Nutr Res Pract.
2017 Feb;11(1):17-24. 10.4162/nrp.2017.11.1.17.
Gelidium amansii extract ameliorates obesity by down-regulating adipogenic transcription factors in diet-induced obese mice
- Affiliations
-
- 1Department of Food Science and Nutrition, Pusan National University, 2, Busandaehak-ro 63beon-gil, Geumjeong-ju, Busan 46241, Korea. hanjs@pusan.ac.kr
- 2Research Institute of Ecology for the Elderly, Pusan National University, Busan 46241, Korea.
- 3Seojin Biotech Co., Ltd., Yongin, Gyeonggi 17015, Korea.
- KMID: 2395289
- DOI: http://doi.org/10.4162/nrp.2017.11.1.17
Abstract
- BACKGROUND/OBJECTIVES
In this study, we investigated whether Gelidium amansii extract (GAE) ameliorates obesity in diet-induced obese (DIO) mice.
MATERIALS/METHODS
The mice were maintained on a high-fat diet (HD) for 5 weeks to generate the DIO mouse model. And then mice fed HD plus 0.5% (GAE1), 1% (GAE2) or 2% (GAE3) for 8 weeks.
RESULTS
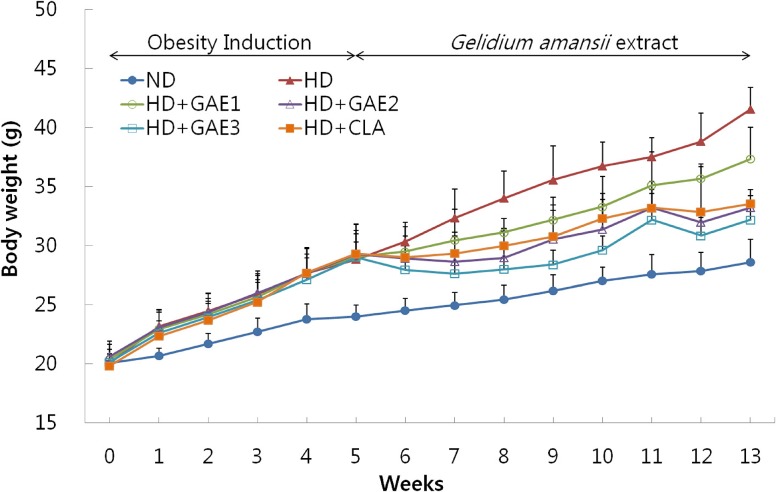
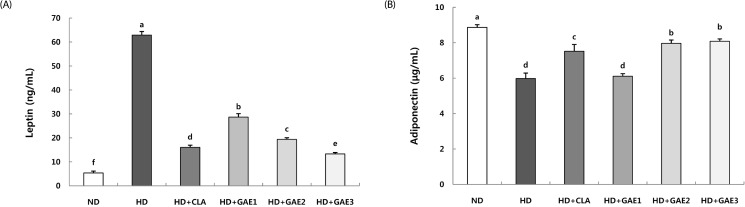
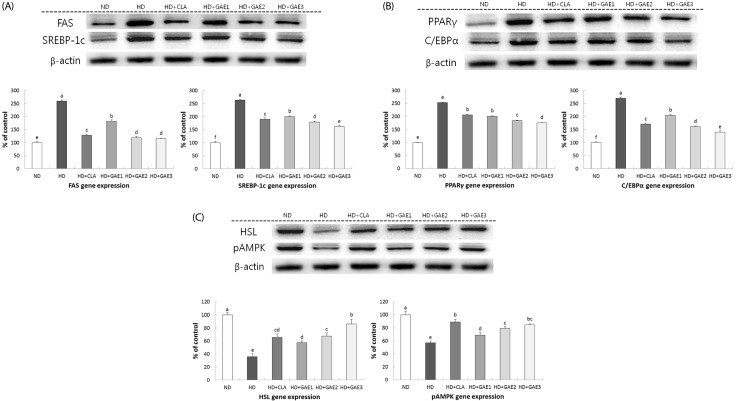
After the experimental period, GAE-supplemented groups were significantly lower than the HD group in body weight gain and liver weight. GAE supplemented groups were significantly lower than the HD group in both epididymal and mesenteric adipose tissue mass. The plasma leptin level was significantly higher in the HD group than in GAE-supplemented groups. The leptin level of HD+GAE3 group was significantly lower than that of the HD+conjugated linoleic acid (CLA) group. In contrast, plasma adiponectin level of the HD group was significantly lower than those of HD+GAE2 and HD+GAE3 groups. The expression levels of adipogenic proteins such as fatty acid synthase, sterol regulatory element-binding protein-1c, peroxisome proliferator-activated receptor γ, and CCAAT/enhancer binding protein α in the GAE supplemented groups were significantly decreased than those in HD group, respectively. In addition, the expression levels of HD+GAE2 and HD+GAE3 groups are significantly decreased compared to those of HD+CLA group. On the contrary, the expression levels of hormone-sensitive lipase and phospho-AMP-activated protein kinase, proteins associated with lipolysis, were significantly increased in the GAE supplemented groups compared to those in the HD group. HD+GAE3 group showed the highest level among the GAE supplemented groups.
CONCLUSIONS
These results suggested that GAE supplementation stimulated the expressions of lipid metabolic factors and reduced weight gain in HD-fed C57BL/6J obese mice.
Keyword
MeSH Terms
-
Adipogenesis
Adiponectin
Adipose Tissue
Animals
Body Weight
Carrier Proteins
Diet, High-Fat
Leptin
Linoleic Acid
Lipolysis
Liver
Mice
Mice, Obese*
Obesity*
Peroxisomes
Plasma
Protein Kinases
Sterol Esterase
Transcription Factors*
Weight Gain
Adiponectin
Carrier Proteins
Leptin
Linoleic Acid
Protein Kinases
Sterol Esterase
Transcription Factors
Figure
Cited by 1 articles
-
Anti-inflammatory effects of Agar free-Gelidium amansii (GA) extracts in high-fat diet-induced obese mice
Yunkyoung Lee, Hyunhee Oh, Myoungsook Lee
Nutr Res Pract. 2018;12(6):479-485. doi: 10.4162/nrp.2018.12.6.479.
Reference
-
1. Nieves JW, Komar L, Cosman F, Lindsay R. Calcium potentiates the effect of estrogen and calcitonin on bone mass: review and analysis. Am J Clin Nutr. 1998; 67:18–24. PMID: 9440370.
Article3. Wang T, Wang Y, Kontani Y, Kobayashi Y, Sato Y, Mori N, Yamashita H. Evodiamine improves diet-induced obesity in a uncoupling protein-1-independent manner: involvement of antiadipogenic mechanism and extracellularly regulated kinase/mitogen-activated protein kinase signaling. Endocrinology. 2008; 149:358–366. PMID: 17884939.
Article4. Choi H, Eo H, Park K, Jin M, Park EJ, Kim SH, Park JE, Kim S. A water-soluble extract from Cucurbita moschata shows anti-obesity effects by controlling lipid metabolism in a high fat diet-induced obesity mouse model. Biochem Biophys Res Commun. 2007; 359:419–425. PMID: 17548058.5. Giri S, Rattan R, Haq E, Khan M, Yasmin R, Won JS, Key L, Singh AK, Singh I. AICAR inhibits adipocyte differentiation in 3T3L1 and restores metabolic alterations in diet-induced obesity mice model. Nutr Metab (Lond). 2006; 3:31. PMID: 16901342.
Article6. Rosen ED, Walkey CJ, Puigserver P, Spiegelman BM. Transcriptional regulation of adipogenesis. Genes Dev. 2000; 14:1293–1307. PMID: 10837022.
Article7. Sztalryd C, Kraemer FB. Regulation of hormone-sensitive lipase in streptozotocin-induced diabetic rats. Metabolism. 1995; 44:1391–1396. PMID: 7476323.
Article8. Lefterova MI, Lazar MA. New developments in adipogenesis. Trends Endocrinol Metab. 2009; 20:107–114. PMID: 19269847.
Article9. Wasan KM, Looije NA. Emerging pharmacological approaches to the treatment of obesity. J Pharm Pharm Sci. 2005; 8:259–271. PMID: 16124937.10. Lee YS, Cha BY, Saito K, Choi SS, Wang XX, Choi BK, Yonezawa T, Teruya T, Nagai K, Woo JT. Effects of a Citrus depressa Hayata (shiikuwasa) extract on obesity in high-fat diet-induced obese mice. Phytomedicine. 2011; 18:648–654. PMID: 21216135.
Article11. Hofbauer KG, Nicholson JR. Pharmacotherapy of obesity. Exp Clin Endocrinol Diabetes. 2006; 114:475–484. PMID: 17115344.
Article13. Choi KM, Lee YS, Shin DM, Lee S, Yoo KS, Lee MK, Lee JH, Kim SY, Lee YM, Hong JT, Yun YP, Yoo HS. Green tomato extract attenuates high-fat-diet-induced obesity through activation of the AMPK pathway in C57BL/6 mice. J Nutr Biochem. 2013; 24:335–342. PMID: 22974972.
Article14. Kadam SU, Prabhasankar P. Marine foods as functional ingredients in bakery and pasta products. Food Res Int. 2010; 43:1975–1980.
Article15. Yan X, Nagata T, Fan X. Antioxidative activities in some common seaweeds. Plant Foods Hum Nutr. 1998; 52:253–262. PMID: 9950086.16. Fu YW, Hou WY, Yeh ST, Li CH, Chen JC. The immunostimulatory effects of hot-water extract of Gelidium amansii via immersion, injection and dietary administrations on white shrimp Litopenaeus vannamei and its resistance against Vibrio alginolyticus. Fish Shellfish Immunol. 2007; 22:673–685. PMID: 17194604.
Article17. Yuan H, Song J, Li X, Li N, Dai J. Immunomodulation and antitumor activity of kappa-carrageenan oligosaccharides. Cancer Lett. 2006; 243:228–234. PMID: 16410037.18. Kang MC, Kang N, Kim SY, Lima IS, Ko SC, Kim YT, Kim YB, Jeung HD, Choi KS, Jeon YJ. Popular edible seaweed, Gelidium amansii prevents against diet-induced obesity. Food Chem Toxicol. 2016; 90:181–187. PMID: 26911551.
Article19. Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972; 18:499–502. PMID: 4337382.
Article20. Folch J, Lees M, Sloane Stanley GH. A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem. 1957; 226:497–509. PMID: 13428781.
Article21. Yamashita Y, Okabe M, Natsume M, Ashida H. Prevention mechanisms of glucose intolerance and obesity by cacao liquor procyanidin extract in high-fat diet-fed C57BL/6 mice. Arch Biochem Biophys. 2012; 527:95–104. PMID: 22465028.
Article22. Lee J, Chae K, Ha J, Park BY, Lee HS, Jeong S, Kim MY, Yoon M. Regulation of obesity and lipid disorders by herbal extracts from Morus alba, Melissa officinalis, and Artemisia capillaris in high-fat diet-induced obese mice. J Ethnopharmacol. 2008; 115:263–270. PMID: 18023310.
Article23. Seo MJ, Lee OH, Choi HS, Lee BY. Extract from edible red seaweed (Gelidium amansii) inhibits lipid accumulation and ROS production during differentiation in 3T3-L1 cells. Prev Nutr Food Sci. 2012; 17:129–135. PMID: 24471074.
Article24. Hokanson JE, Austin MA. Plasma triglyceride level is a risk factor for cardiovascular disease independent of high-density lipoprotein cholesterol level: a meta-analysis of population-based prospective studies. J Cardiovasc Risk. 1996; 3:213–219. PMID: 8836866.
Article25. Miller M, Stone NJ, Ballantyne C, Bittner V, Criqui MH, Ginsberg HN, Goldberg AC, Howard WJ, Jacobson MS, Kris-Etherton PM, Lennie TA, Levi M, Mazzone T, Pennathur S. American Heart Association Clinical Lipidology, Thrombosis, and Prevention Committee of the Council on Nutrition, Physical Activity, and Metabolism. Council on Arteriosclerosis, Thrombosis and Vascular Biology; Council on Cardiovascular Nursing. Council on the Kidney in Cardiovascular Disease. Triglycerides and cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2011; 123:2292–2333. PMID: 21502576.26. den Boer M, Voshol PJ, Kuipers F, Havekes LM, Romijn JA. Hepatic steatosis: a mediator of the metabolic syndrome. Lessons from animal models. Arterioscler Thromb Vasc Biol. 2004; 24:644–649. PMID: 14715643.
Article27. Ferri M, Bin S, Vallini V, Fava F, Michelini E, Roda A, Minnucci G, Bucchi G, Tassoni A. Recovery of polyphenols from red grape pomace and assessment of their antioxidant and anti-cholesterol activities. N Biotechnol. 2016; 33:338–344. PMID: 26705904.
Article28. He ML, Wang Y, You JS, Mir PS, McAllister TA. Effect of a seaweed extract on fatty acid accumulation and glycerol-3-phosphate dehydrogenase activity in 3T3-L1 adipocytes. Lipids. 2009; 44:125–132. PMID: 18982374.
Article29. Körner A, Blüher S, Kapellen T, Garten A, Klammt J, Kratzsch J, Kiess W. Obesity in childhood and adolescence: a review in the interface between adipocyte physiology and clinical challenges. Hormones (Athens). 2005; 4:189–199. PMID: 16613817.
Article30. Staiger H, Tschritter O, Machann J, Thamer C, Fritsche A, Maerker E, Schick F, Häring HU, Stumvoll M. Relationship of serum adiponectin and leptin concentrations with body fat distribution in humans. Obes Res. 2003; 11:368–372. PMID: 12634431.
Article31. Jéquier E. Leptin signaling, adiposity, and energy balance. Ann N Y Acad Sci. 2002; 967:379–388. PMID: 12079865.32. Lee Y, Wang MY, Kakuma T, Wang ZW, Babcock E, McCorkle K, Higa M, Zhou YT, Unger RH. Liporegulation in diet-induced obesity. The antisteatotic role of hyperleptinemia. J Biol Chem. 2001; 276:5629–5635. PMID: 11096093.33. Wu X, Motoshima H, Mahadev K, Stalker TJ, Scalia R, Goldstein BJ. Involvement of AMP-activated protein kinase in glucose uptake stimulated by the globular domain of adiponectin in primary rat adipocytes. Diabetes. 2003; 52:1355–1363. PMID: 12765944.
Article34. Hu E, Liang P, Spiegelman BM. AdipoQ is a novel adipose-specific gene dysregulated in obesity. J Biol Chem. 1996; 271:10697–10703. PMID: 8631877.
Article35. Siegrist M, Rank M, Wolfarth B, Langhof H, Haller B, Koenig W, Halle M. Leptin, adiponectin, and short-term and long-term weight loss after a lifestyle intervention in obese children. Nutrition. 2013; 29:851–857. PMID: 23422541.
Article36. Bruun JM, Lihn AS, Verdich C, Pedersen SB, Toubro S, Astrup A, Richelsen B. Regulation of adiponectin by adipose tissue-derived cytokines: in vivo and in vitro investigations in humans. Am J Physiol Endocrinol Metab. 2003; 285:E527–E533. PMID: 12736161.37. Park J, Rho HK, Kim KH, Choe SS, Lee YS, Kim JB. Overexpression of glucose-6-phosphate dehydrogenase is associated with lipid dysregulation and insulin resistance in obesity. Mol Cell Biol. 2005; 25:5146–5157. PMID: 15923630.
Article38. Schoonjans K, Staels B, Auwerx J. The peroxisome proliferator activated receptors (PPARS) and their effects on lipid metabolism and adipocyte differentiation. Biochim Biophys Acta. 1996; 1302:93–109. PMID: 8695669.
Article39. Rosen ED, Hsu CH, Wang X, Sakai S, Freeman MW, Gonzalez FJ, Spiegelman BM. C/EBPalpha induces adipogenesis through PPARgamma: a unified pathway. Genes Dev. 2002; 16:22–26. PMID: 11782441.40. Kamei R, Kitagawa Y, Kadokura M, Hattori F, Hazeki O, Ebina Y, Nishihara T, Oikawa S. Shikonin stimulates glucose uptake in 3T3-L1 adipocytes via an insulin-independent tyrosine kinase pathway. Biochem Biophys Res Commun. 2002; 292:642–651. PMID: 11922615.
Article41. Kim JB, Wright HM, Wright M, Spiegelman BM. ADD1/SREBP1 activates PPARgamma through the production of endogenous ligand. Proc Natl Acad Sci U S A. 1998; 95:4333–4337. PMID: 9539737.42. Evans RM, Barish GD, Wang YX. PPARs and the complex journey to obesity. Nat Med. 2004; 10:355–361. PMID: 15057233.
Article43. Wang S, Moustaid-Moussa N, Chen L, Mo H, Shastri A, Su R, Bapat P, Kwun I, Shen CL. Novel insights of dietary polyphenols and obesity. J Nutr Biochem. 2014; 25:1–18. PMID: 24314860.
Article44. Mohammed A, Al-Numair KS, Balakrishnan A. Docking studies on the interaction of flavonoids with fat mass and obesity associated protein. Pak J Pharm Sci. 2015; 28:1647–1653. PMID: 26408884.45. Zhang BB, Zhou G, Li C. AMPK: an emerging drug target for diabetes and the metabolic syndrome. Cell Metab. 2009; 9:407–416. PMID: 19416711.
Article46. Jung HA, Jung HJ, Jeong HY, Kwon HJ, Ali MY, Choi JS. Phlorotannins isolated from the edible brown alga Ecklonia stolonifera exert anti-adipogenic activity on 3T3-L1 adipocytes by downregulating C/EBPα and PPARγ. Fitoterapia. 2014; 92:260–269. PMID: 24334103.
Article47. Vingtdeux V, Chandakkar P, Zhao H, Davies P, Marambaud P. Small-molecule activators of AMP-activated protein kinase (AMPK), RSVA314 and RSVA405, inhibit adipogenesis. Mol Med. 2011; 17:1022–1030. PMID: 21647536.
Article48. Hardie DG. AMPK: a key regulator of energy balance in the single cell and the whole organism. Int J Obes (Lond). 2008; 32(Suppl 4):S7–S12. PMID: 18719601.
Article49. Watt MJ, Holmes AG, Pinnamaneni SK, Garnham AP, Steinberg GR, Kemp BE, Febbraio MA. Regulation of HSL serine phosphorylation in skeletal muscle and adipose tissue. Am J Physiol Endocrinol Metab. 2006; 290:E500–E508. PMID: 16188906.
Article50. Bezaire V, Mairal A, Ribet C, Lefort C, Girousse A, Jocken J, Laurencikiene J, Anesia R, Rodriguez AM, Ryden M, Stenson BM, Dani C, Ailhaud G, Arner P, Langin D. Contribution of adipose triglyceride lipase and hormone-sensitive lipase to lipolysis in hMADS adipocytes. J Biol Chem. 2009; 284:18282–18291. PMID: 19433586.
Article51. Son Y, Nam JS, Jang MK, Jung IA, Cho SI, Jung MH. Antiobesity activity of Vigna nakashimae extract in high-fat diet-induced obesity. Biosci Biotechnol Biochem. 2013; 77:332–338. PMID: 23391927.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Anti-inflammatory effects of Agar free-Gelidium amansii (GA) extracts in high-fat diet-induced obese mice
- The anti-obesity effect of Lethariella cladonioides in 3T3-L1 cells and obese mice
- Fumigaclavine C attenuates adipogenesis in 3T3-L1 adipocytes and ameliorates lipid accumulation in high-fat diet-induced obese mice
- A Pilot Study on the Effect of Cannabis Extract on Weight Loss in Diet-induced Obese Mice
- The Effects of the Sasa Borealis Leaves Extract on Plasma Adiponectin, Resistin, C-Reactive Protein and Homocysteine Levels in High Fat Diet-Induced Obese C57/BL6J Mice