J Vet Sci.
2019 Jan;20(1):16-26. 10.4142/jvs.2019.20.1.16.
Correlation of spontaneous adipocyte generation with osteogenic differentiation of porcine skin-derived stem cells
- Affiliations
-
- 1College of Veterinary Medicine and Institute of Veterinary Science, Kangwon National University, Chuncheon 24341, Korea. htcheong@kangwon.ac.kr
- 2College of Animal Life Sciences, Kangwon National University, Chuncheon 24341, Korea.
- KMID: 2434773
- DOI: http://doi.org/10.4142/jvs.2019.20.1.16
Abstract
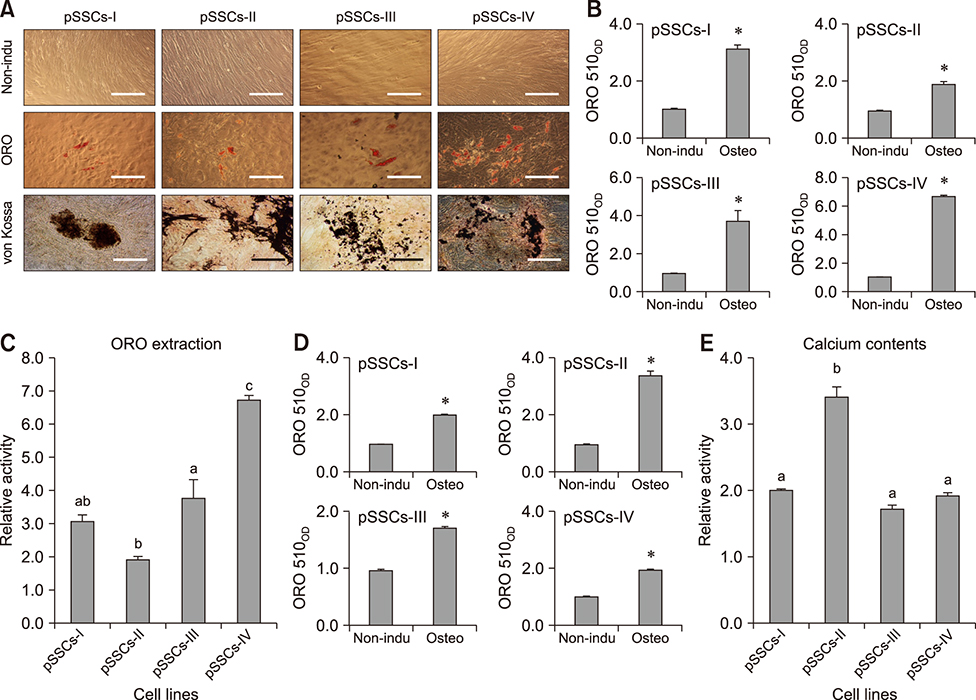
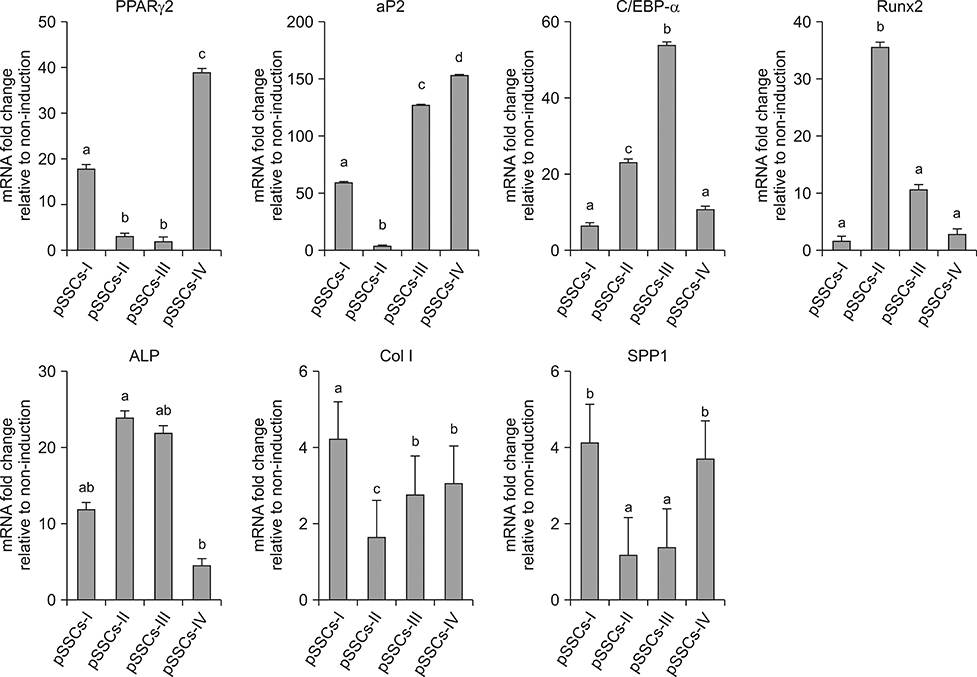
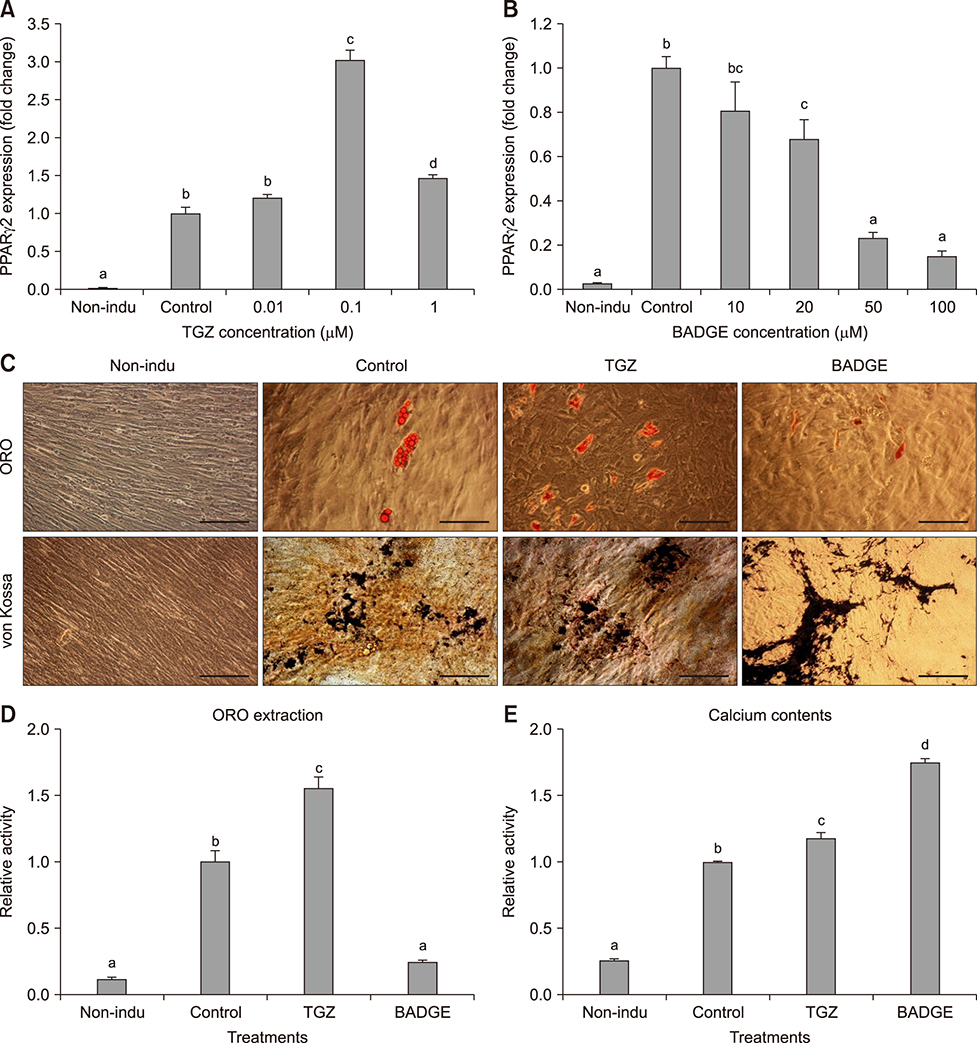
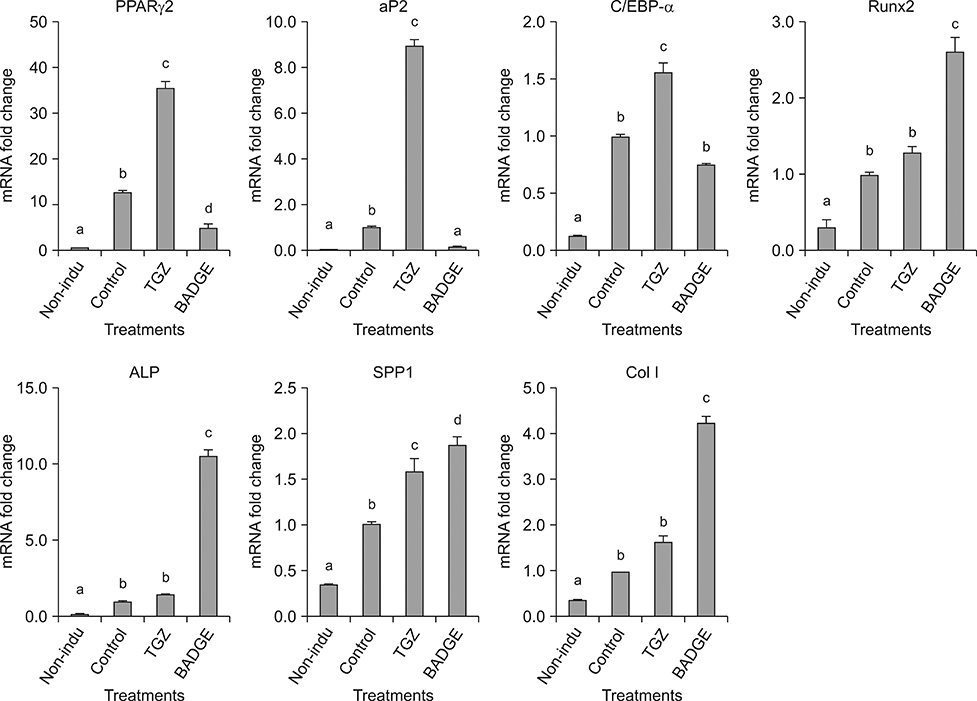
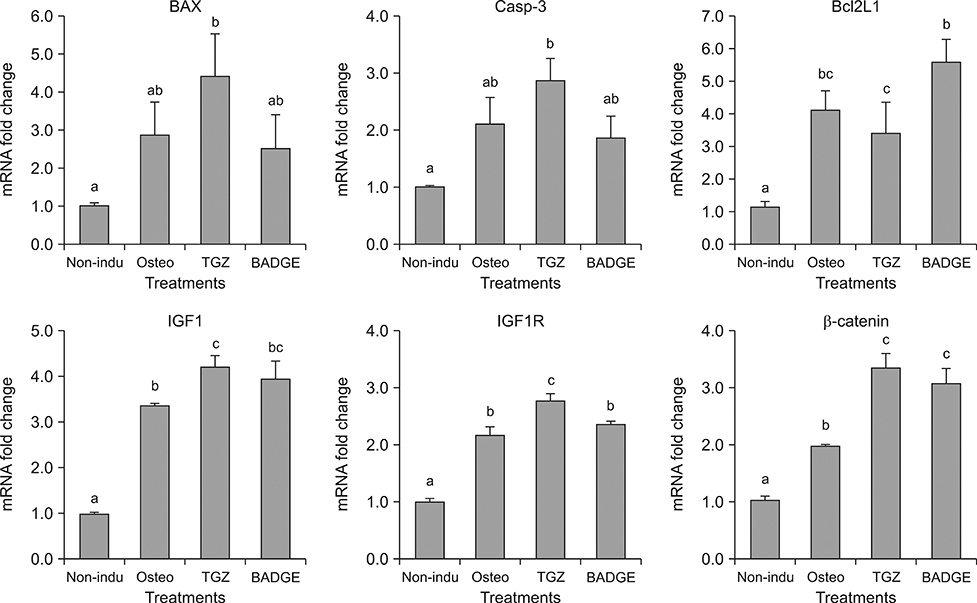
- The objective of this study was to examine effects of spontaneous adipocyte generation on osteogenic differentiation of porcine skin-derived stem cells (pSSCs). Correlation between osteogenic differentiation and adipocyte differentiation induced by osteocyte induction culture was determined using different cell lines. Osteogenic differentiation efficiency of pSSCs was then analyzed by controlling the expression of adipocyte-specific transcription factors during osteogenic induction culture. Among four cell lines, pSSCs-II had the lowest lipid droplet level but the highest calcium content (p < 0.05). It also expressed significantly low levels of peroxisome proliferator-activated receptor gamma 2 (PPARγ2) and adipocyte protein 2 (aP2) mRNAs but very high levels of runt-related transcription factor 2 (Runx2) and alkaline phosphatase (ALP) mRNAs as osteogenic makers (p < 0.05). Oil red O extraction was increased by 0.1 µM troglitazone (TGZ) treatment but decreased by 50 µM bisphenol A diglycidyl ether (BADGE) (p < 0.05). Calcium content was drastically increased after BADGE treatment compared to that in osteogenic induction control and TGZ-treated pSSCs (p < 0.05). Relative expression levels of PPARγ2 and aP2 mRNAs were increased by TGZ but decreased by BADGE. Expression levels of Rucx2 and ALP mRNAs, osteoblast-specific marker genes, were significantly increased by BADGE treatment (p < 0.05). The expression level of BCL2 like 1 was significantly higher in BADGE-treated pSSCs than that in TGZ-treated ones (p < 0.05). The results demonstrate that spontaneous adipocyte generation does not adversely affect osteogenic differentiation. However, reducing spontaneous adipocyte generation by inhibiting PPARγ2 mRNA expression can enhance in vitro osteogenic differentiation of pSSCs.
Keyword
MeSH Terms
Figure
Reference
-
1. Akune T, Ohba S, Kamekura S, Yamaguchi M, Chung UI, Kubota N, Terauchi Y, Harada Y, Azuma Y, Nakamura K, Kadowaki T, Kawaguchi H. PPARγ insufficiency enhances osteogenesis through osteoblast formation from bone marrow progenitors. J Clin Invest. 2004; 113:846–855.
Article2. Aubert RE, Herrera V, Chen W, Haffner SM, Pendergrass M. Rosiglitazone and pioglitazone increase fracture risk in women and men with type 2 diabetes. Diabetes Obes Metab. 2010; 12:716–721.
Article3. Bae HK, Jung BD, Lee S, Park CK, Yang BK, Cheong HT. Chondrogenic differentiation of porcine skin-derived stem cells with different characteristics of spontaneous adipocyte formation. Korean J Embryo Transf. 2017; 32:193–200.
Article4. Beresford JN, Bennett JH, Devlin C, Leboy PS, Owen ME. Evidence for an inverse relationship between the differentiation of adipocytic and osteogenic cells in rat marrow stromal cell cultures. J Cell Sci. 1992; 102:341–351.
Article5. Botolin S, McCabe LR. Inhibition of PPARγ prevents type I diabetic bone marrow adiposity but not bone loss. J Cell Physiol. 2006; 209:967–976.
Article6. Bruedigam C, Eijken M, Koedam M, van de Peppel J, Drabek K, Chiba H, van Leeuwen JP. A new concept underlying stem cell lineage skewing that explains the detrimental effects of thiazolidinediones on bone. Stem Cells. 2010; 28:916–927.
Article7. Chamorro-García R, Kirchner S, Li X, Janesick A, Casey SC, Chow C, Blumberg B. Bisphenol A diglycidyl ether induces adipogenic differentiation of multipotent stromal stem cells through a peroxisome proliferator-activated receptor gamma-independent mechanism. Environ Health Perspect. 2012; 120:984–989.
Article8. Gimble JM, Robinson CE, Wu X, Kelly KA. The function of adipocytes in the bone marrow stroma: an update. Bone. 1996; 19:421–428.
Article9. Hong KM, Belperio JA, Keane MP, Burdick MD, Strieter RM. Differentiation of human circulating fibrocytes as mediated by transforming growth factor-β and peroxisome proliferator-activated receptor γ. J Biol Chem. 2007; 282:22910–22920.
Article10. Hwang IS, Bae HK, Cheong HT. Comparison of the characteristics and multipotential and in vivo cartilage formation capabilities between porcine adipose-derived stem cells and porcine skin-derived stem cell-like cells. Am J Vet Res. 2015; 76:814–821.
Article11. James AW. Review of signaling pathways governing MSC osteogenic and adipogenic differentiation. Scientifica (Cairo). 2013; 2013:684736.
Article12. Krause U, Harris S, Green A, Ylostalo J, Zeitouni S, Lee N, Gregory CA. Pharmaceutical modulation of canonical Wnt signaling in multipotent stromal cells for improved osteoinductive therapy. Proc Natl Acad Sci U S A. 2010; 107:4147–4152.
Article13. Li X, Cui Q, Kao C, Wang GJ, Balian G. Lovastatin inhibits adipogenic and stimulates osteogenic differentiation by suppressing PPARγ and increasing Cbfa1/Runx2 expression in bone marrow mesenchymal cell cultures. Bone. 2003; 33:652–659.
Article14. Meier C, Kraenzlin ME, Bodmer M, Jick SS, Jick H, Meier CR. Use of thiazolidinediones and fracture risk. Arch Intern Med. 2008; 168:820–825.
Article15. Moriishi T, Maruyama Z, Fukuyama R, Ito M, Miyazaki T, Kitaura H, Ohnishi H, Furuichi T, Kawai Y, Masuyama R, Komori H, Takada K, Kawaguchi H, Komori T. Overexpression of Bcl2 in osteoblasts inhibits osteoblast differentiation and induces osteocyte apoptosis. PLoS One. 2011; 6:e27487.
Article16. Muruganandan S, Roman AA, Sinal CJ. Adipocyte differentiation of bone marrow-derived mesenchymal stem cells: cross talk with the osteoblastogenic program. Cell Mol Life Sci. 2009; 66:236–253.
Article17. Nadler J, Scott S. Evidence that pioglitazone increases intracellular free magnesium concentration in freshly isolated rat adipocytes. Biochem Biophys Res Commun. 1994; 202:416–421.
Article18. Oshima K, Nampei A, Matsuda M, Iwaki M, Fukuhara A, Hashimoto J, Yoshikawa H, Shimomura I. Adiponectin increases bone mass by suppressing osteoclast and activating osteoblast. Biochem Biophys Res Commun. 2005; 331:520–526.
Article19. Patel JJ, Butters OR, Arnett TR. PPAR agonists stimulate adipogenesis at the expense of osteoblast differentiation while inhibiting osteoclast formation and activity. Cell Biochem Funct. 2014; 32:368–377.
Article20. Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science. 1999; 284:143–147.
Article21. Reinhart RA. Magnesium metabolism. A review with special reference to the relationship between intracellular content and serum levels. Arch Intern Med. 1988; 148:2415–2420.
Article22. Rochefort GY, Pallu S, Benhamou CL. Osteocyte: the unrecognized side of bone tissue. Osteoporos Int. 2010; 21:1457–1469.
Article23. Takaya J, Higashino H, Kobayashi Y. Intracellular magnesium and insulin resistance. Magnes Res. 2004; 17:126–136.24. Tchoukalova YD, Hausman DB, Dean RG, Hausman GJ. Enhancing effect of troglitazone on porcine adipocyte differentiation in primary culture: a comparison with dexamethasone. Obes Res. 2000; 8:664–672.
Article25. Wabitsch M, Hauner H, Heinze E, Teller WM. The role of growth hormone/insulin-like growth factors in adipocyte differentiation. Metabolism. 1995; 44:10 Suppl 4. 45–49.
Article26. Xian L, Wu X, Pang L, Lou M, Rosen CJ, Qiu T, Crane J, Frassica F, Zhang L, Rodriguez JP, Jia X, Yakar S, Xuan S, Efstratiadis A, Wan M, Cao X. Matrix IGF-1 maintains bone mass by activation of mTOR in mesenchymal stem cells. Nat Med. 2012; 18:1095–1101.
Article27. Yu WH, Li FG, Chen XY, Li JT, Wu YH, Huang LH, Wang Z, Li P, Wang T, Lahn BT, Xiang AP. PPARγ suppression inhibits adipogenesis but does not promote osteogenesis of human mesenchymal stem cells. Int J Biochem Cell Biol. 2012; 44:377–384.
Article28. Zhang W, Pantschenko AG, McCarthy MB, Gronowicz G. Bone-targeted overexpression of Bcl-2 increases osteoblast adhesion and differentiation and inhibits mineralization in vitro. Calcif Tissue Int. 2007; 80:111–122.
Article29. Zhang X, Yang M, Lin L, Chen P, Ma KT, Zhou CY, Ao YF. Runx2 overexpression enhances osteoblastic differentiation and mineralization in adipose--derived stem cells in vitro and in vivo. Calcif Tissue Int. 2006; 79:169–178.
Article30. Zhu RJ, Wu MQ, Li ZJ, Zhang Y, Liu KY. Hematopoietic recovery following chemotherapy is improved by BADGE-induced inhibition of adipogenesis. Int J Hematol. 2013; 97:58–72.
Article31. Zhuang H, Zhang X, Zhu C, Tang X, Yu F, Shang GW, Cai X. Molecular mechanisms of PPAR-γ governing MSC osteogenic and adipogenic differentiation. Curr Stem Cell Res Ther. 2016; 11:255–264.
Article32. Zou L, Zou X, Chen L, Li H, Mygind T, Kassem M, Bünger C. Multilineage differentiation of porcine bone marrow stromal cells associated with specific gene expression pattern. J Orthop Res. 2008; 26:56–64.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Modulation of Osteogenic Differentiation of Adipose-Derived Stromal Cells by Co-Treatment with 3, 4’-Dihydroxyflavone, U0126, and N-Acetyl Cysteine
- Dlx3 and Dlx5 Inhibit Adipogenic Differentiation of Human Dental Pulp Stem Cells
- Osteogenic Differentiation of Human Adipose-derived Stem Cells within PLGA(Poly(D,L-lactic-co-glycolic acid)) Scaffold in the Nude Mouse
- Histological Analysis of In Vitro Co-Culture and In Vivo Mice Co-Transplantation of Stem Cell-Derived Adipocyte and Osteoblast
- The effect of growth factors on osteogenic differentiation of adipose tissue-derived stromal cells