Clin Orthop Surg.
2011 Dec;3(4):325-331. 10.4055/cios.2011.3.4.325.
The Effect of Platelet Rich Plasma from Bone Marrow Aspirate with Added Bone Morphogenetic Protein-2 on the Achilles Tendon-Bone Junction in Rabbits
- Affiliations
-
- 1Department of Orthopaedic Surgery, Korea University College of Medicine, Seoul, Korea. canall@korea.ac.kr
- 2Hanyang University School of Chemical and Biological Engineering, Seoul, Korea.
- 3Department of Bioengineering, Seoul National University, Seoul, Korea.
- KMID: 1097171
- DOI: http://doi.org/10.4055/cios.2011.3.4.325
Abstract
- BACKGROUND
To determine if exogenously injected bone marrow derived platelet-rich plasma (PRP) plus bone morphogenetic protein (BMP)-2 could accelerate the healing of bone-tendon junction injuries and increase the junction holding strength during the early regeneration period.
METHODS
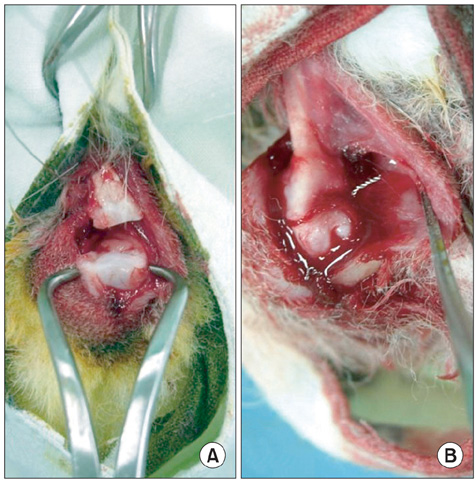
A direct injury model of the bone-tendon junction was made using an Achilles tendon-calcaneus bone junction in a rabbit. In the PRP/BMP-2/fibrin group, 0.05 mL of bone marrow derived PRP and 100 ng/mL of BMP-2 both incorporated into 0.1 mL of fibrin glue were injected into Achilles tendon-calcaneus bone junctions. The effect of the intervention was tested by comparing the results of an intervention group to a control group. The results of biomechanical testing, and histological and gross analyses were compared between the 2 groups at the following time points after surgery: 2 weeks, 4 weeks, and 8 weeks.
RESULTS
Histologic examinations showed that woven bone developed in tendon-bone junctions at 2 weeks after surgery in the PRP/BMP-2/fibrin group. Mechanical test results showed no significant difference between the PRP/BMP-2/fibrin and control groups at 2 and 4 weeks after surgery, but the mean maximal load in the PRP/BMP-2/fibrin group was significantly higher than in the control group (p < 0.05) at 8 weeks after surgery.
CONCLUSIONS
Bone marrow derived PRP and BMP-2 in fibrin glue accelerated healing in a rabbit model of tendon-bone junction injury.
Keyword
MeSH Terms
Figure
Cited by 2 articles
-
Enhanced Tendon-to-Bone Healing of Chronic Rotator Cuff Tears by Bone Marrow Aspirate Concentrate in a Rabbit Model
Xiao Ning Liu, Cheol-Jung Yang, Ji Eui Kim, Zhen Wu Du, Ming Ren, Wei Zhang, Hong Yu Zhao, Kyung Ok Kim, Kyu-Cheol Noh
Clin Orthop Surg. 2018;10(1):99-110. doi: 10.4055/cios.2018.10.1.99.The Effect of Platelet Rich Plasma Dosage on the Tendon Healing in Rabbits
Min-Wook Kim, On Lim, So-Min Hwang, Min-Kyu Hwang, Jong-Seo Lee
J Korean Soc Surg Hand. 2016;21(4):189-197. doi: 10.12790/jkssh.2016.21.4.189.
Reference
-
1. Thomopoulos S, Williams GR, Gimbel JA, Favata M, Soslowsky LJ. Variation of biomechanical, structural, and compositional properties along the tendon to bone insertion site. J Orthop Res. 2003. 21(3):413–419.
Article2. Benjamin M, Evans EJ, Copp L. The histology of tendon attachments to bone in man. J Anat. 1986. 149:89–100.3. St Pierre P, Olson EJ, Elliott JJ, O'Hair KC, McKinney LA, Ryan J. Tendon-healing to cortical bone compared with healing to a cancellous trough: a biomechanical and histological evaluation in goats. J Bone Joint Surg Am. 1995. 77(12):1858–1866.
Article4. Ward JJ, Meyer RD, Lemons JE. Tensile strength comparison of dowel plug technique to standard techniques of tendonbone attachment. Foot Ankle. 1988. 8(5):248–253.
Article5. Wong MW, Qin L, Lee KM, et al. Healing of bone-tendon junction in a bone trough: a goat partial patellectomy model. Clin Orthop Relat Res. 2003. (413):291–302.6. Kim HJ. The Healing effect of bone morphogenetic protein with fibrin glue on an injury of the tendon-bone junction [dissertation]. 2006. Seoul: Korea University.7. Kobayashi K, Healey RM, Sah RL, et al. Novel method for the quantitative assessment of cell migration: a study on the motility of rabbit anterior cruciate (ACL) and medial collateral ligament (MCL) cells. Tissue Eng. 2000. 6(1):29–38.
Article8. Ouyang HW, Goh JC, Thambyah A, Teoh SH, Lee EH. Knitted poly-lactide-co-glycolide scaff old loaded with bone marrow stromal cells in repair and regeneration of rabbit Achilles tendon. Tissue Eng. 2003. 9(3):431–439.
Article9. Wang CJ, Huang HY, Pai CH. Shock wave-enhanced neovascularization at the tendon-bone junction: an experiment in dogs. J Foot Ankle Surg. 2002. 41(1):16–22.
Article10. Wong MW, Qin L, Tai JK, Lee SK, Leung KS, Chan KM. Engineered allogeneic chondrocyte pellet for reconstruction of fibrocartilage zone at bone-tendon junction: a preliminary histological observation. J Biomed Mater Res B Appl Biomater. 2004. 70(2):362–367.11. Hashimoto Y, Yoshida G, Toyoda H, Takaoka K. Generation of tendon-to-bone interface "enthesis" with use of recombinant BMP-2 in a rabbit model. J Orthop Res. 2007. 25(11):1415–1424.
Article12. Arpornmaeklong P, Kochel M, Depprich R, Kubler NR, Wurzler KK. Influence of platelet-rich plasma (PRP) on osteogenic differentiation of rat bone marrow stromal cells: an in vitro study. Int J Oral Maxillofac Surg. 2004. 33(1):60–70.
Article13. Marx RE, Carlson ER, Eichstaedt RM, Schimmele SR, Strauss JE, Georgeff KR. Platelet-rich plasma: growth factor enhancement for bone grafts. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1998. 85(6):638–646.14. Mazor Z, Peleg M, Garg AK, Luboshitz J. Platelet-rich plasma for bone graft enhancement in sinus floor augmentation with simultaneous implant placement: patient series study. Implant Dent. 2004. 13(1):65–72.
Article15. Ogino Y, Ayukawa Y, Tsukiyama Y, Koyano K. The effect of platelet-rich plasma on the cellular response of rat bone marrow cells in vitro. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005. 100(3):302–307.
Article16. Tomoyasu A, Higashio K, Kanomata K, et al. Platelet-rich plasma stimulates osteoblastic differentiation in the presence of BMPs. Biochem Biophys Res Commun. 2007. 361(1):62–67.
Article17. de Mos M, van der Windt AE, Jahr H, et al. Can plateletrich plasma enhance tendon repair? A cell culture study. Am J Sports Med. 2008. 36(6):1171–1178.18. Nishimoto S, Oyama T, Matsuda K. Simultaneous concentration of platelets and marrow cells: a simple and useful technique to obtain source cells and growth factors for regenerative medicine. Wound Repair Regen. 2007. 15(1):156–162.
Article19. Forslund C, Aspenberg P. Improved healing of transected rabbit Achilles tendon after a single injection of cartilage-derived morphogenetic protein-2. Am J Sports Med. 2003. 31(4):555–559.
Article20. Jones CM, Dale L, Hogan BL, Wright CV, Smith JC. Bone morphogenetic protein-4 (BMP-4) acts during gastrula stages to cause ventralization of Xenopus embryos. Development. 1996. 122(5):1545–1554.
Article21. Wozney JM, Rosen V. Bone morphogenetic protein and bone morphogenetic protein gene family in bone formation and repair. Clin Orthop Relat Res. 1998. (346):26–37.
Article22. Rodeo SA, Arnoczky SP, Torzilli PA, Hidaka C, Warren RF. Tendon-healing in a bone tunnel: a biomechanical and histological study in the dog. J Bone Joint Surg Am. 1993. 75(12):1795–1803.
Article23. Lu H, Qin L, Lee K, Wong W, Chan K, Leung K. Healing compared between bone to tendon and cartilage to tendon in a partial inferior patellectomy model in rabbits. Clin J Sport Med. 2008. 18(1):62–69.
Article24. Cooper RR, Misol S. Tendon and ligament insertion: a light and electron microscopic study. J Bone Joint Surg Am. 1970. 52(1):1–20.25. Woo SL, Orlando CA, Gomez MA, Frank CB, Akeson WH. Tensile properties of the medial collateral ligament as a function of age. J Orthop Res. 1986. 4(2):133–141.
Article26. Aspenberg P, Forslund C. Bone morphogenetic proteins and tendon repair. Scand J Med Sci Sports. 2000. 10(6):372–375.
Article27. Chong AK, Ang AD, Goh JC, et al. Bone marrow-derived mesenchymal stem cells influence early tendon-healing in a rabbit achilles tendon model. J Bone Joint Surg Am. 2007. 89(1):74–81.
Article28. Rodeo SA, Suzuki K, Deng XH, Wozney J, Warren RF. Use of recombinant human bone morphogenetic protein-2 to enhance tendon healing in a bone tunnel. Am J Sports Med. 1999. 27(4):476–488.
Article29. Jackson MR, MacPhee MJ, Drohan WN, Alving BM. Fibrin sealant: current and potential clinical applications. Blood Coagul Fibrinolysis. 1996. 7(8):737–746.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effect of Platelet Rich Plasma Dosage on the Tendon Healing in Rabbits
- Effect of platelet-rich plasma in Achilles tendon allograft in rabbits
- The Healing Effectof Bone Morphogenic Protein with Fibrin Glue on an Injury of the Tendon-Bone Junction
- Enhanced Tendon-to-Bone Healing of Chronic Rotator Cuff Tears by Bone Marrow Aspirate Concentrate in a Rabbit Model
- The Effect of PRF and PRP for New Bone Formation of beta-TCP in Skull of White Rabbit