Cancer Res Treat.
2019 Jul;51(3):861-875. 10.4143/crt.2018.214.
A Phage Display-Identified Peptide Selectively Binds to Kidney Injury Molecule-1 (KIM-1) and Detects KIM-1–Overexpressing Tumors In Vivo
- Affiliations
-
- 1Department of Biochemistry and Cell Biology, School of Medicine, Kyungpook National University, Daegu, Korea. leebh@knu.ac.kr
- 2BK21 Plus KNU Biomedical Convergence Program, School of Medicine, Kyungpook National University, Daegu, Korea.
- 3CMRI, School of Medicine, Kyungpook National University, Daegu, Korea.
- 4Laboratory Animal Center, Daegu Gyeongbuk Medical Innovation Foundation, Daegu, Korea.
- KMID: 2454279
- DOI: http://doi.org/10.4143/crt.2018.214
Abstract
- PURPOSE
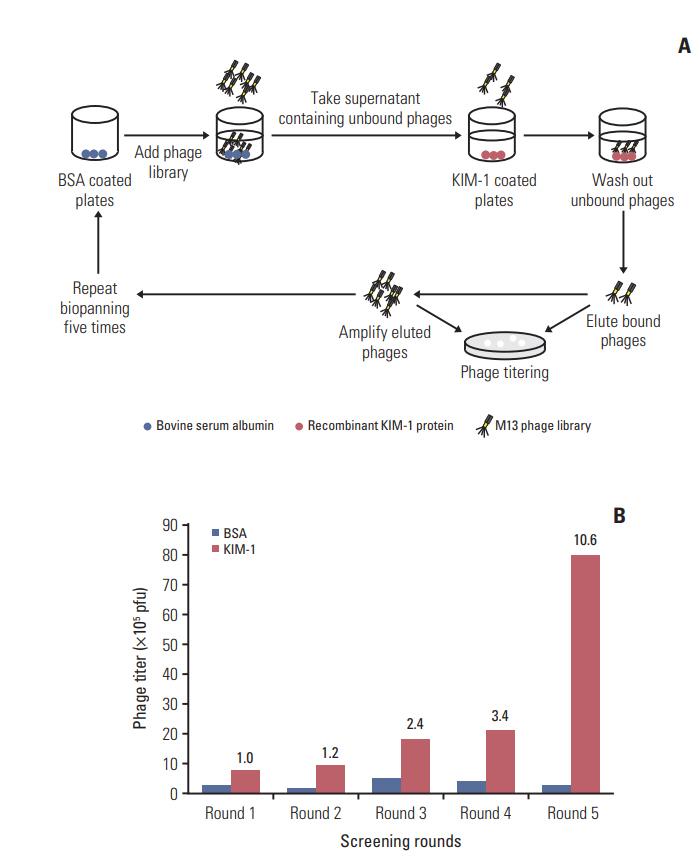
This study was carried out to identify a peptide that selectively binds to kidney injury molecule-1 (KIM-1) by screening a phage-displayed peptide library and to use the peptide for the detection of KIM-1overexpressing tumors in vivo.
MATERIALS AND METHODS
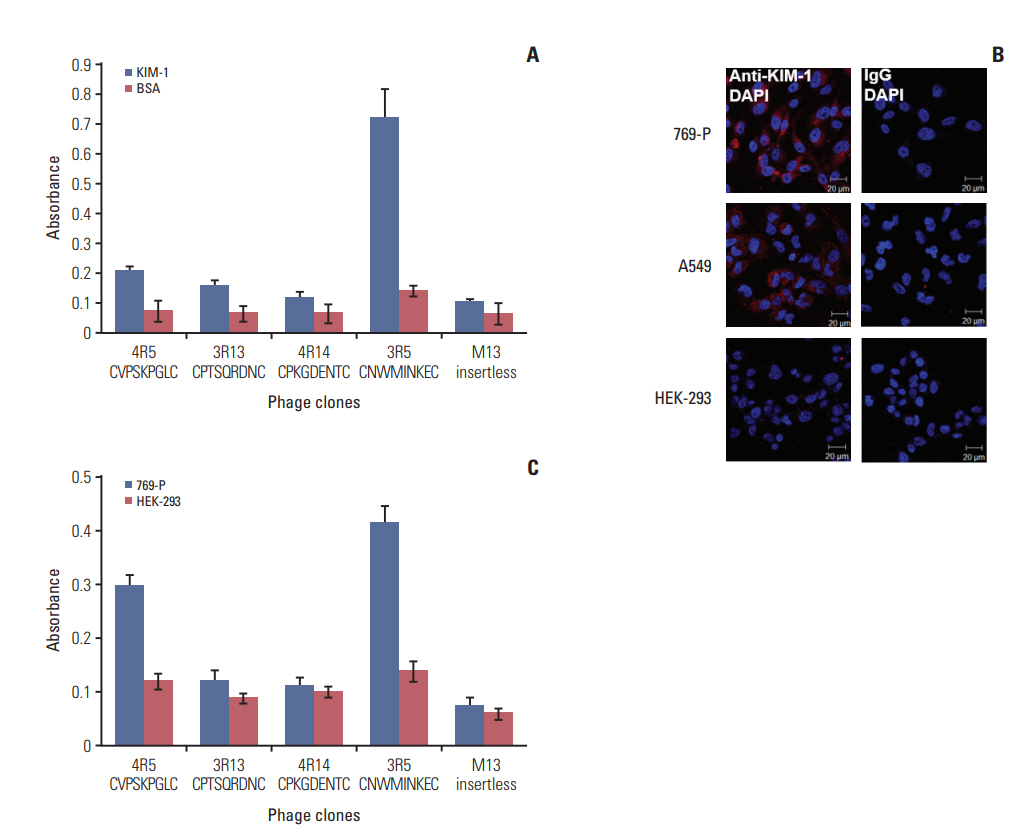
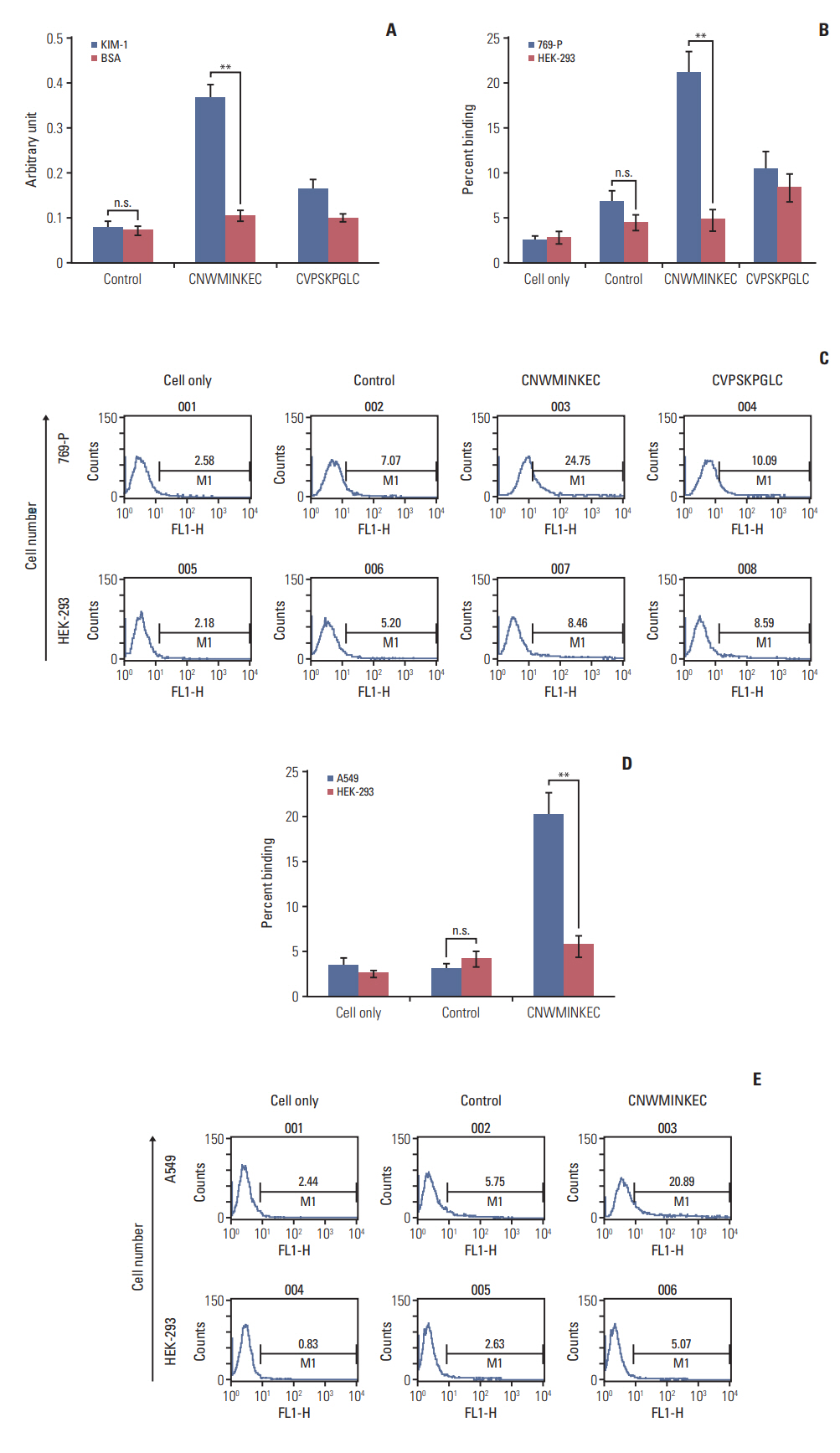
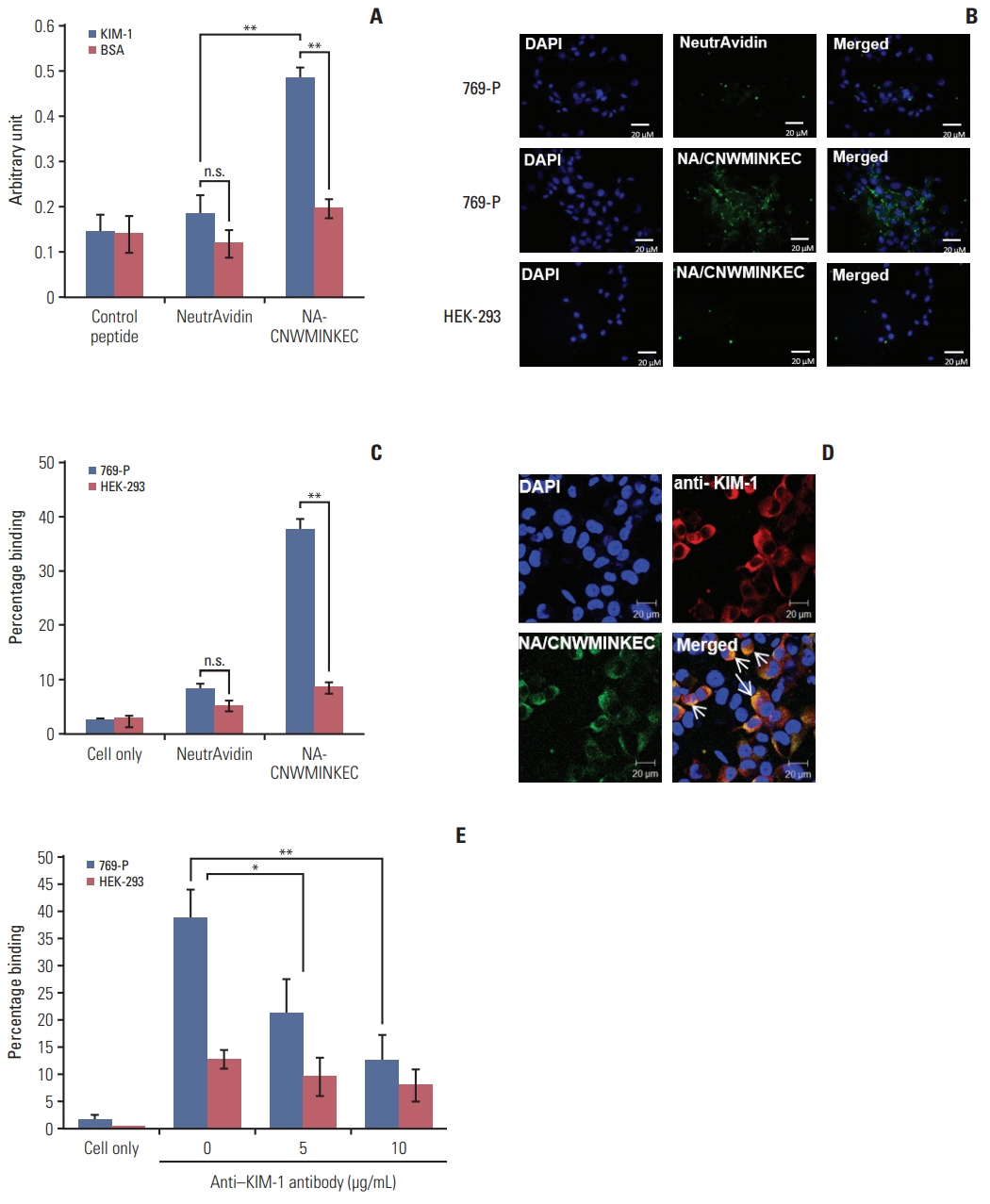
Biopanning of a phage-displayed peptide library was performed on KIM-1-coated plates. The binding of phage clones, peptides, and a peptide multimer to the KIM-1 protein and KIM-1-overexpressing and KIM-1-low expressing cells was examined by enzyme-linked immunosorbent assay, fluorometry, and flow cytometry. A biotin-peptide multimer was generated using NeutrAvidin. In vivo homing of the peptide to KIM-1-overexpressing and KIM1-low expressing tumors in mice was examined by whole-body fluorescence imaging.
RESULTS
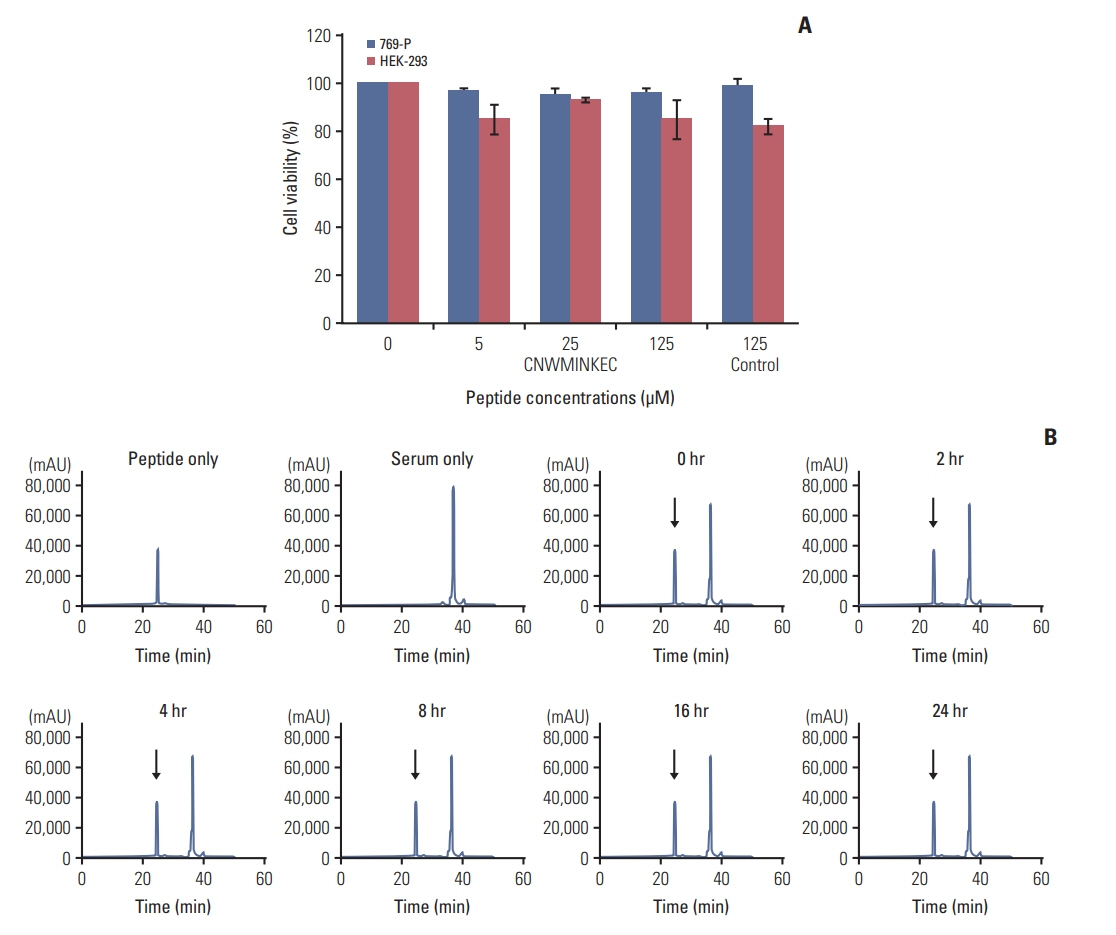
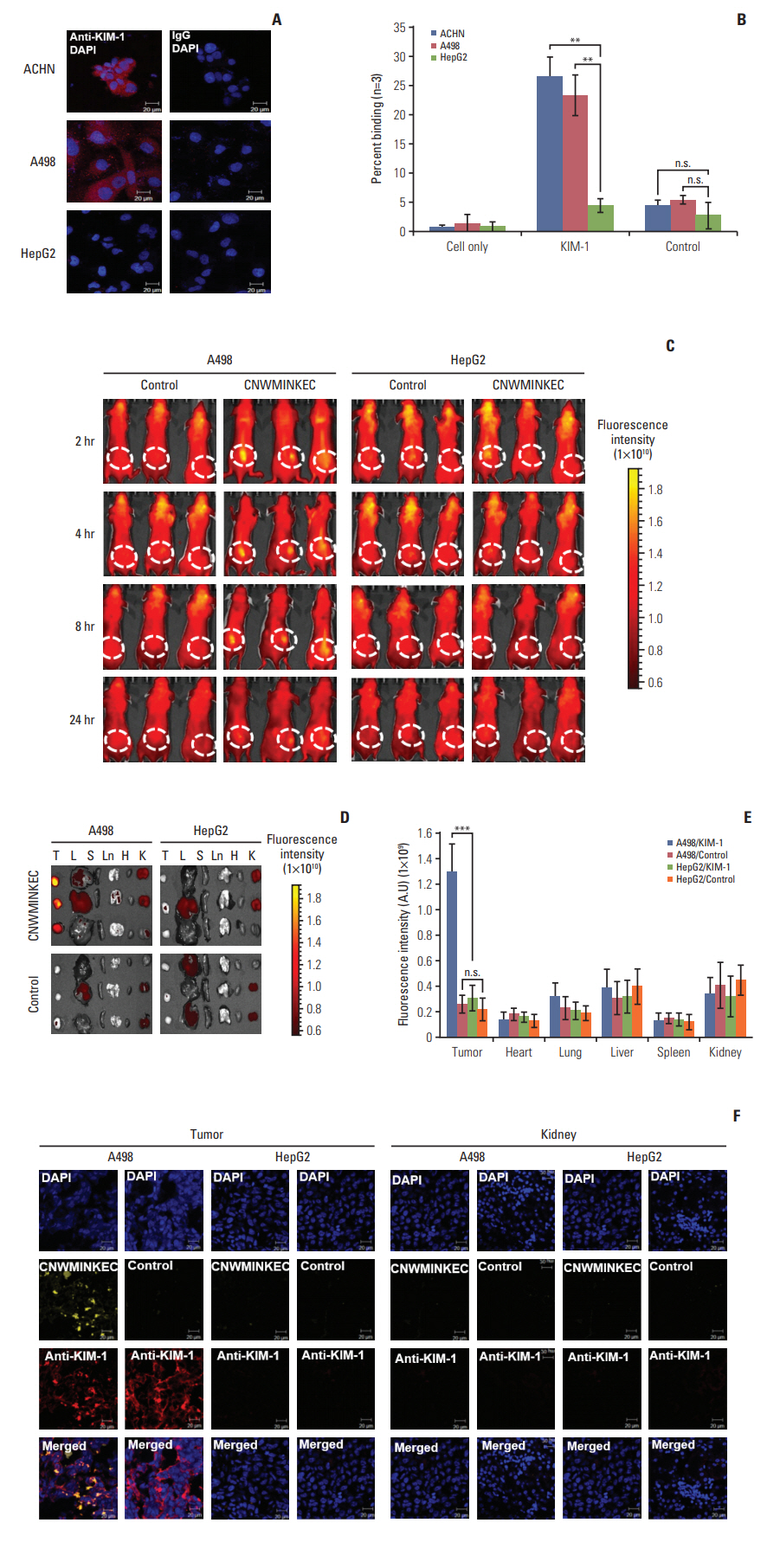
A phage clone displaying the CNWMINKEC peptide showed higher binding affinity to KIM-1 and KIM-1-overexpressing 769-P renal tumor cells compared to other phage clones selected after biopanning. The CNWMINKEC peptide and a NeutrAvidin/biotin-CNWMINKEC multimer selectively bound to KIM-1 over albumin and to KIM-1-overexpressing 769-P cells and A549 lung tumor cells compared to KIM-1-low expressing HEK293 normal cells. Co-localization and competition assays using an anti-KIM-1 antibody demonstrated that the binding of the CNWMINKEC peptide to 769-P cells was specifically mediated by KIM-1. The CNWMINKEC peptide was not cytotoxic to cells and was stable for up to 24 hours in the presence of serum. Whole-body fluorescence imaging demonstrated selective homing of the CNWM-INKEC peptide to KIM-1-overexpressing A498 renal tumor compared to KIM1-low expressing HepG2 liver tumor in mice.
CONCLUSION
The CNWMINKEC peptide is a promising probe for in vivo imaging and detection of KIM-1"’overexpressing tumors.
MeSH Terms
Figure
Reference
-
References
1. Ichimura T, Bonventre JV, Bailly V, Wei H, Hession CA, Cate RL, et al. Kidney injury molecule-1 (KIM-1), a putative epithelial cell adhesion molecule containing a novel immunoglobulin domain, is up-regulated in renal cells after injury. J Biol Chem. 1998; 273:4135–42.
Article2. Ichimura T, Hung CC, Yang SA, Stevens JL, Bonventre JV. Kidney injury molecule-1: a tissue and urinary biomarker for nephrotoxicant-induced renal injury. Am J Physiol Renal Physiol. 2004; 286:F552–63.
Article3. Bailly V, Zhang Z, Meier W, Cate R, Sanicola M, Bonventre JV. Shedding of kidney injury molecule-1, a putative adhesion protein involved in renal regeneration. J Biol Chem. 2002; 277:39739–48.
Article4. Vaidya VS, Ramirez V, Ichimura T, Bobadilla NA, Bonventre JV. Urinary kidney injury molecule-1: a sensitive quantitative biomarker for early detection of kidney tubular injury. Am J Physiol Renal Physiol. 2006; 290:F517–29.
Article5. Liangos O, Tighiouart H, Perianayagam MC, Kolyada A, Han WK, Wald R, et al. Comparative analysis of urinary biomarkers for early detection of acute kidney injury following cardiopulmonary bypass. Biomarkers. 2009; 14:423–31.
Article6. Han WK, Waikar SS, Johnson A, Betensky RA, Dent CL, Devarajan P, et al. Urinary biomarkers in the early diagnosis of acute kidney injury. Kidney Int. 2008; 73:863–9.
Article7. Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008; 58:71–96.
Article8. Chow WH, Devesa SS, Warren JL, Fraumeni JF Jr. Rising incidence of renal cell cancer in the United States. JAMA. 1999; 281:1628–31.
Article9. Hock LM, Lynch J, Balaji KC. Increasing incidence of all stages of kidney cancer in the last 2 decades in the United States: an analysis of surveillance, epidemiology and end results program data. J Urol. 2002; 167:57–60.
Article10. Motzer RJ, Bander NH, Nanus DM. Renal-cell carcinoma. N Engl J Med. 1996; 335:865–75.
Article11. Zambrano NR, Lubensky IA, Merino MJ, Linehan WM, Walther MM. Histopathology and molecular genetics of renal tumors toward unification of a classification system. J Urol. 1999; 162:1246–58.
Article12. Martignoni G, Pea M, Brunelli M, Chilosi M, Zamo A, Bertaso M, et al. CD10 is expressed in a subset of chromophobe renal cell carcinomas. Mod Pathol. 2004; 17:1455–63.
Article13. Avery AK, Beckstead J, Renshaw AA, Corless CL. Use of antibodies to RCC and CD10 in the differential diagnosis of renal neoplasms. Am J Surg Pathol. 2000; 24:203–10.
Article14. Chu P, Arber DA. Paraffin-section detection of CD10 in 505 nonhematopoietic neoplasms. Frequent expression in renal cell carcinoma and endometrial stromal sarcoma. Am J Clin Pathol. 2000; 113:374–82.15. Han WK, Alinani A, Wu CL, Michaelson D, Loda M, McGovern FJ, et al. Human kidney injury molecule-1 is a tissue and urinary tumor marker of renal cell carcinoma. J Am Soc Nephrol. 2005; 16:1126–34.
Article16. Zhang PL, Mashni JW, Sabbisetti VS, Schworer CM, Wilson GD, Wolforth SC, et al. Urine kidney injury molecule-1: a potential non-invasive biomarker for patients with renal cell carcinoma. Int Urol Nephrol. 2014; 46:379–88.
Article17. Vila MR, Kaplan GG, Feigelstock D, Nadal M, Morote J, Porta R, et al. Hepatitis A virus receptor blocks cell differentiation and is overexpressed in clear cell renal cell carcinoma. Kidney Int. 2004; 65:1761–73.18. Lin F, Zhang PL, Yang XJ, Shi J, Blasick T, Han WK, et al. Human kidney injury molecule-1 (hKIM-1): a useful immunohistochemical marker for diagnosing renal cell carcinoma and ovarian clear cell carcinoma. Am J Surg Pathol. 2007; 31:371–81.
Article19. Thomas LJ, Vitale L, O'Neill T, Dolnick RY, Wallace PK, Minderman H, et al. Development of a novel antibody-drug conjugate for the potential treatment of ovarian, lung, and renal cell carcinoma expressing TIM-1. Mol Cancer Ther. 2016; 15:2946–54.
Article20. Ladner RC, Sato AK, Gorzelany J, de Souza M. Phage displayderived peptides as therapeutic alternatives to antibodies. Drug Discov Today. 2004; 9:525–9.
Article21. Lee S, Xie J, Chen X. Peptides and peptide hormones for molecular imaging and disease diagnosis. Chem Rev. 2010; 110:3087–111.
Article22. Pasqualini R, Koivunen E, Ruoslahti E. Alpha v integrins as receptors for tumor targeting by circulating ligands. Nat Biotechnol. 1997; 15:542–6.23. Hong HY, Lee HY, Kwak W, Yoo J, Na MH, So IS, et al. Phage display selection of peptides that home to atherosclerotic plaques: IL-4 receptor as a candidate target in atherosclerosis. J Cell Mol Med. 2008; 12:2003–14.
Article24. Chi L, Na MH, Jung HK, Vadevoo SM, Kim CW, Padmanaban G, et al. Enhanced delivery of liposomes to lung tumor through targeting interleukin-4 receptor on both tumor cells and tumor endothelial cells. J Control Release. 2015; 209:327–36.
Article25. Wang K, Purushotham S, Lee JY, Na MH, Park H, Oh SJ, et al. In vivo imaging of tumor apoptosis using histone H1-targeting peptide. J Control Release. 2010; 148:283–91.26. Padmanaban G, Park H, Choi JS, Cho YW, Kang WC, Moon CI, et al. Identification of peptides that selectively bind to myoglobin by biopanning of phage displayed-peptide library. J Biotechnol. 2014; 187:43–50.
Article27. Cuadros T, Trilla E, Vila MR, de Torres I, Vilardell J, Messaoud NB, et al. Hepatitis A virus cellular receptor 1/kidney injury molecule-1 is a susceptibility gene for clear cell renal cell carcinoma and hepatitis A virus cellular receptor/kidney injury molecule-1 ectodomain shedding a predictive biomarker of tumour progression. Eur J Cancer. 2013; 49:2034–47.
Article28. Liu Y. The place of FDG PET/CT in renal cell carcinoma: value and limitations. Front Oncol. 2016; 6:201.
Article29. Gergel L, Forsberg E, Pilsmaker C, Boyer J, Round S, Borrelli K, et al. Preclinical efficacy of an antibody-drug conjugate targeting TIM-1 in ovarian, renal and lung tumor models. FASEB J. 2015; 29(1 Suppl):945.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Terminal Protein-specific scFv Production by Phage Display
- Identification of high-affinity VEGFR3-binding peptides through a phage-displayed random peptide library
- Development of a Pancreatic Cancer Specific Binding Peptide Using Phage Display
- Engineering a High-Affinity PD-1 Peptide for Optimized Immune Cell-Mediated Tumor Therapy
- Peptide Mimotopes of Neisseria meningitidis Group B Capsular Polysaccharide