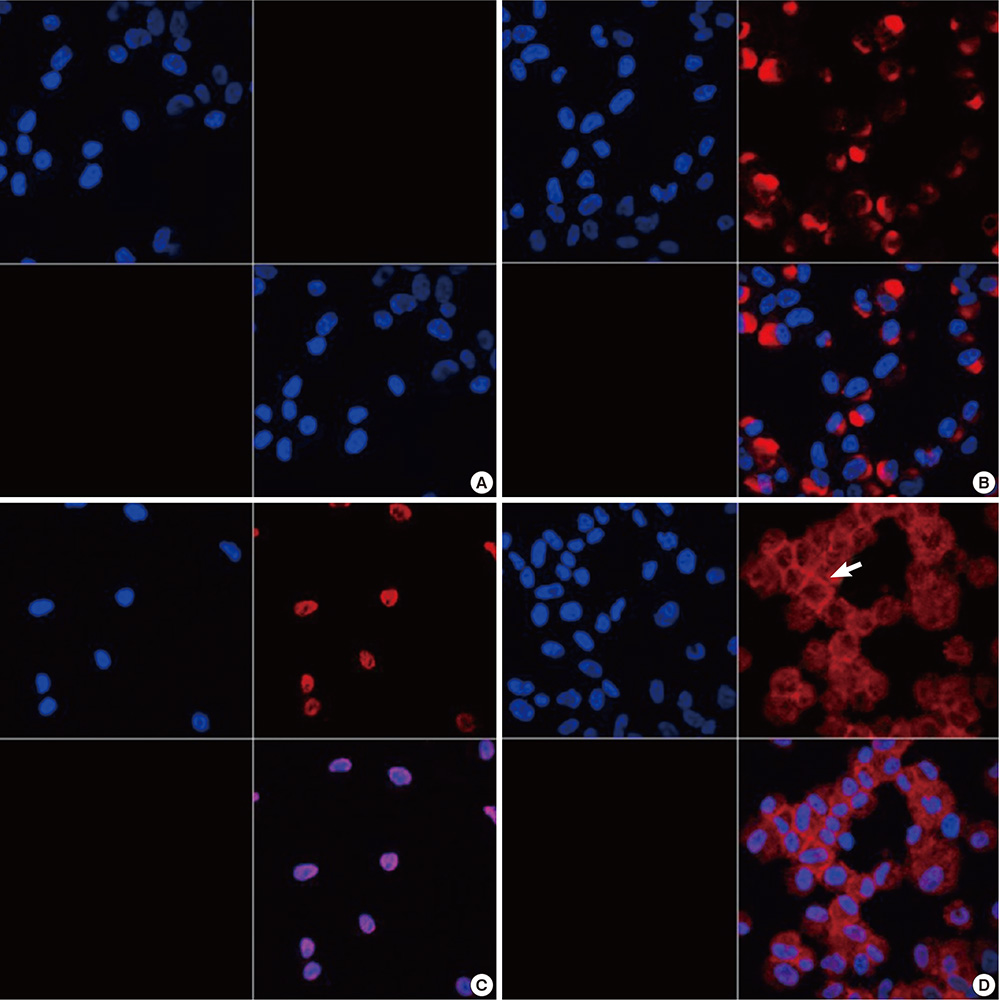
- Neuropilin 1 (NP1) is a part of essential receptor complexes mediating both semaphorin3A (SEMA3A) and vascular endothelial growth factor (VEGF) which is one of important mediators involved in the pathogenesis of asthma. Therefore, it is possible that SEMA3A plays a role in the pathogenesis of asthma through attenuation of VEGF-mediated effects. In the present study, we aimed to evaluate expression levels of SEMA3A and NP1 using induced sputum of asthmatics and a murine model of asthma. Firstly, SEMA3A and NP1 expressions in induced sputum of asthmatics and SEMA3A and NP1 expression on bronchoalveolar lavage (BAL) cells and lung homogenates of asthmatic mice were determined. Then we evaluated the immunolocalization of VEGF receptor 1 (VEGFR1), VEGF receptor 2 (VEGFR2), and NP1 expressions on asthmatic mice lung tissue and their subcellular distributions using fibroblast and BEAS2B cell lines. Sputum SEMA3A and NP1 expressions were significantly higher in asthmatics than controls. Similarly, SEMA3A and NP1 expressions on BAL cells and lung homogenates were significantly elevated in asthmatic mice compared to control mice. Immunohistochemical analysis showed that VEGFR1, VEGFR2, and NP1 expressions were also uniformly increased in asthmatic mice. Our observations suggest that SEMA3A and NP1 may play important roles in the pathogenesis of asthma.
MeSH Terms
-
Animals
Asthma/metabolism/pathology/*physiopathology
Bronchoalveolar Lavage Fluid/cytology
Cell Line
Disease Models, Animal
Female
Fibroblasts/metabolism
*Gene Expression Regulation
Immunohistochemistry
Lung/metabolism
Male
Mice
Mice, Inbred C57BL
Neuropilin-1/*genetics/metabolism
Semaphorin-3A/*genetics/metabolism
Sputum/metabolism
Vascular Endothelial Growth Factor Receptor-1/metabolism
Vascular Endothelial Growth Factor Receptor-2/metabolism
Neuropilin-1
Semaphorin-3A
Vascular Endothelial Growth Factor Receptor-1
Vascular Endothelial Growth Factor Receptor-2
Figure
Reference
-
1. Holgate ST. Airway inflammation and remodeling in asthma: current concepts. Mol Biotechnol. 2002; 22:179–189.2. Gerber HP, Dixit V, Ferrara N. Vascular endothelial growth factor induces expression of the antiapoptotic proteins Bcl-2 and A1 in vascular endothelial cells. J Biol Chem. 1998; 273:13313–13316.3. Clauss M. Molecular biology of the VEGF and the VEGF receptor family. Semin Thromb Hemost. 2000; 26:561–569.4. Kanazawa H, Hirata K, Yoshikawa J. Involvement of vascular endothelial growth factor in exercise induced bronchoconstriction in asthmatic patients. Thorax. 2002; 57:885–888.5. Hoshino M, Takahashi M, Aoike N. Expression of vascular endothelial growth factor, basic fibroblast growth factor, and angiogenin immunoreactivity in asthmatic airways and its relationship to angiogenesis. J Allergy Clin Immunol. 2001; 107:295–301.6. Asai K, Kanazawa H, Otani K, Shiraishi S, Hirata K, Yoshikawa J. Imbalance between vascular endothelial growth factor and endostatin levels in induced sputum from asthmatic subjects. J Allergy Clin Immunol. 2002; 110:571–575.7. Lee CG, Link H, Baluk P, Homer RJ, Chapoval S, Bhandari V, Kang MJ, Cohn L, Kim YK, McDonald DM, et al. Vascular endothelial growth factor (VEGF) induces remodeling and enhances TH2-mediated sensitization and inflammation in the lung. Nat Med. 2004; 10:1095–1103.8. Tseliou E, Bakakos P, Kostikas K, Hillas G, Mantzouranis K, Emmanouil P, Simoes D, Alchanatis M, Papiris S, Loukides S. Increased levels of angiopoietins 1 and 2 in sputum supernatant in severe refractory asthma. Allergy. 2012; 67:396–402.9. Soker S, Fidder H, Neufel G, Klagsbrun M. Characterization of novel vascular endothelial growth factor (VEGF) receptors on tumor cells that bind VEGF165 via its exon 7-encoded domain. J Biol Chem. 1996; 271:5761–5767.10. Soker S, Takashima S, Miao HQ, Neufeld G, Klagsbrun M. Neuropilin-1 is expressed by endothelial and tumor cells as an isoform-specific receptor for vascular endothelial growth factor. Cell. 1998; 92:735–745.11. Kolodkin AL, Levengood D, Rowe E, Tai YT, Giger RJ, Ginty DD. Neuropilin is a semaphorin III receptor. Cell. 1997; 90:753–762.12. He Z, Tessier-Lavigne M. Neuropilin is a receptor for the axonal chemorepellent semaphorin III. Cell. 1997; 90:739–751.13. Bachelder RE, Lipscomb EA, Lin X, Wendt MA, Chadborn NH, Eickholt BJ, Mercurio AM. Competing autocrine pathways involving alternative neuropilin-1 ligands regulate chemotaxis of carcinoma cells. Cancer Res. 2003; 63:5230–5233.14. Pan Q, Chanthery Y, Liang WC, Stawicki S, Mak J, Rathore N, Tong RK, Kowalski J, Yee SF, Pacheco G, et al. Blocking neuropilin-1 function has an additive effect with anti-VEGF to inhibit tumor growth. Cancer Cell. 2007; 11:53–67.15. Yamaguchi J, Nakamura F, Aihara M, Yamashita N, Usui H, Hida T, Takei K, Nagashima Y, Ikezawa Z, Goshima Y. Semaphorin 3A alleviates skin lesions and scratching behavior in NC/Nga mice, an atopic dermatitis model. J Invest Dermatol. 2008; 128:2842–2849.16. Koczy-Baron E, Jochem J, Kasperska-Zajac A. Increased plasma concentration of vascular endothelial growth factor in patients with atopic dermatitis and its relation to disease severity and platelet activation. Inflamm Res. 2012; 61:1405–1409.17. Haixia D, Jingsong Z, Lei J, Hairong D, Jun W, Hang X, Weixian C. Gene expression of neuropilin-1 and its receptors, VEGF/Semaphorin 3a, in normal and cancer cells. Cell Biochem Biophys. 2011; 59:39–47.18. Staton CA, Shaw LA, Valluru M, Hoh L, Koay I, Cross SS, Reed MW, Brown NJ. Expression of class 3 semaphorins and their receptors in human breast neoplasia. Histopathology. 2011; 59:274–282.19. US Department of Health and Human Services, National Institute of Health, National Heart, Lung, and Blood Institute. Expert Panel Report 3: guidelines for the diagnosis and management of asthma. Accessed 13 Feb 2013. Available at: http://www.nhlbi.nih.gov/guidelines/asthma.20. Ungar WJ, Champman KR, Santos MT. Assessment of a medication-based asthma index for population research. Am J Respir Crit Care Med. 2002; 165:190–194.21. Sohn SW, Lee HS, Park HW, Chang YS, Kim YK, Cho SH, Kim YY, Min KU. Evaluation of cytokine mRNA in induced sputum from patients with allergic rhinitis: relationship to airway hyperresponsiveness. Allergy. 2008; 63:268–273.22. Shim EJ, Chun E, Lee HS, Bang BR, Kim TW, Cho SH, Min KU, Park HW. The role of high-mobility group box-1 (HMGB1) in the pathogenesis of asthma. Clin Exp Allergy. 2012; 42:958–965.23. Brambilla E, Constantin B, Drabkin H, Roche J. Semaphorin. SEMA3F localization in malignant human lung and cell lines: a suggested role in cell adhesion and migration. Am J Pathol. 2000; 156:939–950.24. Catalano A, Caprari P, Rodilossi S, Betta P, Castellucci M, Casazza A, Tamagnone L, Procopio A. Cross-talk between vascular endothelial growth factor and semaphorin-3A pathway in the regulation of normal and malignant mesothelial cell proliferation. FASEB J. 2004; 18:358–360.25. Messersmith EK, Leonardo ED, Shatz CJ, Tessier-Lavigne M, Goodman CS, Kolodkin AL. Cross-talk between vascular endothelial growth factor and semaphorin-3A pathway in the regulation of normal and malignant mesothelial cell proliferation. Neuron. 1995; 14:949–959.26. Becker PM, Tran TS, Delannoy MJ, He C, Shannon JM, McGrath-Morrow S. Semaphorin 3A contributes to distal pulmonary epithelial cell differentiation and lung morphogenesis. PLoS One. 2011; 6:e27449.27. Ito T, Kagoshima M, Sasaki Y, Sasaki Y, Li C, Udaka N, Kitsukawa T, Fujisawa H, Taniguchi M, Yagi T, et al. Repulsive axon guidance molecule Sema3A inhibits branching morphogenesis of fetal mouse lung. Mech Dev. 2000; 97:35–45.28. Lepelletier Y, Moura IC, Hadj-Slimane R, Renand A, Fiorentino S, Baude C, Shirvan A, Barzilai A, Hermine O. Immunosuppressive role of semaphorin-3A on T cell proliferation is mediated by inhibition of actin cytoskeleton reorganization. Eur J Immunol. 2006; 36:1782–1793.29. Le A, Zielinski R, He C, Crow MT, Biswal S, Tuder RM, Becker PM. Pulmonary epithelial neuropilin-1 deletion enhances development of cigarette smoke-induced emphysema. Am J Respir Crit Care Med. 2009; 180:396–406.30. Davies DE. The bronchial epithelium in chronic and severe asthma. Curr Allergy Asthma Rep. 2001; 1:127–133.31. Roche J, Drabkin H, Brambilla E. Neuropilin and its ligands in normal lung and cancer. Adv Exp Med Biol. 2002; 515:103–114.32. Lee HS, Myers A, Kim J. Vascular endothelial growth factor drives autocrine epithelial cell proliferation and survival in chronic rhinosinusitis with nasal polyposis. Am J Respir Crit Care Med. 2009; 180:1056–1067.33. Lee HS, Kim J. Cigarette smoke inhibits nasal airway epithelial cell growth and survival. Int Forum Allergy Rhinol. 2013; 3:188–192.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Expression and Function of Plexin A1 in Rheumatoid Synoviocytes
- Treatment of Severe Asthma
- MicroRNA-143 and -145 modulate the phenotype of synovial fibroblasts in rheumatoid arthritis
- ERRATUM: Spelling Correction. Suppressive effects of long-term treatment with inhaled steroids on hypothalamic-pituitary-adrenal axis in asthma
- Anti-SEMA3A Antibody: A Novel Therapeutic Agent to Suppress Glioblastoma Tumor Growth