Prog Med Phys.
2018 Mar;29(1):29-41. 10.14316/pmp.2018.29.1.29.
Preliminary Phantom Experiments to Map Amino Acids and Neurotransmitters Using MRI
- Affiliations
-
- 1Department of Biomedical Engineering, Graduate School, Kyung Hee University, Yongin, Korea.
- 2Biomedical Research Center, Asan Institute for Life Sciences, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 3Department of Radiology, Kyung Hee University Hospital at Gangdong, College of Medicine, Kyung Hee University, Seoul, Korea. ghjahng@gmail.com
- 4Department of Biomedical Engineering, College of Electronics and Information, Kyung Hee University, Yongin, Korea.
- KMID: 2411951
- DOI: http://doi.org/10.14316/pmp.2018.29.1.29
Abstract
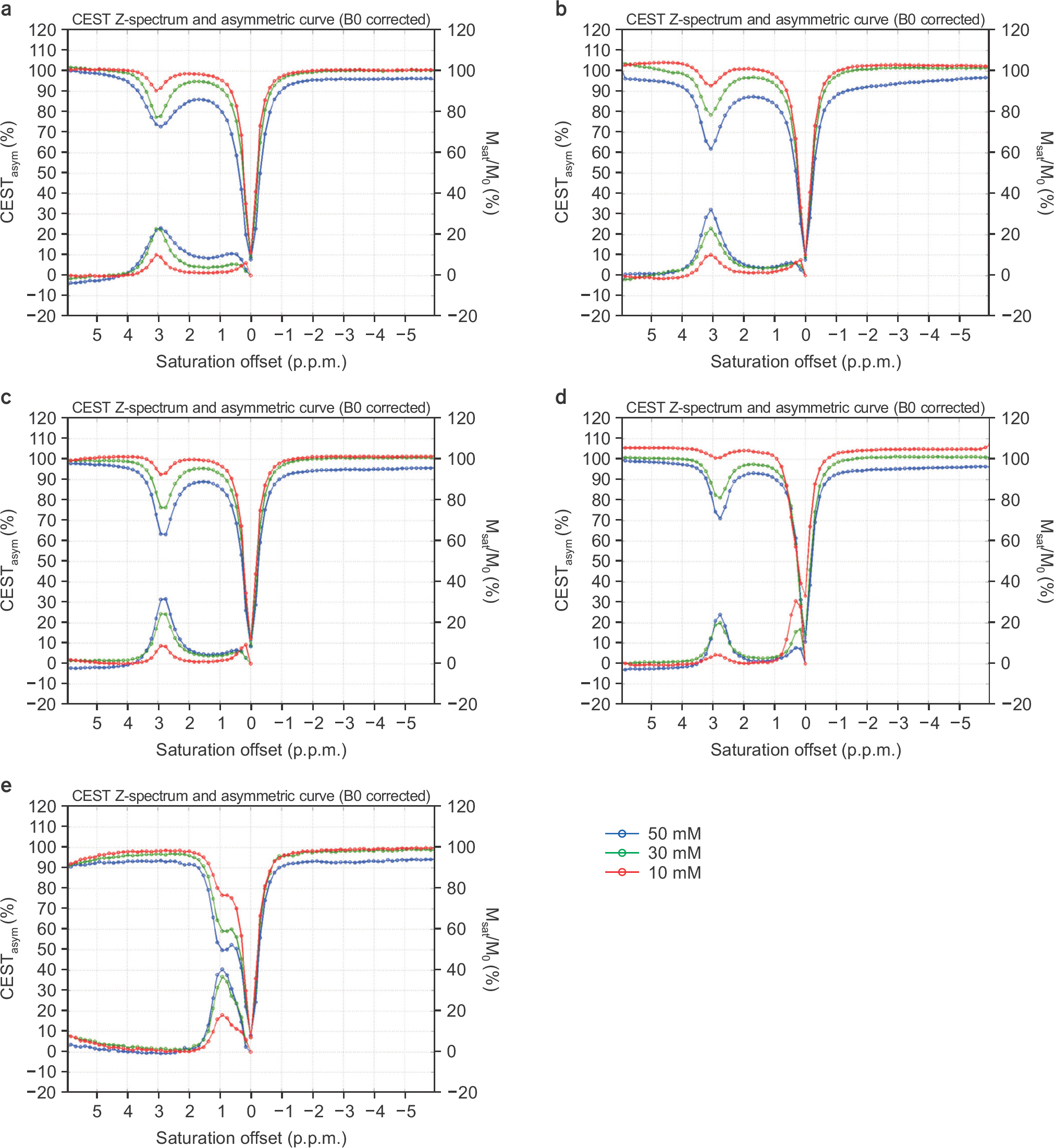
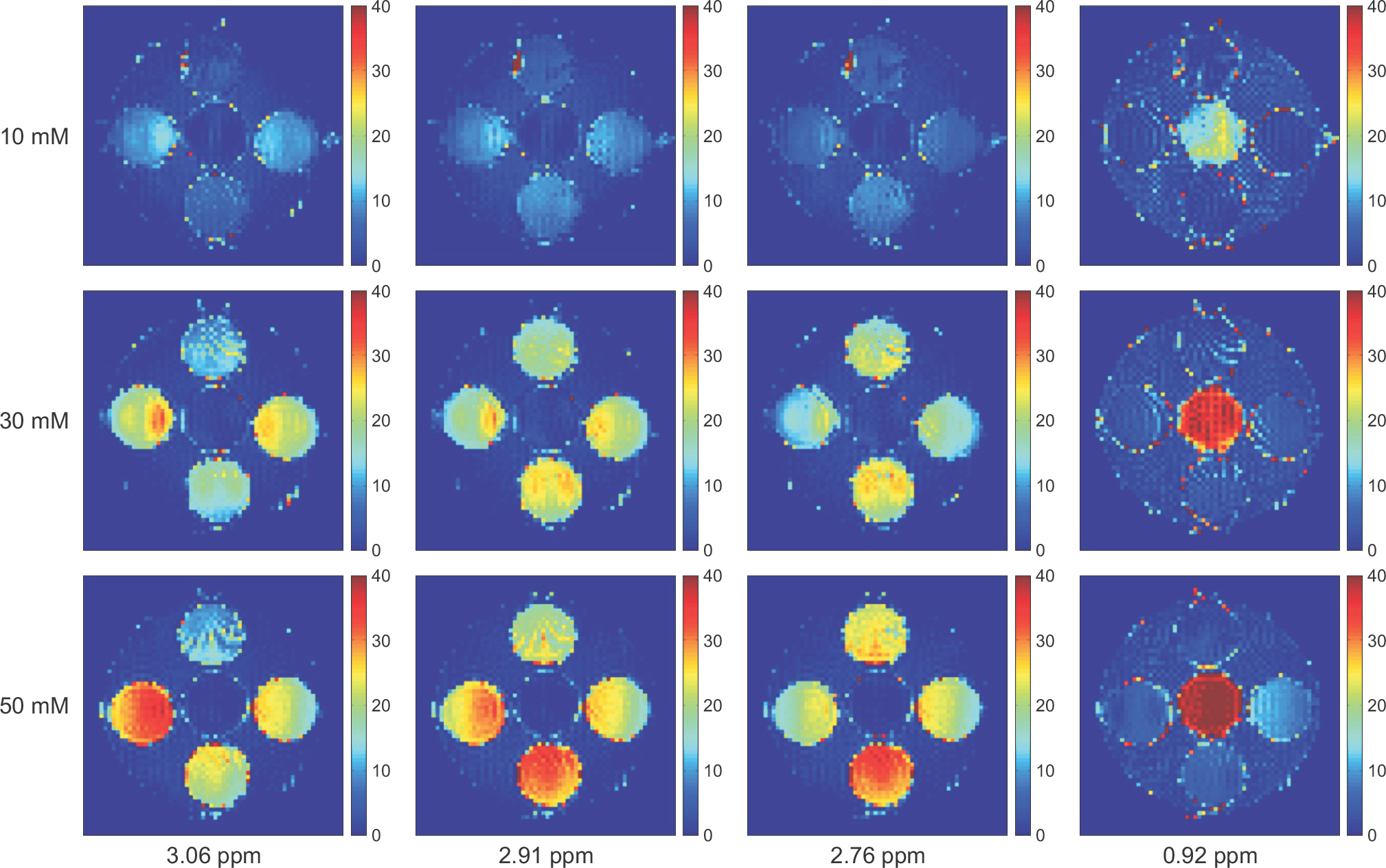
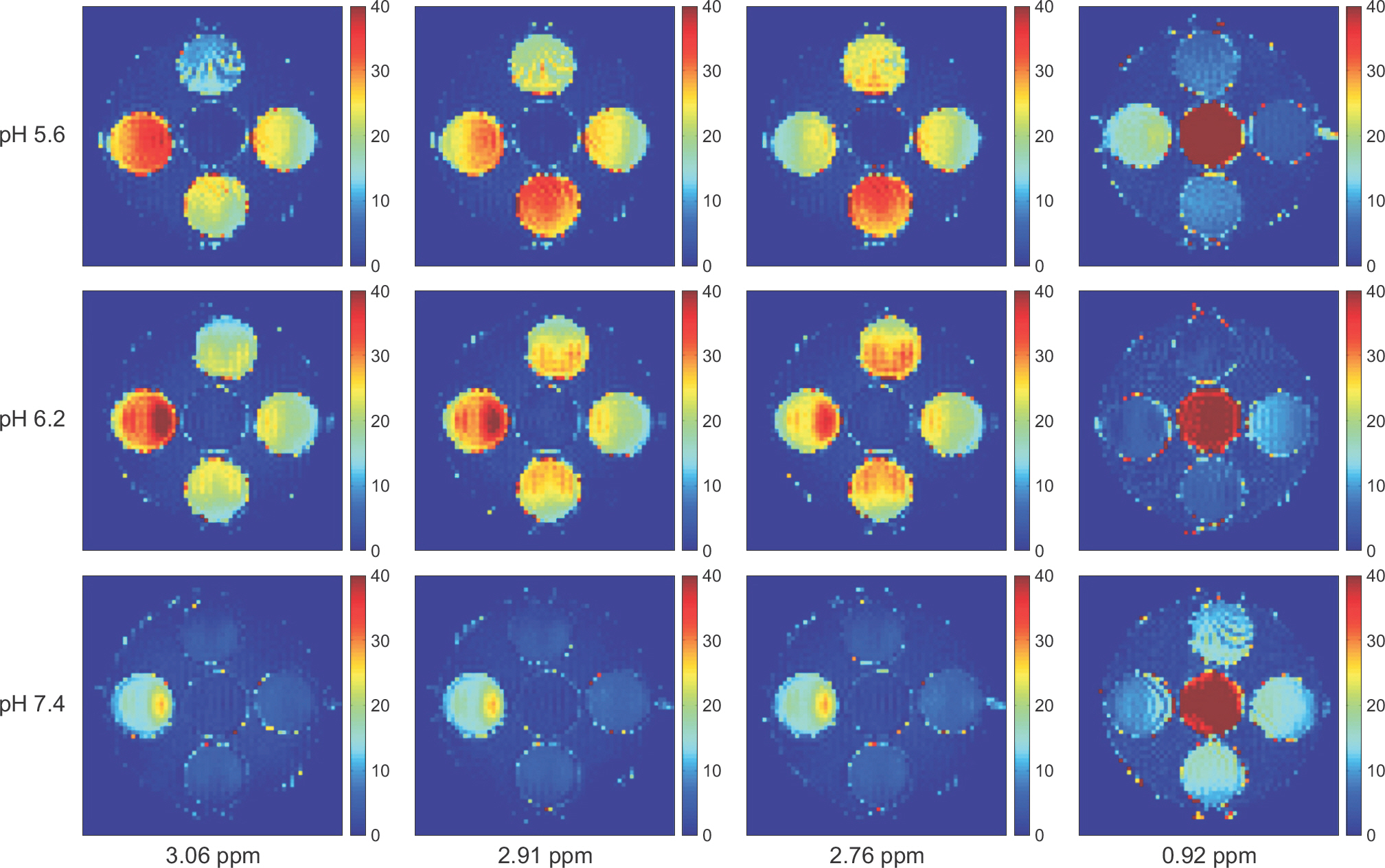
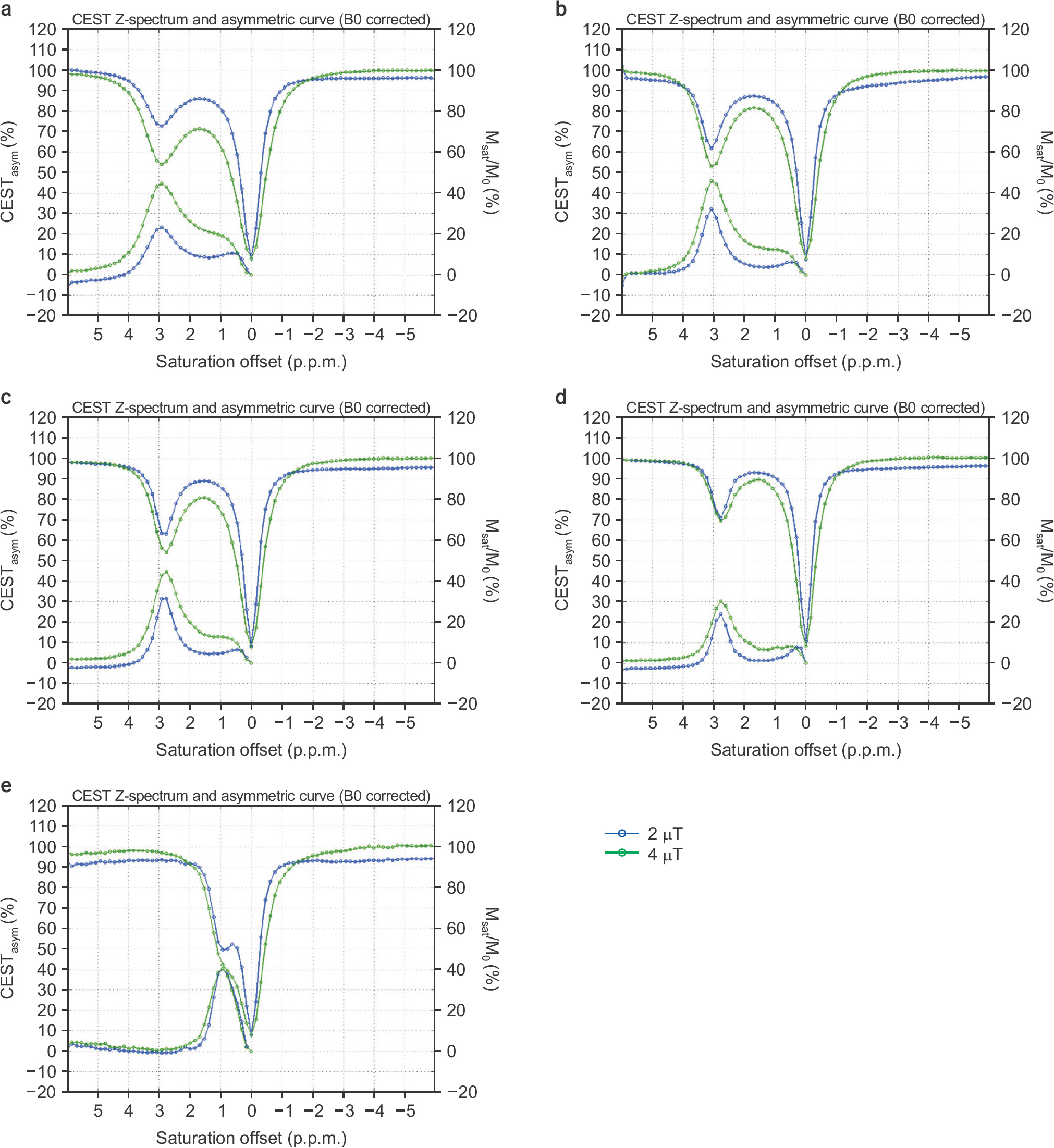
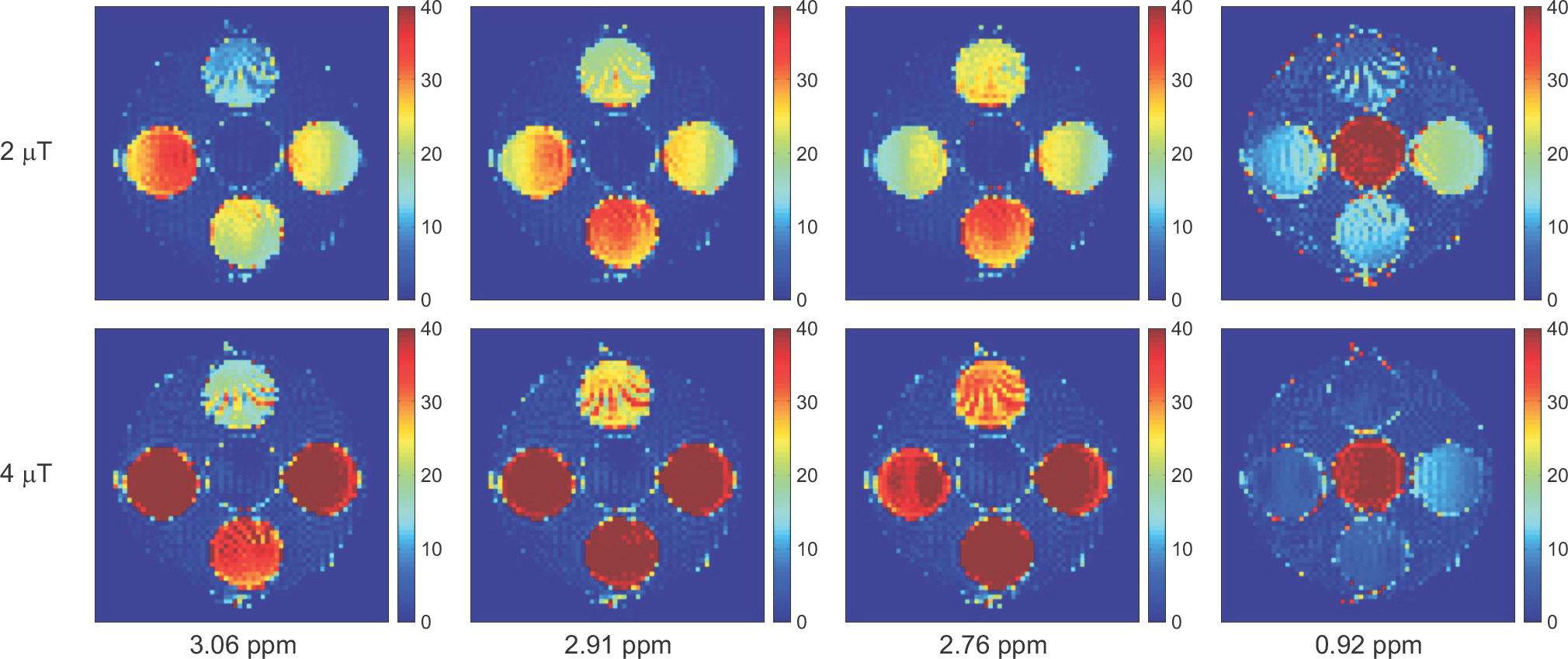
- The objective of this study was to evaluate the chemical exchange saturation transfer (CEST) effect of amino acids and neurotransmitters, which exist in the human brain, depending on the concentration, pH, and amplitude of the saturation radiofrequency field. Phantoms were developed with asparagine (Asn), γ-aminobutyric acid (GABA), glutamate (Glu), glycine (Gly), and myoinositol (MI). Each chemical had three different concentrations of 10, 30, and 50 mM and three different pH values of 5.6, 6.2, and 7.4. Full Z-spectrum CEST images for each phantom were acquired with a continuous-wave radiofrequency (RF) saturation pulse with two different Bâ‚ amplitudes of 2 µT and 4 µT using an animal 9.4T MRI system. A voxel-based CEST asymmetry was mapped to evaluate exchangeable protons based on amide (-NH), amine (-NHâ‚‚), and hydroxyl (-OH) groups for the five target molecules. For all target molecules, the CEST effect was increased with increasing concentration and B1 amplitude; however, the CEST effect with varying pH displayed a different trend depending on the characteristics of the molecule. On CEST asymmetric maps, Glu and MI were well visualized around 3.0 and 0.9 ppm, respectively, and were well separated macroscopically at a pH of 7.4. The exchange rates of Asn, Glu, BABA, and Gly usually decreased with increasing pH. The CEST effect was dependent on the concentration, acidity of the target molecules, and B1 amplitude of the saturation RF pulse. The CEST effect for Asn can be observed in a 9.4T MRI system. The results of this study are based on applying the CEST technique in patients with neurodegenerative diseases when proteins in the brain are increased with disease progression.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Magnetic Resonance Imaging: Historical Overview, Technical Developments, and Clinical Applications
Geon-Ho Jahng, Soonchan Park, Chang-Woo Ryu, Zang-Hee Cho
Prog Med Phys. 2020;31(3):35-53. doi: 10.14316/pmp.2020.31.3.35.
Reference
-
References
1. Guivel-Scharen V, Sinnwell T, Wolff SD, Balaban RS. Detection of proton chemical exchange between metabolites and water in biological tissues. J Magn Reson. 1998; 133:36–45.
Article2. Dagher AP, Aletras A, Choyke P, Balaban RS. Imaging of urea using chemical exchange-dependent saturation transfer at 1.5T. J Magn Reson Imaging. 2000; 12:745–748.
Article3. Cai K, Haris M, Singh A, et al. Magnetic resonance imaging of glutamate. Nat Med. 2012; 18:302–306.
Article4. Ahmed M, Davis J, Aucoin D, et al. Structural conversion of neurotoxic amyloid-beta(1–42) oligomers to fibrils. Nat Struct Mol Biol. 2010; 17:561–567.5. Rauscher S, Baud S, Miao M, Keeley FW, Pomes R. Proline and glycine control protein self-organization into elastomeric or amyloid fibrils. Structure. 2006; 14:1667–1676.
Article6. Harmeier A, Wozny C, Rost BR, et al. Role of amyloid-beta glycine 33 in oligomerization, toxicity, and neuronal plasticity. J Neurosci. 2009; 29:7582–7590.7. Kogan F, Hariharan H, Reddy R. Chemical Exchange Saturation Transfer (CEST) Imaging: Description of Technique and Potential Clinical Applications. Curr Radiol Rep. 2013; 1:102–114.
Article8. Haris M, Singh A, Cai K, et al. MICEST: a potential tool for non-invasive detection of molecular changes in Alzheimer's disease. J Neurosci Methods. 2013; 212:87–93.
Article9. Haris M, Cai K, Singh A, Hariharan H, Reddy R. In vivo mapping of brain myo-inositol. Neuroimage. 2011; 54:2079–2085.
Article10. Oh JH, Kim HG, Woo DC, Jeong HK, Lee SY, Jahng GH. Chemical-Exchange-Saturation-Transfer Magnetic Resonance Imaging to Map Gamma-Aminobutyric Acid, Glutamate, Myoinositol, Glycine, and Asparagine: Phantom Experiments. J Korean Phys Soc. 2017; 70:545–553.
Article11. McVicar N, Li AX, Meakin SO, Bartha R. Imaging chemical exchange saturation transfer (CEST) effects following tumor-selective acidification using lonidamine. NMR Biomed. 2015; 28:566–575.
Article12. McMahon MT, Gilad AA, Zhou J, Sun PZ, Bulte JW, van Zijl PC. Quantifying exchange rates in chemical exchange saturation transfer agents using the saturation time and saturation power dependencies of the magnetization transfer effect on the magnetic resonance imaging signal (QUEST and QUESP): Ph calibration for poly-L-lysine and a starburst dendrimer. Magn Reson Med. 2006; 55:836–847.
Article13. Jahng GH, Oh JH. Physical Modeling of Chemical Exchange Saturation Transfer Imaging. Progress in Medical Physics. 2017; 28:135–143.
Article14. Zaiss M, Bachert P. Chemical exchange saturation transfer (CEST) and MR Z-spectroscopy in vivo: a review of theoretical approaches and methods. Phys Med Biol. 2013; 58:R221–269.15. McVicar N, Li AX, Goncalves DF, et al. Quantitative tissue pH measurement during cerebral ischemia using amine and amide concentration-independent detection (AACID) with MRI. J Cereb Blood Flow Metab. 2014; 34:690–698.
Article16. Zong X, Wang P, Kim SG, Jin T. Sensitivity and source of amine-proton exchange and amide-proton transfer magnetic resonance imaging in cerebral ischemia. Magn Reson Med. 2014; 71:118–132.
Article17. Ward KM, Balaban RS. Determination of pH using water protons and chemical exchange dependent saturation transfer (CEST). Magn Reson Med. 2000; 44:799–802.
Article18. van Zijl PC, Yadav NN. Chemical exchange saturation transfer (CEST): what is in a name and what isn't? Magn Reson Med. 2011; 65:927–948.
Article19. Schmitt B, Zaiss M, Zhou J, Bachert P. Optimization of pulse train presaturation for CEST imaging in clinical scanners. Magn Reson Med. 2011; 65:1620–1629.
Article20. Zu Z, Li K, Janve VA, Does MD, Gochberg DF. Optimizing pulsed-chemical exchange saturation transfer imaging sequences. Magn Reson Med. 2011; 66:1100–1108.
Article21. Tee YK, Khrapitchev AA, Sibson NR, Payne SJ, Chappell MA. Evaluating the use of a continuous approximation for model-based quantification of pulsed chemical exchange saturation transfer (CEST). J Magn Reson. 2012; 222:88–95.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Age estimation from tooth dentine using the racemization of amino acids
- Characteristics of Magnetic Resonance-Based Attenuation Correction Map on Phantom Study in Positron Emission Tomography/Magnetic Resonance Imaging System
- The Effects of Repeated Toluene Exposure on Amino Acid Neurotransmitters in the Rat Brain
- Expression and functional characterization of amino acid transport system L in Saos2 human osteogenic sarcoma cells
- Mechanisms of amino acid sensing in mTOR signaling pathway