Allergy Asthma Immunol Res.
2018 Jan;10(1):77-82. 10.4168/aair.2018.10.1.77.
T-Helper Type 2 Cells Direct Antigen-Induced Eosinophilic Skin Inflammation in Mice
- Affiliations
-
- 1Center for Life Science Research, University of Yamanashi, Yamanashi, Japan. osamuk@yamanashi.ac.jp
- 2Allergy and Immunology Project, Tokyo Metropolitan Institute of Medical Science, Tokyo, Japan.
- 3Clinical Research Center for Allergy and Rheumatology, National Sagamihara Hospital, Kanagawa, Japan.
- 4RIKEN Bioresource Center, Ibaraki, Japan.
- KMID: 2428852
- DOI: http://doi.org/10.4168/aair.2018.10.1.77
Abstract
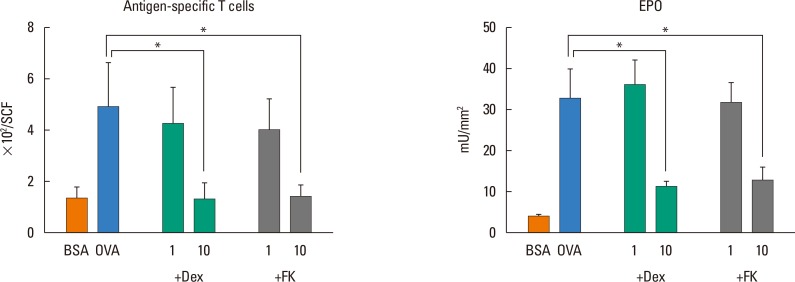
- Eosinophilic inflammation in combination with immunoglobulin E (IgE) production is a characteristic feature of atopic dermatitis. Although activated T-helper type (Th) 2 cells play critical roles in the local accumulation and activation of eosinophils, whether they induce eosinophilic skin inflammation, independent of the IgE-mediated pathway has been unclear. To address the functional role of T cells in allergic skin diseases, we herein transferred Th1/Th2-differentiated or naive DO11.10 T cells into unprimed BALB/c mice. Ovalbumin-specific Th2 cells, as well as eosinophils, accumulated in the skin upon antigen challenge, despite the absence of antigen-specific IgE. Neither antigen-specific Th1 nor naive T cells induced eosinophil accumulation, although Th1 cells by themselves migrated into the skin. Interleukin (IL)-4, IL-5, and eotaxin were specifically produced in the skin of antigen-challenged, Th2 cell-transferred mice, whereas interferon (IFN)-γ and regulated on activation, normal T cell expressed and secreted (RANTES) were preferentially produced in Th1 cells-transferred mice. Production of monocyte chemoattractant protein (MCP)-1 and MCP-3 was enhanced by both Th1 and Th2 cells. The accumulation of eosinophils and Th2 cells in the skin was suppressed by both dexamethasone and FK506, indicating an essential role of Th2 cells in eosinophil recruitment. We conclude that Th2 cells can induce eosinophilic infiltration into the skin in the absence of antigen-specific IgE.
MeSH Terms
-
Animals
Chemokines
Cytokines
Dermatitis, Atopic
Dexamethasone
Eosinophils*
Immunoglobulin E
Immunoglobulins
Inflammation*
Interferons
Interleukin-5
Interleukins
Mice*
Monocytes
Skin Diseases
Skin*
T-Lymphocytes
Tacrolimus
Th1 Cells
Th2 Cells
Chemokines
Cytokines
Dexamethasone
Immunoglobulin E
Immunoglobulins
Interferons
Interleukin-5
Interleukins
Tacrolimus
Figure
Reference
-
1. Leiferman KM. Eosinophils in atopic dermatitis. J Allergy Clin Immunol. 1994; 94:1310–1317. PMID: 7798571.
Article2. Leiferman KM. A current perspective on the role of eosinophils in dermatologic diseases. J Am Acad Dermatol. 1991; 24:1101–1112. PMID: 2071704.
Article3. Ott NL, Gleich GJ, Peterson EA, Fujisawa T, Sur S, Leiferman KM. Assessment of eosinophil and neutrophil participation in atopic dermatitis: comparison with the IgE-mediated late-phase reaction. J Allergy Clin Immunol. 1994; 94:120–128. PMID: 8027490.
Article4. Peters MS, Schroeter AL, Kephart GM, Gleich GJ. Localization of eosinophil granule major basic protein in chronic urticaria. J Invest Dermatol. 1983; 81:39–43. PMID: 6345685.
Article5. Walker C, Kägi MK, Ingold P, Braun P, Blaser K, Bruijnzeel-Koomen CA, et al. Atopic dermatitis: correlation of peripheral blood T cell activation, eosinophilia and serum factors with clinical severity. Clin Exp Allergy. 1993; 23:145–153. PMID: 8448682.
Article6. Czech W, Krutmann J, Schöpf E, Kapp A. Serum eosinophil cationic protein (ECP) is a sensitive measure for disease activity in atopic dermatitis. Br J Dermatol. 1992; 126:351–355. PMID: 1571256.
Article7. Kägi MK, Joller-Jemelka H, Wüthrich B. Correlation of eosinophils, eosinophil cationic protein and soluble interleukin-2 receptor with the clinical activity of atopic dermatitis. Dermatology. 1992; 185:88–92. PMID: 1421636.8. Walsh GM. Eosinophil granule proteins and their role in disease. Curr Opin Hematol. 2001; 8:28–33. PMID: 11138623.
Article9. Hoffman DR, Yamamoto FY, Geller B, Haddad Z. Specific IgE antibodies in atopic eczema. J Allergy Clin Immunol. 1975; 55:256–267.
Article10. Sampson HA, Albergo R. Comparison of results of skin tests, RAST, and double-blind, placebo-controlled food challenges in children with atopic dermatitis. J Allergy Clin Immunol. 1984; 74:26–33. PMID: 6547461.
Article11. Gordon JR, Burd PR, Galli SJ. Mast cells as a source of multifunctional cytokines. Immunol Today. 1990; 11:458–464. PMID: 2073318.
Article12. Coyle AJ, Wagner K, Bertrand C, Tsuyuki S, Bews J, Heusser C. Central role of immunoglobulin (Ig) E in the induction of lung eosinophil infiltration and T helper 2 cell cytokine production: inhibition by a non-anaphylactogenic anti-IgE antibody. J Exp Med. 1996; 183:1303–1310. PMID: 8666888.
Article14. Park HJ, Kim EJ, Yoon D, Lee JK, Chang WS, Lim YM, et al. Prevalence of self-reported allergic diseases and IgE levels: a 2010 KNHANES analysis. Allergy Asthma Immunol Res. 2017; 9:329–339. PMID: 28497920.
Article15. Mehlhop PD, van de Rijn M, Goldberg AB, Brewer JP, Kurup VP, Martin TR, et al. Allergen-induced bronchial hyperreactivity and eosinophilic inflammation occur in the absence of IgE in a mouse model of asthma. Proc Natl Acad Sci U S A. 1997; 94:1344–1349. PMID: 9037055.
Article16. Hamelmann E, Takeda K, Schwarze J, Vella AT, Irvin CG, Gelfand EW. Development of eosinophilic airway inflammation and airway hyperresponsiveness requires interleukin-5 but not immunoglobulin E or B lymphocytes. Am J Respir Cell Mol Biol. 1999; 21:480–489. PMID: 10502558.
Article17. Ogawa K, Kaminuma O, Kikkawa H, Kameda R, Ikezawa K, Suko M, et al. Primary role of CD4+ T cells and supplemental role of mast cells in allergic pulmonary eosinophilia. Int Arch Allergy Immunol. 1999; 120(Suppl 1):15–18. PMID: 10529596.
Article18. Nishimura T, Saeki M, Kaminuma O, Matsuoka K, Yonekawa H, Mori A, et al. Existence of antigen-specific immunoglobulin E is not sufficient for allergic nasal eosinophil infiltration in mice. Int Arch Allergy Immunol. 2013; 161(Suppl 2):125–128. PMID: 23711863.
Article19. Corrigan CJ, Kay AB. T cells and eosinophils in the pathogenesis of asthma. Immunol Today. 1992; 13:501–507. PMID: 1361126.
Article20. Kay AB. “Helper” (CD4+) T cells and eosinophils in allergy and asthma. Am Rev Respir Dis. 1992; 145:S22–S26. PMID: 1346489.
Article21. Lever R, Turbitt M, Sanderson A, MacKie R. Immunophenotyping of the cutaneous infiltrate and of the mononuclear cells in the peripheral blood in patients with atopic dermatitis. J Invest Dermatol. 1987; 89:4–7. PMID: 3298446.
Article22. Romagnani S. Human TH1 and TH2 subsets: doubt no more. Immunol Today. 1991; 12:256–257. PMID: 1680337.
Article23. Kouro T, Takatsu K. IL-5- and eosinophil-mediated inflammation: from discovery to therapy. Int Immunol. 2009; 21:1303–1309. PMID: 19819937.
Article24. Murphy KM, Heimberger AB, Loh DY. Induction by antigen of intrathymic apoptosis of CD4+CD8+TCRlo thymocytes in vivo. Science. 1990; 250:1720–1723. PMID: 2125367.25. Watanabe N, Kaminuma O, Kitamura N, Hiroi T. Induced Treg cells augment the Th17-mediated intestinal inflammatory response in a CTLA4-dependent manner. PLoS One. 2016; 11:e0150244. PMID: 26950218.
Article26. Kaminuma O, Ohtomo T, Mori A, Nagakubo D, Hieshima K, Ohmachi Y, et al. Selective down-regulation of Th2 cell-mediated airway inflammation in mice by pharmacological intervention of CCR4. Clin Exp Allergy. 2012; 42:315–325. PMID: 22092376.
Article27. Kaminuma O, Fujimura H, Fushimi K, Nakata A, Sakai A, Chishima S, et al. Dynamics of antigen-specific helper T cells at the initiation of airway eosinophilic inflammation. Eur J Immunol. 2001; 31:2669–2679. PMID: 11536165.
Article28. Pettipher ER, Salter ED, Showell HJ. Effect of in vivo desensitization to leukotriene B4 on eosinophil infiltration in response to C5a in guinea-pig skin. Br J Pharmacol. 1994; 113:117–120. PMID: 7812600.29. Kaminuma O, Mori A, Ogawa K, Nakata A, Kikkawa H, Naito K, et al. Successful transfer of late phase eosinophil infiltration in the lung by infusion of helper T cell clones. Am J Respir Cell Mol Biol. 1997; 16:448–454. PMID: 9115756.
Article30. Nishimura T, Kaminuma O, Saeki M, Kitamura N, Matsuoka K, Yonekawa H, et al. Essential contribution of CD4+ T cells to antigen-induced nasal hyperresponsiveness in experimental allergic rhinitis. PLoS One. 2016; 11:e0146686. PMID: 26752722.
Article31. Ponath PD, Qin S, Post TW, Wang J, Wu L, Gerard NP, et al. Molecular cloning and characterization of a human eotaxin receptor expressed selectively on eosinophils. J Exp Med. 1996; 183:2437–2448. PMID: 8676064.
Article32. Daugherty BL, Siciliano SJ, DeMartino JA, Malkowitz L, Sirotina A, Springer MS. Cloning, expression, and characterization of the human eosinophil eotaxin receptor. J Exp Med. 1996; 183:2349–2354. PMID: 8642344.
Article33. Velazquez JR, Teran LM. Chemokines and their receptors in the allergic airway inflammatory process. Clin Rev Allergy Immunol. 2011; 41:76–88. PMID: 20352527.
Article34. Warringa RA, Schweizer RC, Maikoe T, Kuijper PH, Bruijnzeel PL, Koendermann L. Modulation of eosinophil chemotaxis by interleukin-5. Am J Respir Cell Mol Biol. 1992; 7:631–636. PMID: 1449809.
Article35. Sehmi R, Wardlaw AJ, Cromwell O, Kurihara K, Waltmann P, Kay AB. Interleukin-5 selectively enhances the chemotactic response of eosinophils obtained from normal but not eosinophilic subjects. Blood. 1992; 79:2952–2959. PMID: 1316789.
Article36. Collins PD, Marleau S, Griffiths-Johnson DA, Jose PJ, Williams TJ. Cooperation between interleukin-5 and the chemokine eotaxin to induce eosinophil accumulation in vivo. J Exp Med. 1995; 182:1169–1174. PMID: 7561691.37. Kita H, Abu-Ghazaleh R, Sanderson CJ, Gleich GJ. Effect of steroids on immunoglobulin-induced eosinophil degranulation. J Allergy Clin Immunol. 1991; 87:70–77. PMID: 1991924.
Article38. Hossain M, Okubo Y, Sekiguchi M. Effects of various drugs (staurosporine, herbimycin A, ketotifen, theophylline, FK506 and cyclosporin A) on eosinophil viability. Arerugi. 1994; 43:711–717. PMID: 7520689.39. Mori A, Suko M, Nishizaki Y, Kaminuma O, Kobayashi S, Matsuzaki G, et al. IL-5 production by CD4+ T cells of asthmatic patients is suppressed by glucocorticoids and the immunosuppressants FK506 and cyclosporin A. Int Immunol. 1995; 7:449–457. PMID: 7540862.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- CD4+ T Helper Cells Engineered to Produce IL-10 Reverse Allergen-induced Airway Hyperreactivity and Inflammation
- Regulation and Function of Th17 Cells
- Human Cytotoxic T Lymphocyte-associated Antigen 4-immunoglobulin Fusion Protein Inhibits Airway Inflammation and Hyperresponsiveness in a Murine Model of Asthma
- Signals from the various immune cells in promoting food allergy-induced eosinophilic esophagitis like disease
- Eosinophils and childhood asthma