Ann Dermatol.
2019 Oct;31(5):530-537. 10.5021/ad.2019.31.5.530.
RE-ORGA, a Korean Herb Extract, Can Prevent Hair Loss Induced by Dihydrotestosterone in Human Dermal Papilla Cells
- Affiliations
-
- 1Department of Predictive Toxicology, Korea Institute of Toxicology, Daejeon, Korea. sjyoon@kitox.re.kr
- 2Research Institute, Wooridul Huebrain Ltd., Seoul, Korea. bskwon9@gmail.com
- 3Research Institute, WINNOVA Co., Ltd., Seoul, Korea.
- KMID: 2456230
- DOI: http://doi.org/10.5021/ad.2019.31.5.530
Abstract
- BACKGROUND
Androgenic alopecia (AGA) is the most common type of hair loss. It is likely inherited genetically and is promoted by dihydrotestosterone. 5α-reductase has been proven a good target through finasteride use. However, the pathogenesis of AGA cannot be fully explained based only on dihydrotestosterone levels.
OBJECTIVE
To identify similar hairloss inhibition activity of RE-ORGA with mode of action other than finasteride.
METHODS
We prepared RE-ORGA from Korean herb mixtures. We performed MTT assays for cytotoxicity, Cell Counting Kit-8 assays for cell proliferation, and western blot to identify expression levels of 5α-reductase and Bax. RNA-sequencing was performed for the expression patterns of genes in dihydrotestosterone-activated pathways. Anti-inflammatory activity was also assessed by the expression levels of tumor necrosis factor-alpha (TNF-α) and interleukin 6.
RESULTS
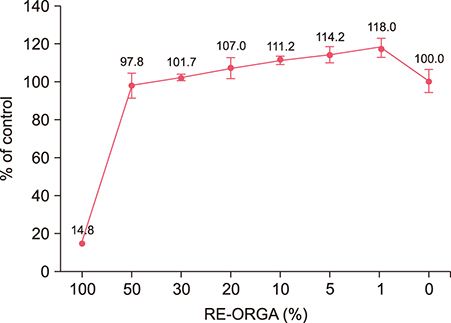
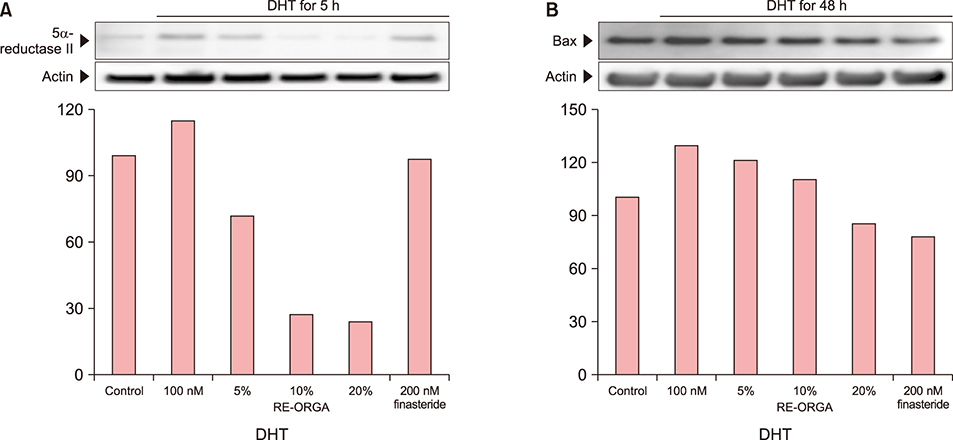
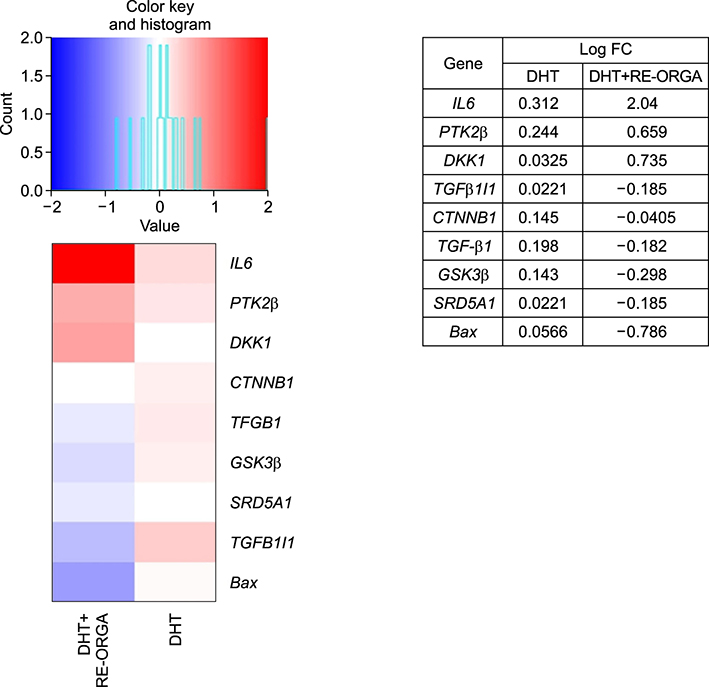
REORGA could promote the proliferation of human dermal papilla cells and showed low cytotoxicity. It also inhibited the expression of 5α-reductases and Bax in the cells. RNA-sequencing results verified that the mRNA expressions of SRD5A1, Bax, transforming growth factor-beta 1 (TGF-β1), and TGF-β1 induced transcript 1 (TGFβ1I1) were decreased, whereas expression of protein tyrosine kinase 2 beta (PTK2β) was more elevated. REORGA also showed anti-inflammatory activity through decreased mRNA levels of TNF-α.
CONCLUSION
Transcriptionally, up-regulation of PTK2β and concomitant down-regulation of TGFβ1I1 imply that RE-ORGA can modulate androgen receptor sensitivity, decreasing the expression of 5α-reductase type II and Bax together with TGF-β1 transcripts; RE-ORGA also showed partial anti-inflammatory activity. Overall, RE-ORGA is expected to alleviate hair loss by regulating 5α-reductase activity and the receptor's androgen sensitivity.
MeSH Terms
-
Alopecia
Blotting, Western
Cell Count
Cell Proliferation
Cholestenone 5 alpha-Reductase
Dihydrotestosterone*
Down-Regulation
Finasteride
Hair*
Humans*
Interleukin-6
Protein-Tyrosine Kinases
Receptors, Androgen
RNA, Messenger
Tumor Necrosis Factor-alpha
Up-Regulation
Cholestenone 5 alpha-Reductase
Dihydrotestosterone
Finasteride
Interleukin-6
Protein-Tyrosine Kinases
RNA, Messenger
Receptors, Androgen
Tumor Necrosis Factor-alpha
Figure
Reference
-
1. Courtois M, Loussouarn G, Hourseau C, Grollier JF. Hair cycle and alopecia. Skin Pharmacol. 1994; 7:84–89.
Article2. Choudhry R, Hodgins MB, Van der Kwast TH, Brinkmann AO, Boersma WJ. Localization of androgen receptors in human skin by immunohistochemistry: implications for the hormonal regulation of hair growth, sebaceous glands and sweat glands. J Endocrinol. 1992; 133:467–475.
Article3. Hibberts NA, Howell AE, Randall VA. Balding hair follicle dermal papilla cells contain higher levels of androgen receptors than those from non-balding scalp. J Endocrinol. 1998; 156:59–65.
Article4. Sawaya ME, Price VH. Different levels of 5alpha-reductase type I and II, aromatase, and androgen receptor in hair follicles of women and men with androgenetic alopecia. J Invest Dermatol. 1997; 109:296–300.
Article5. Kaufman KD, Olsen EA, Whiting D, Savin R, DeVillez R, Bergfeld W, et al. Finasteride in the treatment of men with androgenetic alopecia. Finasteride Male Pattern Hair Loss Study Group. J Am Acad Dermatol. 1998; 39(4 Pt 1):578–589.6. Schweikert HU, Wilson JD. Regulation of human hair growth by steroid hormones. II. Androstenedione metabolism in isolated hairs. J Clin Endocrinol Metab. 1974; 39:1012–1019.
Article7. Inui S, Fukuzato Y, Nakajima T, Yoshikawa K, Itami S. Identification of androgen-inducible TGF-beta1 derived from dermal papilla cells as a key mediator in androgenetic alopecia. J Investig Dermatol Symp Proc. 2003; 8:69–71.
Article8. Kwack MH, Kim MK, Kim JC, Sung YK. Dickkopf 1 promotes regression of hair follicles. J Invest Dermatol. 2012; 132:1554–1560.
Article9. Yu M, Kissling S, Freyschmidt-Paul P, Hoffmann R, Shapiro J, McElwee KJ. Interleukin-6 cytokine family member oncostatin M is a hair-follicle-expressed factor with hair growth inhibitory properties. Exp Dermatol. 2008; 17:12–19.
Article10. Inui S, Fukuzato Y, Nakajima T, Kurata S, Itami S. Androgen receptor co-activator Hic-5/ARA55 as a molecular regulator of androgen sensitivity in dermal papilla cells of human hair follicles. J Invest Dermatol. 2007; 127:2302–2306.
Article11. Kaliyadan F, Nambiar A, Vijayaraghavan S. Androgenetic alopecia: an update. Indian J Dermatol Venereol Leprol. 2013; 79:613–625.
Article12. Kwack MH, Ahn JS, Kim MK, Kim JC, Sung YK. Dihydrotestosterone-inducible IL-6 inhibits elongation of human hair shafts by suppressing matrix cell proliferation and promotes regression of hair follicles in mice. J Invest Dermatol. 2012; 132:43–49.
Article13. Inui S, Itami S. Molecular basis of androgenetic alopecia: from androgen to paracrine mediators through dermal papilla. J Dermatol Sci. 2011; 61:1–6.
Article14. Uchiyama C, Ishida K, Tsutsui T, Naito A, Kurita K, Hanihara H, et al. Effects of Hura crepitans and its active ingredient, daphne factor F3, on dihydrotestosterone-induced neurotrophin-4 activation and hair retardation. Biol Pharm Bull. 2012; 35:42–47.
Article15. Upton JH, Hannen RF, Bahta AW, Farjo N, Farjo B, Philpott MP. Oxidative stress-associated senescence in dermal papilla cells of men with androgenetic alopecia. J Invest Dermatol. 2015; 135:1244–1252.
Article16. Audet-Walsh É, Yee T, Tam IS, Giguère V. Inverse regulation of DHT synthesis enzymes 5α-reductase types 1 and 2 by the androgen receptor in prostate cancer. Endocrinology. 2017; 158:1015–1021.
Article17. Shin H, Yoo HG, Inui S, Itami S, Kim IG, Cho AR, et al. Induction of transforming growth factor-beta 1 by androgen is mediated by reactive oxygen species in hair follicle dermal papilla cells. BMB Rep. 2013; 46:460–464.
Article18. Hoffmann R. The potential role of cytokines and T cells in alopecia areata. J Investig Dermatol Symp Proc. 1999; 4:235–238.
Article19. Philpott MP, Sanders DA, Bowen J, Kealey T. Effects of interleukins, colony-stimulating factor and tumour necrosis factor on human hair follicle growth in vitro: a possible role for interleukin-1 and tumour necrosis factor-alpha in alopecia areata. Br J Dermatol. 1996; 135:942–948.
Article20. Kasumagic-Halilovic E, Prohic A, Cavaljuga S. Tumor necrosis factor-alpha in patients with alopecia areata. Indian J Dermatol. 2011; 56:494–496.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Chrysanthemum zawadskii Extract on Dermal Papilla Cell Proliferation and Hair Growth
- Effects of Alopecia Areata Serum on Proliferation of Cultured Dermal Papilla Cells
- Effects of Vaseular Endothelial Growth Factors on Hair Growth in Vitro
- Epigallocatechin Gallate-Mediated Alteration of the MicroRNA Expression Profile in 5α-Dihydrotestosterone-Treated Human Dermal Papilla Cells
- Enhancing the Angiogenic and Proliferative Capacity of Dermal Fibroblasts with Mulberry ( Morus alba. L) Root Extract