Korean Circ J.
2020 Mar;50(3):236-247. 10.4070/kcj.2019.0195.
Novel Associations between Related Proteins and Cellular Effects of High-Density Lipoprotein
- Affiliations
-
- 1Cardiovascular Research Institute, Yonsei University College of Medicine, Seoul, Korea. shl1106@yuhs.ac
- 2Center for Theragnosis, Biomedical Research Institute, Korean Institute of Science and Technology, Seoul, Korea. jelee9137@kist.re.kr
- 3Department of Biochemistry, College of Life Science and Biotechnology, Yonsei University, Seoul, Korea.
- 4Department of Chemistry, Sookmyung Women's University, Seoul, Korea.
- 5Proteometech Inc., Seoul, Korea.
- 6Division of Cardiology, Department of Internal Medicine, Severance Hospital, Yonsei University College of Medicine, Seoul, Korea.
- 7KHU-KIST Department of Converging Science and Technology, Kyung Hee University, Seoul, Korea.
- 8Division of Bio-Medical Science & Technology, KIST School, Korea University of Science and Technology, Seoul, Korea.
- KMID: 2470908
- DOI: http://doi.org/10.4070/kcj.2019.0195
Abstract
- BACKGROUND AND OBJECTIVES
Recent studies have examined the structure-function relationship of high-density lipoprotein (HDL). This study aimed to identify and rank HDL-associated proteins involved in several biological function of HDL.
METHODS
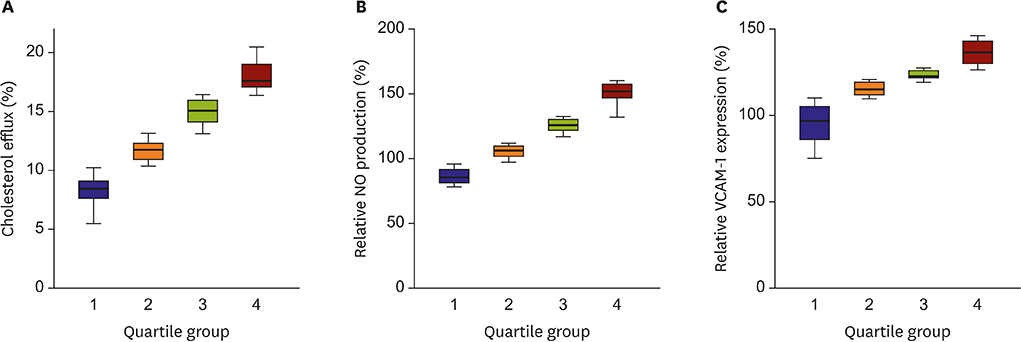
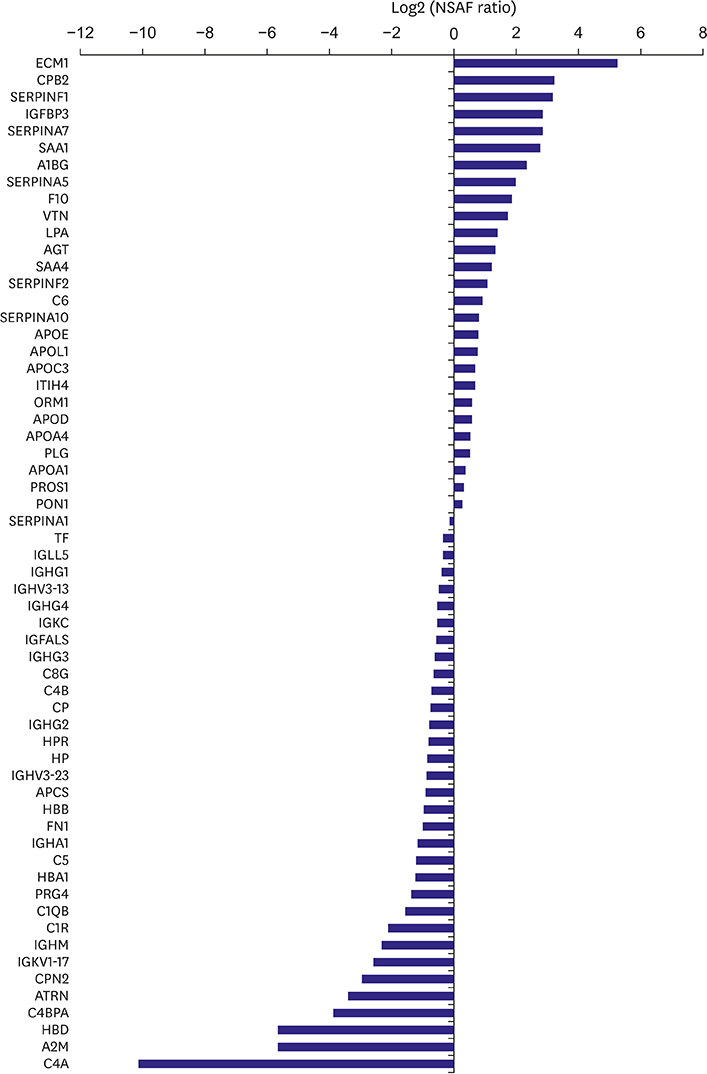
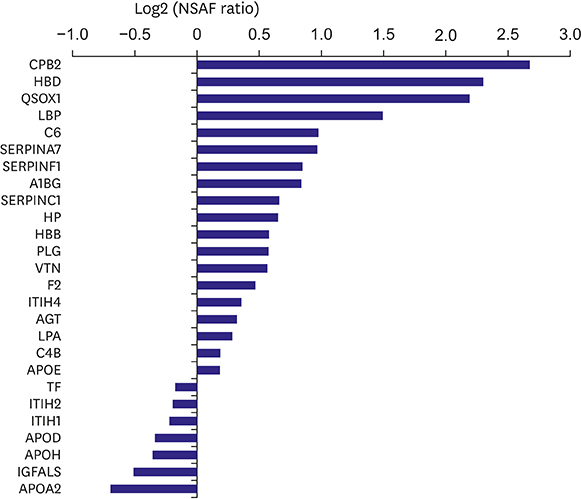
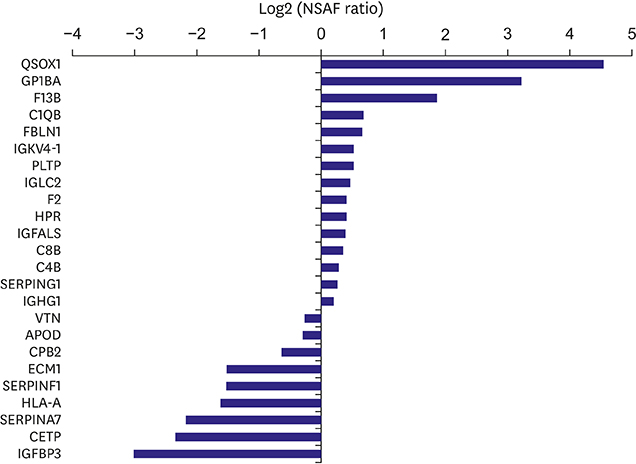
HDLs isolated from 48 participants were analyzed. Cholesterol efflux capacity, effect of HDL on nitric oxide production, and vascular cell adhesion molecule-1 expression were assessed. The relative abundance of identified proteins in the highest vs. lowest quartile was expressed using the normalized spectral abundance factor ratio.
RESULTS
After adjustment by multiple testing, six proteins, thyroxine-binding globulin, alpha-1B-glycoprotein, plasma serine protease inhibitor, vitronectin, angiotensinogen, and serum amyloid A-4, were more abundant (relative abundance ratio ≥2) in HDLs with the highest cholesterol efflux capacity. In contrast, three proteins, complement C4-A, alpha-2-macroglobulin, and immunoglobulin mu chain C region, were less abundant (relative abundance ratio <0.5). In terms of nitric oxide production and vascular cell adhesion molecule-1 expression, no proteins showed abundance ratios ≥2 or <0.5 after adjustment. Proteins correlated with the functional parameters of HDL belonged to diverse biological categories.
CONCLUSIONS
In summary, this study ranked proteins showing higher or lower abundance in HDLs with high functional capacities and newly identified multiple proteins linked to cholesterol efflux capacity.
MeSH Terms
-
Amyloid
Angiotensinogen
Atherosclerosis
Cardiovascular Diseases
Cholesterol
Complement System Proteins
Immunoglobulin mu-Chains
Lipoproteins*
Nitric Oxide
Plasma
Proteomics
Serine Proteases
Thyroxine-Binding Globulin
Vascular Cell Adhesion Molecule-1
Vitronectin
Amyloid
Angiotensinogen
Cholesterol
Complement System Proteins
Immunoglobulin mu-Chains
Lipoproteins
Nitric Oxide
Serine Proteases
Thyroxine-Binding Globulin
Vascular Cell Adhesion Molecule-1
Vitronectin
Figure
Cited by 1 articles
-
The Long and Winding Road: To the Proper Understanding of High-density Lipoprotein
Jidong Sung
Korean Circ J. 2020;50(3):248-249. doi: 10.4070/kcj.2020.0020.
Reference
-
1. Gordon SM, Hofmann S, Askew DS, Davidson WS. High density lipoprotein: it's not just about lipid transport anymore. Trends Endocrinol Metab. 2011; 22:9–15.
Article2. Vaisar T. Proteomics investigations of HDL: challenges and promise. Curr Vasc Pharmacol. 2012; 10:410–421.
Article3. Riwanto M, Rohrer L, Roschitzki B, et al. Altered activation of endothelial anti- and proapoptotic pathways by high-density lipoprotein from patients with coronary artery disease: role of high-density lipoprotein-proteome remodeling. Circulation. 2013; 127:891–904.4. Kopecky C, Haidinger M, Birner-Grünberger R, et al. Restoration of renal function does not correct impairment of uremic HDL properties. J Am Soc Nephrol. 2015; 26:565–575.
Article5. Darabi M, Guillas-Baudouin I, Le Goff W, Chapman MJ, Kontush A. Therapeutic applications of reconstituted HDL: when structure meets function. Pharmacol Ther. 2016; 157:28–42.
Article6. Davidson WS, Silva RA, Chantepie S, Lagor WR, Chapman MJ, Kontush A. Proteomic analysis of defined HDL subpopulations reveals particle-specific protein clusters: relevance to antioxidative function. Arterioscler Thromb Vasc Biol. 2009; 29:870–876.7. Besler C, Heinrich K, Rohrer L, et al. Mechanisms underlying adverse effects of HDL on eNOS-activating pathways in patients with coronary artery disease. J Clin Invest. 2011; 121:2693–2708.
Article8. Rosenson RS, Brewer HB Jr, Ansell B, et al. Translation of high-density lipoprotein function into clinical practice: current prospects and future challenges. Circulation. 2013; 128:1256–1267.9. Gordon SM, Chung JH, Playford MP, et al. High density lipoprotein proteome is associated with cardiovascular risk factors and atherosclerosis burden as evaluated by coronary CT angiography. Atherosclerosis. 2018; 278:278–285.
Article10. Pamir N, Hutchins PM, Ronsein GE, et al. Plasminogen promotes cholesterol efflux by the ABCA1 pathway. JCI Insight. 2017; 2:e92176.
Article11. Luo M, Liu A, Wang S, et al. ApoCIII enrichment in HDL impairs HDL-mediated cholesterol efflux capacity. Sci Rep. 2017; 7:2312.
Article12. Rhainds D, Tardif JC. From HDL-cholesterol to HDL-function: cholesterol efflux capacity determinants. Curr Opin Lipidol. 2019; 30:101–107.13. Zhou A, Huntington JA, Pannu NS, Carrell RW, Read RJ. How vitronectin binds PAI-1 to modulate fibrinolysis and cell migration. Nat Struct Biol. 2003; 10:541–544.
Article14. Singh B, Su YC, Riesbeck K. Vitronectin in bacterial pathogenesis: a host protein used in complement escape and cellular invasion. Mol Microbiol. 2010; 78:545–560.
Article15. Vaisar T, Pennathur S, Green PS, et al. Shotgun proteomics implicates protease inhibition and complement activation in the antiinflammatory properties of HDL. J Clin Invest. 2007; 117:746–756.
Article16. Stonik JA, Remaley AT, Demosky SJ, Neufeld EB, Bocharov A, Brewer HB. Serum amyloid A promotes ABCA1-dependent and ABCA1-independent lipid efflux from cells. Biochem Biophys Res Commun. 2004; 321:936–941.
Article17. Abe-Dohmae S, Kato KH, Kumon Y, et al. Serum amyloid A generates high density lipoprotein with cellular lipid in an ABCA1- or ABCA7-dependent manner. J Lipid Res. 2006; 47:1542–1550.
Article18. van der Westhuyzen DR, Cai L, de Beer MC, de Beer FC. Serum amyloid A promotes cholesterol efflux mediated by scavenger receptor B-I. J Biol Chem. 2005; 280:35890–35895.
Article19. Han CY, Tang C, Guevara ME, et al. Serum amyloid A impairs the antiinflammatory properties of HDL. J Clin Invest. 2016; 126:266–281.
Article20. Ceda GP, Dall'Aglio E, Magnacavallo A, et al. The insulin-like growth factor axis and plasma lipid levels in the elderly. J Clin Endocrinol Metab. 1998; 83:499–502.
Article21. Lam CS, Chen MH, Lacey SM, et al. Circulating insulin-like growth factor-1 and its binding protein-3: metabolic and genetic correlates in the community. Arterioscler Thromb Vasc Biol. 2010; 30:1479–1484.22. Min HK, Maruyama H, Jang BK, et al. Suppression of IGF binding protein-3 by palmitate promotes hepatic inflammatory responses. FASEB J. 2016; 30:4071–4082.
Article23. Zhang Q, Steinle JJ. IGFBP-3 inhibits TNF-á production and TNFR-2 signaling to protect against retinal endothelial cell apoptosis. Microvasc Res. 2014; 95:76–81.
Article24. Kawachi S, Takeda N, Sasaki A, et al. Circulating insulin-like growth factor-1 and insulin-like growth factor binding protein-3 are associated with early carotid atherosclerosis. Arterioscler Thromb Vasc Biol. 2005; 25:617–621.
Article25. Craword SE, Fitchev P, Veliceasa D, Volpert OV. The many facets of PEDF in drug discovery and disease: a diamond in the rough or split personality disorder? Expert Opin Drug Discov. 2013; 8:769–792.
Article26. Tahara N, Yamagishi S, Tahara A, et al. Serum level of pigment epithelium-derived factor is a marker of atherosclerosis in humans. Atherosclerosis. 2011; 219:311–315.
Article27. Yamagishi S, Nakamura K, Ueda S, Kato S, Imaizumi T. Pigment epithelium-derived factor (PEDF) blocks angiotensin II signaling in endothelial cells via suppression of NADPH oxidase: a novel anti-oxidative mechanism of PEDF. Cell Tissue Res. 2005; 320:437–445.
Article28. Khan AA, Mundra PA, Straznicky NE, et al. Weight loss and exercise alter the high-density lipoprotein lipidome and improve high-density lipoprotein functionality in metabolic syndrome. Arterioscler Thromb Vasc Biol. 2018; 38:438–447.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Correlation between the Triglyceride to High Density Lipoprotein Cholesterol Ratio and Computed Tomography-Measured Visceral Fat and Cardiovascular Disease Risk Factors in Local Adult Male Subjects
- Hypolipidemic Effects and Safety of Lovastatin in Patients with Primary Hypercholesterolemia
- Changes in the Serum Level of High Density Lipoprotein-cholesterol after Smoking Cessation among Adult Men
- Associations between Oxidized LDL to LDL Ratio, HDL and Vascular Calcification in the Feet of Hemodialysis Patients
- Monocytes and High-density Lipoprotein Cholesterol in Branch Retinal Vein Occlusion