Endocrinol Metab.
2017 Dec;32(4):475-484. 10.3803/EnM.2017.32.4.475.
Rebound Feeding in the Wake of Short-Term Suspension of Food Intake Differs in the Presence of Estrous Cycle Peak versus Nadir Levels of Estradiol
- Affiliations
-
- 1Department of Basic Pharmaceutical Sciences, School of Pharmacy, College of Health and Pharmaceutical Sciences, The University of Louisiana Monroe, Monroe, LA, USA. briski@ulm.edu
- KMID: 2406192
- DOI: http://doi.org/10.3803/EnM.2017.32.4.475
Abstract
- BACKGROUND
Short-term interruption of feeding is ordinary in modern life but negatively impacts appetite control and body weight. Estradiol (E) imposes long-term inhibitory tonus on food consumption; however, E influence on energy repletion secondary to food deprivation (FD) is unclear. This study investigated the hypothesis that E signal strength regulates hyperphagic responses to FD of varying duration.
METHODS
Ovariectomized female rats were implanted with E-containing silastic capsules (30 [E-30] or 300 µg [E-300]/mL) to replicate plasma concentrations at cycle nadir versus peak levels.
RESULTS
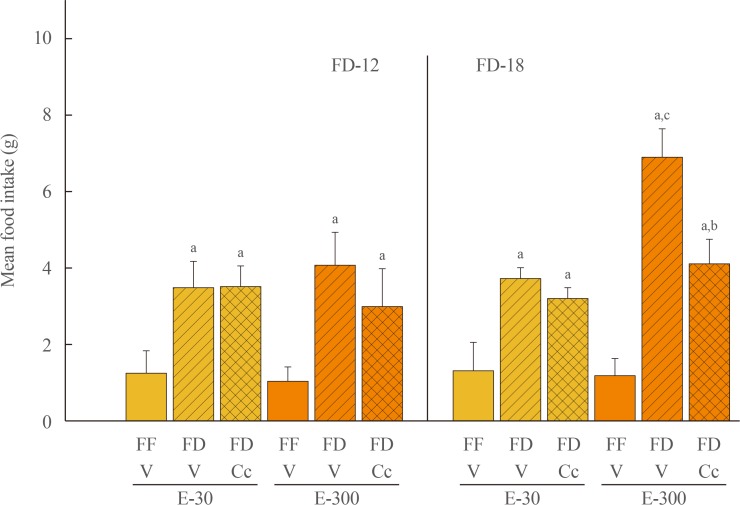
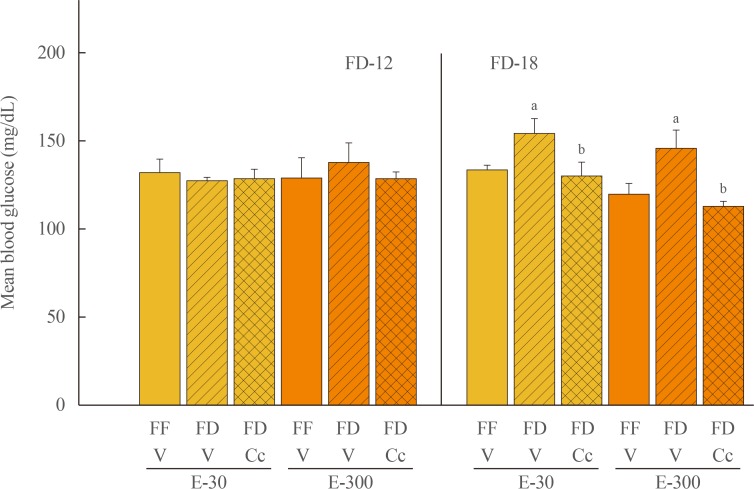
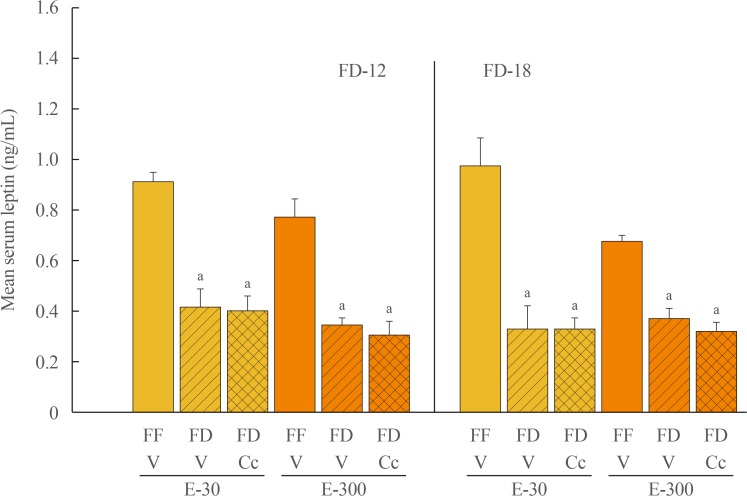
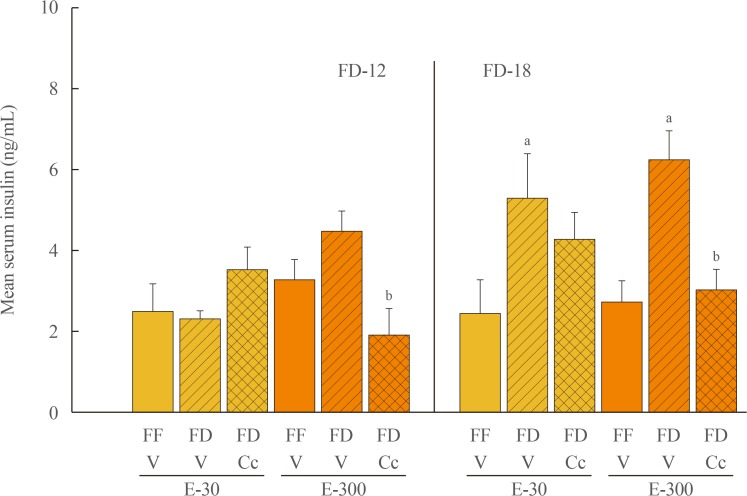
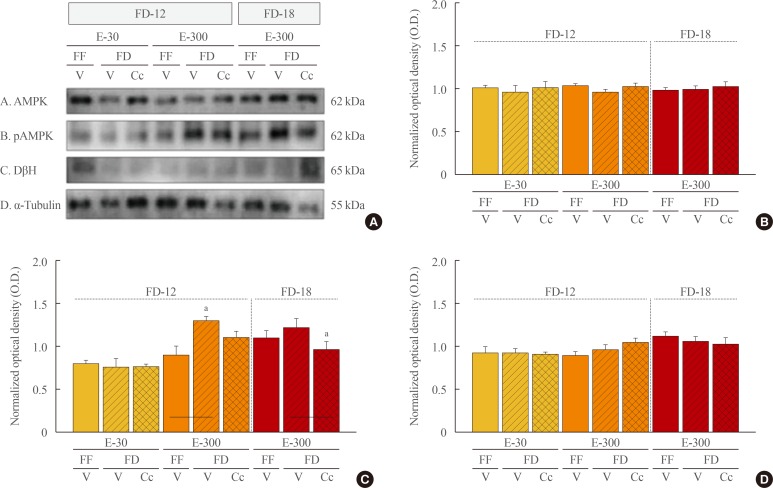
Data show that food intake was increased equally in E-30 and E-300 rats after 12 hours of food deprivation (FD-12); yet, FD of 18 hours (FD-18) amplified refeeding by E-300 versus E-30. Caudal fourth ventricular administration of the 5"²-monophosphate-activated protein kinase (AMPK) inhibitor compound C (Cc) did not modify FD-induced hyperphagia in E-30 (regardless of FD interval) or E-300 animals exposed to FD-12, but diminished refeeding after FD-18 in E-300 rats. Cc-reversible hyperglycemia occurred in refed FD-18 groups. Serum insulin was resistant to FD-12 plus refeeding, but was elevated by AMPK-dependent mechanisms in refed E-300 FD-18 rats; equivalent Cc-insensitive decrements in circulating leptin occurred in all FD groups.
CONCLUSION
Current results show that estrous cycle peak, but not baseline, E levels engage hindbrain AMPK signaling to intensify hyperphagia in response to prolongation of FD. Observations of hindbrain AMPK-dependent hyperglycemia, alongside elevated insulin secretion, in refed rats exposed to FD-18 implicate this sensor in insulin resistance mechanisms of glucose partitioning in response to this metabolic imbalance.
MeSH Terms
-
AMP-Activated Protein Kinases
Animals
Appetite
Body Weight
Capsules
Eating*
Estradiol*
Estrous Cycle*
Female
Food Deprivation
Glucose
Humans
Hyperglycemia
Hyperphagia
Insulin
Insulin Resistance
Leptin
Plasma
Protein Kinases
Rats
Rhombencephalon
AMP-Activated Protein Kinases
Capsules
Estradiol
Glucose
Insulin
Leptin
Protein Kinases
Figure
Reference
-
1. Wade GN, Schneider JE. Metabolic fuels and reproduction in female mammals. Neurosci Biobehav Rev. 1992; 16:235–272. PMID: 1630733.
Article2. Leidy HJ, Campbell WW. The effect of eating frequency on appetite control and food intake: brief synopsis of controlled feeding studies. J Nutr. 2011; 141:154–157. PMID: 21123467.
Article3. Stote KS, Baer DJ, Spears K, Paul DR, Harris GK, Rumpler WV, et al. A controlled trial of reduced meal frequency without caloric restriction in healthy, normal-weight, middle-aged adults. Am J Clin Nutr. 2007; 85:981–988. PMID: 17413096.
Article4. Ibrahim BA, Briski KP. Deferred feeding and body weight responses to short-term interruption of fuel acquisition: impact of estradiol. Horm Metab Res. 2015; 47:611–621. PMID: 25230326.
Article5. Hardie DG. Minireview: the AMP-activated protein kinase cascade. The key sensor of cellular energy status. Endocrinology. 2003; 144:5179–5183. PMID: 12960015.
Article6. Kahn BB, Alquier T, Carling D, Hardie DG. AMP-activated protein kinase: ancient energy gauge provides clues to modern understanding of metabolism. Cell Metab. 2005; 1:15–25. PMID: 16054041.
Article7. Hayes MR, Skibicka KP, Bence KK, Grill HJ. Dorsal hindbrain 5′-adenosine monophosphate-activated protein kinase as an intracellular mediator of energy balance. Endocrinology. 2009; 150:2175–2182. PMID: 19116341.
Article8. Alenazi FS, Ibrahim BA, Al-Hamami H, Shakiya M, Briski KP. Role of estradiol in intrinsic hindbrain AMPK regulation of hypothalamic AMPK, metabolic neuropeptide, and norepinephrine activity and food intake in the female rat. Neuroscience. 2016; 314:35–46. PMID: 26628404.
Article9. Ibrahim BA, Tamrakar P, Gujar AD, Cherian AK, Briski KP. Caudal fourth ventricular administration of the AMPK activator 5-aminoimidazole-4-carboxamide-riboside regulates glucose and counterregulatory hormone profiles, dorsal vagal complex metabolosensory neuron function, and hypothalamic Fos expression. J Neurosci Res. 2013; 91:1226–1238. PMID: 23825033.
Article10. Thammacharoen S, Lutz TA, Geary N, Asarian L. Hindbrain administration of estradiol inhibits feeding and activates estrogen receptor-alpha-expressing cells in the nucleus tractus solitarius of ovariectomized rats. Endocrinology. 2008; 149:1609–1617. PMID: 18096668.11. Briski KP, Cherian AK, Genabai NK, Vavaiya KV. In situ coexpression of glucose and monocarboxylate transporter mRNAs in metabolic-sensitive caudal dorsal vagal complex catecholaminergic neurons: transcriptional reactivity to insulin-induced hypoglycemia and caudal hindbrain glucose or lactate repletion during insulin-induced hypoglycemia. Neuroscience. 2009; 164:1152–1160. PMID: 19744543.
Article12. Cherian AK, Briski KP. A2 noradrenergic nerve cell metabolic transducer and nutrient transporter adaptation to hypoglycemia: impact of estrogen. J Neurosci Res. 2012; 90:1347–1358. PMID: 22431334.13. Butcher RL, Collins WE, Fugo NW. Plasma concentration of LH, FSH, prolactin, progesterone and estradiol-17beta throughout the 4-day estrous cycle of the rat. Endocrinology. 1974; 94:1704–1708. PMID: 4857496.14. Goodman RL. A quantitative analysis of the physiological role of estradiol and progesterone in the control of tonic and surge secretion of luteinizing hormone in the rat. Endocrinology. 1978; 102:142–150. PMID: 570477.
Article15. ter Haar MB. Circadian and estrual rhythms in food intake in the rat. Horm Behav. 1972; 3:213–219. PMID: 4681745.16. Asarian L, Geary N. Cyclic estradiol treatment normalizes body weight and restores physiological patterns of spontaneous feeding and sexual receptivity in ovariectomized rats. Horm Behav. 2002; 42:461–471. PMID: 12488112.
Article17. Asarian L, Geary N. Modulation of appetite by gonadal steroid hormones. Philos Trans R Soc Lond B Biol Sci. 2006; 361:1251–1263. PMID: 16815802.
Article18. Briski KP, Marshall ES, Sylvester PW. Effects of estradiol on glucoprivic transactivation of catecholaminergic neurons in the female rat caudal brainstem. Neuroendocrinology. 2001; 73:369–377. PMID: 11408778.
Article19. Briski KP, Alenazi FSH, Shakya M, Sylvester PW. Hindbrain A2 noradrenergic neuron adenosine 5′-monophosphate-activated protein kinase activation, upstream kinase/phosphorylase protein expression, and receptivity to hormone and fuel reporters of short-term food deprivation are regulated by estradiol. J Neurosci Res. 2017; 95:1427–1437. PMID: 27618227.
Article20. Baskin DG, Figlewicz Lattemann D, Seeley RJ, Woods SC, Porte D Jr, Schwartz MW. Insulin and leptin: dual adiposity signals to the brain for the regulation of food intake and body weight. Brain Res. 1999; 848:114–123. PMID: 10612703.
Article21. Woods SC, Lutz TA, Geary N, Langhans W. Pancreatic signals controlling food intake; insulin, glucagon and amylin. Philos Trans R Soc Lond B Biol Sci. 2006; 361:1219–1235. PMID: 16815800.
Article22. Klok MD, Jakobsdottir S, Drent ML. The role of leptin and ghrelin in the regulation of food intake and body weight in humans: a review. Obes Rev. 2007; 8:21–34. PMID: 17212793.
Article23. Briski KP, Nedungadi TP. Adaptation of feeding and counter-regulatory hormone responses to intermediate insulin-induced hypoglycaemia in the ovariectomized female rat: effects of oestradiol. J Neuroendocrinol. 2009; 21:578–585. PMID: 19500228.24. Kale AY, Paranjape SA, Briski KP. I.c.v. administration of the nonsteroidal glucocorticoid receptor antagonist, CP-472555, prevents exacerbated hypoglycemia during repeated insulin administration. Neuroscience. 2006; 140:555–565. PMID: 16626867.
Article25. Butera PC. Estradiol and the control of food intake. Physiol Behav. 2010; 99:175–180. PMID: 19555704.
Article26. Giles ED, Jackman MR, Johnson GC, Schedin PJ, Houser JL, MacLean PS. Effect of the estrous cycle and surgical ovariectomy on energy balance, fuel utilization, and physical activity in lean and obese female rats. Am J Physiol Regul Integr Comp Physiol. 2010; 299:R1634–R1642. PMID: 20926768.
Article27. Chen JQ, Brown TR, Russo J. Regulation of energy metabolism pathways by estrogens and estrogenic chemicals and potential implications in obesity associated with increased exposure to endocrine disruptors. Biochim Biophys Acta. 2009; 1793:1128–1143. PMID: 19348861.
Article28. Everett JW, Sawyer CH, Markee JE. A neurogenic timing factor in control of the ovulatory discharge of luteinizing hormone in the cyclic rat. Endocrinology. 1949; 44:234–250. PMID: 18115074.29. Everett JW, Sawyer CH. A 24-hour periodicity in the “LH-release apparatus” of female rats, disclosed by barbiturate sedation. Endocrinology. 1950; 47:198–218. PMID: 14793479.30. Legan SJ, Coon GA, Karsch FJ. Role of estrogen as initiator of daily LH surges in the ovariectomized rat. Endocrinology. 1975; 96:50–56. PMID: 1109905.31. Legan SJ, Karsch FJ. A daily signal for the LH surge in the rat. Endocrinology. 1975; 96:57–62. PMID: 1167356.
Article32. Kalra SP. Mandatory neuropeptide-steroid signaling for the preovulatory luteinizing hormone-releasing hormone discharge. Endocr Rev. 1993; 14:507–538. PMID: 8262004.
Article33. Edwards CM, Abusnana S, Sunter D, Murphy KG, Ghatei MA, Bloom SR. The effect of the orexins on food intake: comparison with neuropeptide Y, melanin-concentrating hormone and galanin. J Endocrinol. 1999; 160:R7–R12. PMID: 10077743.
Article34. Kalra SP, Kalra PS. Neuropeptide Y: a physiological orexigen modulated by the feedback action of ghrelin and leptin. Endocrine. 2003; 22:49–56. PMID: 14610298.
Article35. Leibowitz SF. Regulation and effects of hypothalamic galanin: relation to dietary fat, alcohol ingestion, circulating lipids and energy homeostasis. Neuropeptides. 2005; 39:327–332. PMID: 15944030.
Article36. Sahu A. Leptin decreases food intake induced by melanin-concentrating hormone (MCH), galanin (GAL) and neuropeptide Y (NPY) in the rat. Endocrinology. 1998; 139:4739–4742. PMID: 9794487.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Menstruation and Sleep
- Regulation of Estrogen Receptor mRNA in Rat Anterior Pituitary Gland
- Establishment of an In Vivo Rat Model to Investigate Female Vaginal Arousal Response
- Serum leptin concentrations, leptin mRNA expression, and food intake during the estrous cycle in rats
- Expression of Luteinizing Hormone (LH) and Its Receptor Gene in Uterus from Cycling Rats