J Periodontal Implant Sci.
2019 Dec;49(6):366-381. 10.5051/jpis.2019.49.6.366.
Bioactive characteristics of an implant surface coated with a pH buffering agent: an in vitro study
- Affiliations
-
- 1Department of Periodontology, Research Institute for Periodontal Regeneration, Yonsei University College of Dentistry, Seoul, Korea. shchoi726@yuhs.ac
- 2Implant R&D Center, Osstem Implant Co., Ltd., Busan, Korea.
- KMID: 2465381
- DOI: http://doi.org/10.5051/jpis.2019.49.6.366
Abstract
- PURPOSE
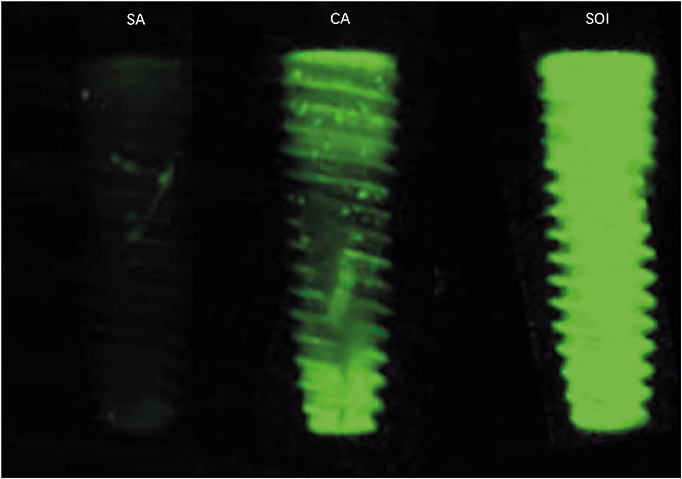
The purpose of this study was to evaluate the effectiveness of conventional sandblasted, large-grit, acid-etched (SLA) surface coated with a pH buffering solution based on surface wettability, blood protein adhesion, osteoblast affinity, and platelet adhesion and activation.
METHODS
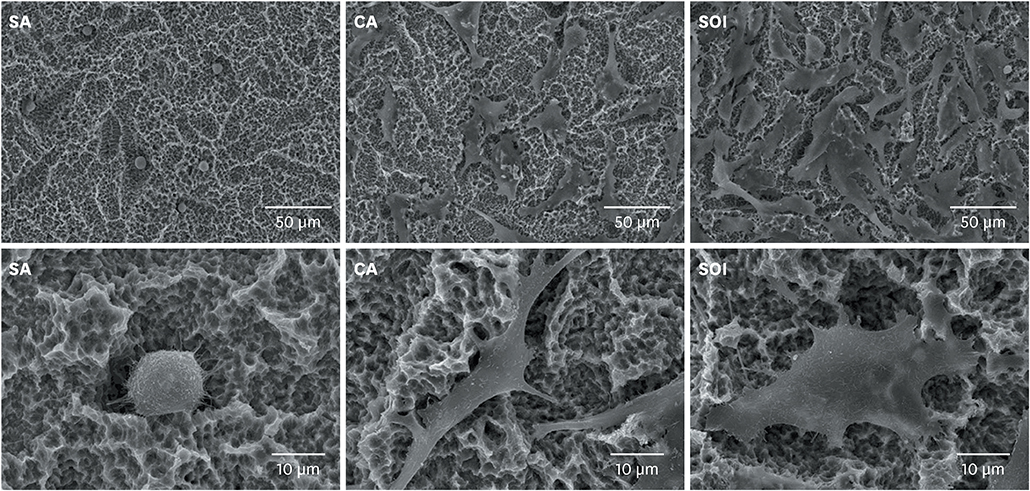
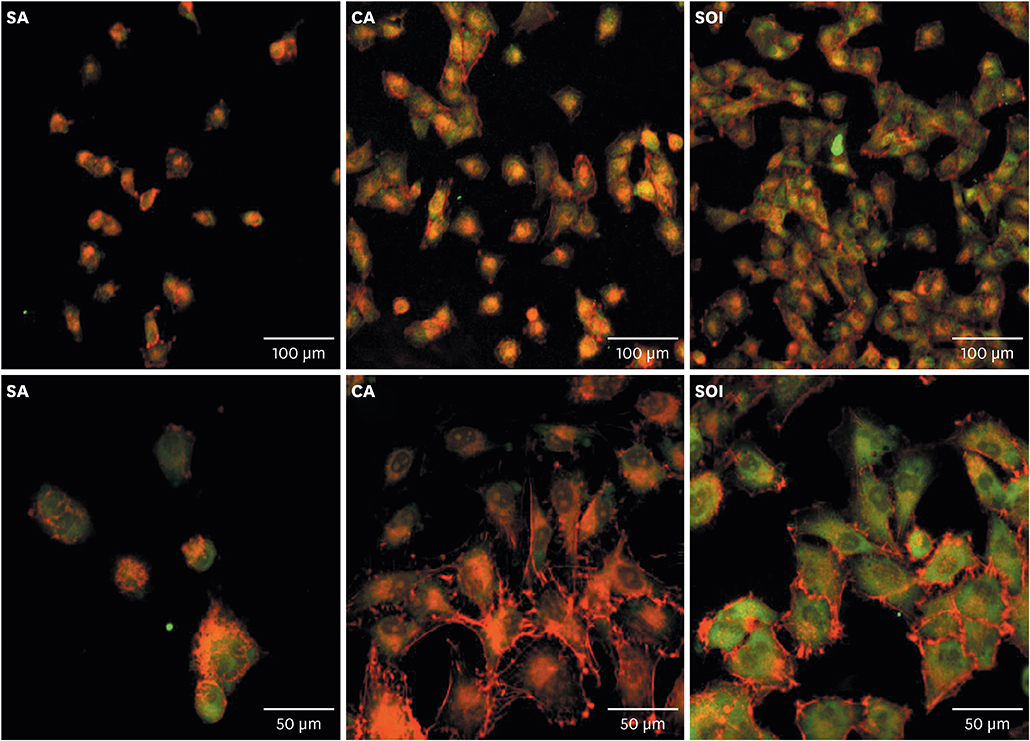
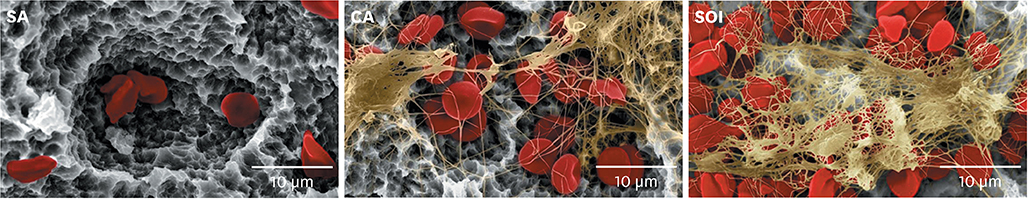
Titanium discs and implants with conventional SLA surface (SA), SLA surface in an aqueous calcium chloride solution (CA), and SLA surface with a pH buffering agent (SOI) were prepared. The wetting velocity was measured by the number of threads wetted by blood over an interval of time. Serum albumin adsorption was tested using the bicinchoninic acid assay and by measuring fluorescence intensity. Osteoblast activity assays (osteoblast adhesion, proliferation, differentiation, mineralization, and migration) were also performed, and platelet adhesion and activation assays were conducted.
RESULTS
In both the wetting velocity test and the serum albumin adsorption assay, the SOI surface displayed a significantly higher wetting velocity than the SA surface (P=0.000 and P=0.000, respectively). In the osteoblast adhesion, proliferation, differentiation, and mineralization tests, the mean values for SOI were all higher than those for SA and CA. On the osteoblast migration, platelet adhesion, and activation tests, SOI also showed significantly higher values than SA (P=0.040, P=0.000, and P=0.000, respectively).
CONCLUSIONS
SOI exhibited higher hydrophilicity and affinity for proteins, cells, and platelets than SA. Within the limits of this study, it may be concluded that coating an implant with a pH buffering agent can induce the attachment of platelets, proteins, and cells to the implant surface. Further studies should be conducted to directly compare SOI with other conventional surfaces with regard to its safety and effectiveness in clinical settings.
MeSH Terms
-
Adsorption
Blood Platelets
Calcium Chloride
Coated Materials, Biocompatible
Dental Implants
Fluorescence
Hydrogen-Ion Concentration*
Hydrophobic and Hydrophilic Interactions
Immunoassay
In Vitro Techniques*
Miners
Osteoblasts
Serum Albumin
Surface Properties
Titanium
Wettability
Calcium Chloride
Coated Materials, Biocompatible
Dental Implants
Serum Albumin
Titanium
Figure
Reference
-
1. Massaro C, Rotolo P, De Riccardis F, Milella E, Napoli A, Wieland M, et al. Comparative investigation of the surface properties of commercial titanium dental implants. Part I: chemical composition. J Mater Sci Mater Med. 2002; 13:535–548.2. Albrektsson T, Wennerberg A. Oral implant surfaces: Part 2--review focusing on clinical knowledge of different surfaces. Int J Prosthodont. 2004; 17:544–564.3. Esposito M, Coulthard P, Thomsen P, Worthington HV. The role of implant surface modifications, shape and material on the success of osseointegrated dental implants. A Cochrane systematic review. Eur J Prosthodont Restor Dent. 2005; 13:15–31.4. Puleo DA, Thomas MV. Implant surfaces. Dent Clin North Am. 2006; 50:323–338. v
Article5. Le Guéhennec L, Soueidan A, Layrolle P, Amouriq Y. Surface treatments of titanium dental implants for rapid osseointegration. Dent Mater. 2007; 23:844–854.
Article6. Morton D, Bornstein MM, Wittneben JG, Martin WC, Ruskin JD, Hart CN, et al. Early loading after 21 days of healing of nonsubmerged titanium implants with a chemically modified sandblasted and acid-etched surface: two-year results of a prospective two-center study. Clin Implant Dent Relat Res. 2010; 12:9–17.
Article7. Li D, Ferguson SJ, Beutler T, Cochran DL, Sittig C, Hirt HP, et al. Biomechanical comparison of the sandblasted and acid-etched and the machined and acid-etched titanium surface for dental implants. J Biomed Mater Res. 2002; 60:325–332.
Article8. Lang NP, Salvi GE, Huynh-Ba G, Ivanovski S, Donos N, Bosshardt DD. Early osseointegration to hydrophilic and hydrophobic implant surfaces in humans. Clin Oral Implants Res. 2011; 22:349–356.
Article9. Gittens RA, Scheideler L, Rupp F, Hyzy SL, Geis-Gerstorfer J, Schwartz Z, et al. A review on the wettability of dental implant surfaces II: biological and clinical aspects. Acta Biomater. 2014; 10:2907–2918.
Article10. Buser D, Broggini N, Wieland M, Schenk RK, Denzer AJ, Cochran DL, et al. Enhanced bone apposition to a chemically modified SLA titanium surface. J Dent Res. 2004; 83:529–533.
Article11. Scarano A, Piattelli A, Quaranta A, Lorusso F. Bone response to two dental implants with different sandblasted/acid-etched implant surfaces: a histological and histomorphometrical study in rabbits. BioMed Res Int. 2017; 2017:8724951.
Article12. Soares PB, Moura CC, da Rocha Júnior HA, Dechichi P, Zanetta-Barbosa D. Biological characterization of implant surfaces - in vitro study. Rev Odontol UNESP. 2015; 44:195–199.13. Martin JY, Schwartz Z, Hummert TW, Schraub DM, Simpson J, Lankford J Jr, et al. Effect of titanium surface roughness on proliferation, differentiation, and protein synthesis of human osteoblast-like cells (MG63). J Biomed Mater Res. 1995; 29:389–401.
Article14. Eriksson C, Nygren H, Ohlson K. Implantation of hydrophilic and hydrophobic titanium discs in rat tibia: cellular reactions on the surfaces during the first 3 weeks in bone. Biomaterials. 2004; 25:4759–4766.
Article15. Hong J, Kurt S, Thor A. A hydrophilic dental implant surface exhibits thrombogenic properties in vitro . Clin Implant Dent Relat Res. 2013; 15:105–112.
Article16. Di Iorio D, Traini T, Degidi M, Caputi S, Neugebauer J, Piattelli A. Quantitative evaluation of the fibrin clot extension on different implant surfaces: an in vitro study. J Biomed Mater Res B Appl Biomater. 2005; 74:636–642.17. Kohn DH, Sarmadi M, Helman JI, Krebsbach PH. Effects of pH on human bone marrow stromal cells in vitro: implications for tissue engineering of bone. J Biomed Mater Res. 2002; 60:292–299.
Article18. Arnett TR. Extracellular pH regulates bone cell function. J Nutr. 2008; 138:415S–8S.
Article19. Kaysinger KK, Ramp WK. Extracellular pH modulates the activity of cultured human osteoblasts. J Cell Biochem. 1998; 68:83–89.
Article20. Marumo M, Suehiro A, Kakishita E, Groschner K, Wakabayashi I. Extracellular pH affects platelet aggregation associated with modulation of store-operated Ca2+ entry. Thromb Res. 2001; 104:353–360.
Article21. Engvall E. Enzyme immunoassay ELISA and EMIT. Methods Enzymol. 1980; 70:419–439.22. Jung K, Pergande M. Influence of inorganic phosphate on the activity determination of isoenzymes of alkaline phosphatase in various buffer systems. Clin Chim Acta. 1980; 102:215–219.
Article23. Harada M, Hiraoka BY, Fukasawa K, Fukasawa KM. Purification and properties of bovine dental-pulp alkaline-phosphatase. Arch Oral Biol. 1982; 27:69–74.
Article24. Jones JV, Mansour M, James H, Sadi D, Carr RI. A substrate amplification system for enzyme-linked immunoassays. II. Demonstration of its applicability for measuring anti-DNA antibodies. J Immunol Methods. 1989; 118:79–84.25. Rausch-fan X, Qu Z, Wieland M, Matejka M, Schedle A. Differentiation and cytokine synthesis of human alveolar osteoblasts compared to osteoblast-like cells (MG63) in response to titanium surfaces. Dent Mater. 2008; 24:102–110.
Article26. Valarmathi MT, Yost MJ, Goodwin RL, Potts JD. The influence of proepicardial cells on the osteogenic potential of marrow stromal cells in a three-dimensional tubular scaffold. Biomaterials. 2008; 29:2203–2216.
Article27. Sudo H, Kodama HA, Amagai Y, Yamamoto S, Kasai S. In vitro differentiation and calcification in a new clonal osteogenic cell line derived from newborn mouse calvaria. J Cell Biol. 1983; 96:191–198.
Article28. Gregory CA, Gunn WG, Peister A, Prockop DJ. An Alizarin red-based assay of mineralization by adherent cells in culture: comparison with cetylpyridinium chloride extraction. Anal Biochem. 2004; 329:77–84.
Article29. Malladi P, Xu Y, Chiou M, Giaccia AJ, Longaker MT. Effect of reduced oxygen tension on chondrogenesis and osteogenesis in adipose-derived mesenchymal cells. Am J Physiol Cell Physiol. 2006; 290:C1139–46.
Article30. Kanthan SR, Kavitha G, Addi S, Choon DS, Kamarul T. Platelet-rich plasma (PRP) enhances bone healing in non-united critical-sized defects: a preliminary study involving rabbit models. Injury. 2011; 42:782–789.
Article31. Tamada Y, Kulik EA, Ikada Y. Simple method for platelet counting. Biomaterials. 1995; 16:259–261.
Article32. Grunkemeier JM, Tsai WB, Horbett TA. Hemocompatibility of treated polystyrene substrates: contact activation, platelet adhesion, and procoagulant activity of adherent platelets. J Biomed Mater Res. 1998; 41:657–670.
Article33. Park JY, Davies JE. Red blood cell and platelet interactions with titanium implant surfaces. Clin Oral Implants Res. 2000; 11:530–539.
Article34. Klein MO, Grötz KA, Walter C, Wegener J, Wagner W, Al-Nawas B. Functional rehabilitation of mandibular continuity defects using autologous bone and dental implants - prognostic value of bone origin, radiation therapy and implant dimensions. Eur Surg Res. 2009; 43:269–275.
Article35. Justus CR, Leffler N, Ruiz-Echevarria M, Yang LV. In vitro cell migration and invasion assays. J Vis Exp. 2014; 51046.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Influence of Implant Surface Coated with pH Buffering Agent on Early Osseointegration
- The Effects of Citric Acid on HA coated Implant Surface
- CLINICAL STUDY OF ENDOSSEOUS HYDROXYAPATITE COATED IMPLANTS
- Effects of surface coating on the screw release of dental implant screw
- The effect of Ca-P coatings of anodized implant surface on response of osteoblast-like cells in vitro