Clin Exp Otorhinolaryngol.
2012 Dec;5(4):222-227. 10.3342/ceo.2012.5.4.222.
The Effect of Nano-Silver on Allergic Rhinitis Model in Mice
- Affiliations
-
- 1Department of Otorhinolaryngology, Catholic University of Daegu School of Medicine, Daegu, Korea. hsseung@cu.ac.kr
- KMID: 1489083
- DOI: http://doi.org/10.3342/ceo.2012.5.4.222
Abstract
OBJECTIVES
Silver has long been known as a strong antimicrobial and disinfectant. Several types of nano-silver coated products have been developed. However, the antimicrobial and disinfectant characteristics of nano-silver have not been well studied. The aim of this study was to investigate the effect of nano-silver on allergic inflammation in a mouse model.
METHODS
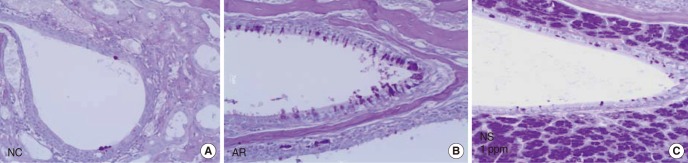
Female BALB/C mice were sensitized by intraperitoneal injection of ovalbumin (OVA) and aluminium hydroxide on days 0, 7, 14, and 21. Mice were challenged with intranasal instillation of OVA. Nano-silver was also administered nasally prior to intranasal instillation of OVA. Severity of allergic rhinitis was assessed according to nasal symptoms, serum OVA-specific IgE level, interleukin (IL)-4, IL-10, and interferon (INF)-gamma levels in nasal lavage fluid. Hematoxylin-eosin stain and periodic acid-Schiff stain were performed for evaluation of histological change.
RESULTS
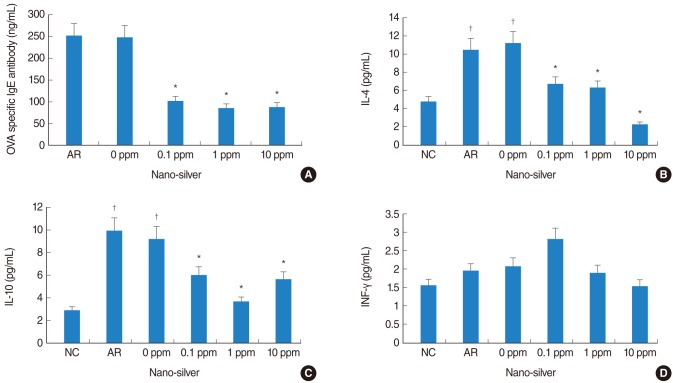
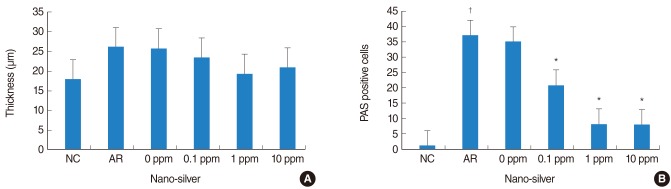
Nano-silver attenuated manifestation of nasal symptoms in sensitized mice and inhibited production of OVA-specific IgE, IL-4, and IL-10, however, it had no effect on INF-gamma level. In addition, the degree of inflammatory cell infiltration and goblet cell hyperplasia was attenuated by nano-silver.
CONCLUSION
These results suggest that nano-silver may effectively reduce allergic inflammation in a mouse model of allergic rhinitis. Through its properties as an anti-inflammatory agent, nano-silver may be a useful therapeutic strategy.
Keyword
MeSH Terms
-
Animals
Female
Goblet Cells
Humans
Hydroxides
Hyperplasia
Immunoglobulin E
Inflammation
Injections, Intraperitoneal
Interferons
Interleukin-10
Interleukin-4
Interleukins
Mice
Nasal Lavage Fluid
Ovalbumin
Ovum
Rhinitis
Rhinitis, Allergic, Perennial
Silver
Hydroxides
Immunoglobulin E
Interferons
Interleukin-10
Interleukin-4
Interleukins
Ovalbumin
Silver
Figure
Reference
-
1. Slawson RM, Lee H, Trevors JT. Bacterial interactions with silver. Biol Met. 1990; 3(3-4):151–154. PMID: 2073456.
Article2. Shvedova AA, Kisin ER, Mercer R, Murray AR, Johnson VJ, Potapovich AI, et al. Unusual inflammatory and fibrogenic pulmonary responses to single-walled carbon nanotubes in mice. Am J Physiol Lung Cell Mol Physiol. 2005; 11. 289(5):L698–L708. PMID: 15951334.
Article3. Magrez A, Kasas S, Salicio V, Pasquier N, Seo JW, Celio M, et al. Cellular toxicity of carbon-based nanomaterials. Nano Lett. 2006; 6. 6(6):1121–1125. PMID: 16771565.
Article4. Shin SH, Ye MK, Kim HS, Kang HS. The effects of nano-silver on the proliferation and cytokine expression by peripheral blood mononuclear cells. Int Immunopharmacol. 2007; 12. 7(13):1813–1818. PMID: 17996693.
Article5. Chen X, Schluesener HJ. Nanosilver: a nanoproduct in medical application. Toxicol Lett. 2008; 1. 176(1):1–12. PMID: 18022772.
Article6. Kim JH, Mun YJ, Ahn SH, Park JS, Woo WH. Induction of oral tolerance to Japanese cedar pollen. Arch Pharm Res. 2001; 12. 24(6):557–563. PMID: 11794535.
Article7. Murasugi T, Nakagami Y, Yoshitomi T, Hirahara K, Yamashita M, Taniguchi Y, et al. Oral administration of a T cell epitope inhibits symptoms and reactions of allergic rhinitis in Japanese cedar pollen allergen-sensitized mice. Eur J Pharmacol. 2005; 3. 510(1-2):143–148. PMID: 15740735.
Article8. Newacheck PW, Stoddard JJ. Prevalence and impact of multiple childhood chronic illnesses. J Pediatr. 1994; 1. 124(1):40–48. PMID: 7506774.
Article9. Chen HW, Su SF, Chien CT, Lin WH, Yu SL, Chou CC, et al. Titanium dioxide nanoparticles induce emphysema-like lung injury in mice. FASEB J. 2006; 11. 20(13):2393–2395. PMID: 17023518.
Article10. Xia T, Kovochich M, Brant J, Hotze M, Sempf J, Oberley T, et al. Comparison of the abilities of ambient and manufactured nanoparticles to induce cellular toxicity according to an oxidative stress paradigm. Nano Lett. 2006; 8. 6(8):1794–1807. PMID: 16895376.
Article11. Cyrys J, Stolzel M, Heinrich J, Kreyling WG, Menzel N, Wittmaack K, et al. Elemental composition and sources of fine and ultrafine ambient particles in Erfurt, Germany. Sci Total Environ. 2003; 4. 305(1-3):143–156. PMID: 12670764.
Article12. Nel A. Atmosphere, air pollution-related illness: effects of particles. Science. 2005; 5. 308(5723):804–806. PMID: 15879201.13. Brunner TJ, Wick P, Manser P, Spohn P, Grass RN, Limbach LK, et al. In vitro cytotoxicity of oxide nanoparticles: comparison to asbestos, silica, and the effect of particle solubility. Environ Sci Technol. 2006; 7. 40(14):4374–4381. PMID: 16903273.
Article14. Roh JY, Sim SJ, Yi J, Park K, Chung KH, Ryu DY, et al. Ecotoxicity of silver nanoparticles on the soil nematode Caenorhabditis elegans using functional ecotoxicogenomics. Environ Sci Technol. 2009; 5. 43(10):3933–3940. PMID: 19544910.
Article15. Feng QL, Wu J, Chen GQ, Cui FZ, Kim TN, Kim JO. A mechanistic study of the antibacterial effect of silver ions on Escherichia coli and Staphylococcus aureus. J Biomed Mater Res. 2000; 12. 52(4):662–668. PMID: 11033548.16. Park HS, Kim KH, Jang S, Park JW, Cha HR, Lee JE, et al. Attenuation of allergic airway inflammation and hyperresponsiveness in a murine model of asthma by silver nanoparticles. Int J Nanomedicine. 2010; 8. 5:505–515. PMID: 20957173.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Allergic Rhinitis Mouse Model
- The Inhibitory Effect of BCG on the Allergic Inflammation in House Dustmite-Induced Mouse
- The Effect of Interleukin-12 on the Immune Response in Allergic Rhinitis Mouse Model
- Principles and Application of Mouse Model of Allergic Rhinitis
- The Role of Aviation Medical Examiners in the Diagnosis, Treatment and Aeromedical Assessment of Patients with Allergic Rhinitis