Korean J Physiol Pharmacol.
2017 Nov;21(6):591-598. 10.4196/kjpp.2017.21.6.591.
The bifunctional effect of propofol on thromboxane agonist (U46619)-induced vasoconstriction in isolated human pulmonary artery
- Affiliations
-
- 1Department of Anesthesiology, Guangdong Second Provincial General Hospital, Guangzhou 510317, China.
- 2Department of Anesthesia, Guangdong General Hospital, Guangdong Academy of Medical Sciences, Guangzhou 510080, China. cuijianxiu@163.com
- 3Medical Research Center of Guangdong General Hospital, Guangdong Academy of Medical Sciences, Guangdong Provincial Cardiovascular Institute, Guangzhou 510080, China.
- 4Surgical Training Physician, Guangdong General Hospital, Guangdong Academy of Medical Sciences, Guangzhou 510080, China.
- KMID: 2395253
- DOI: http://doi.org/10.4196/kjpp.2017.21.6.591
Abstract
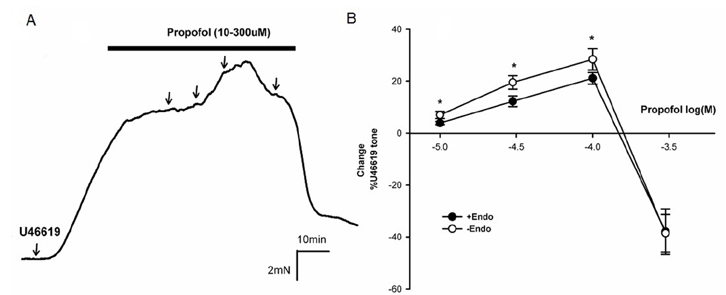
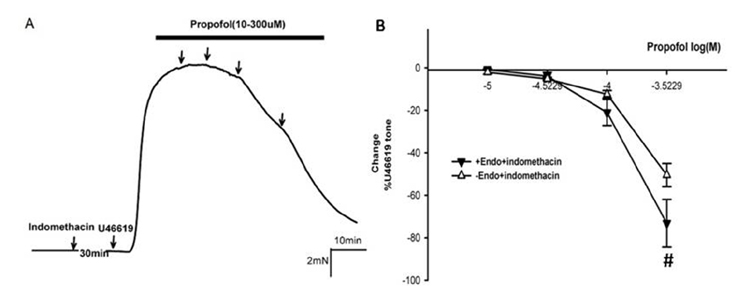
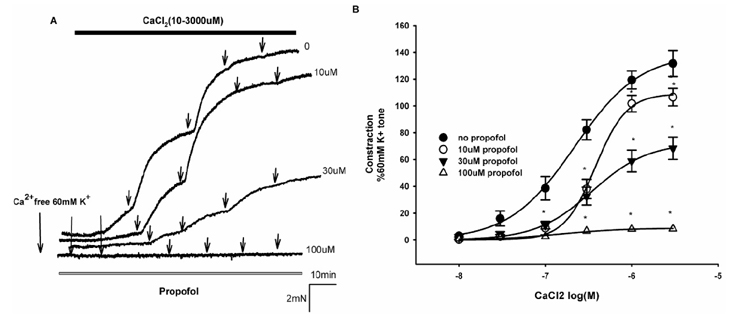
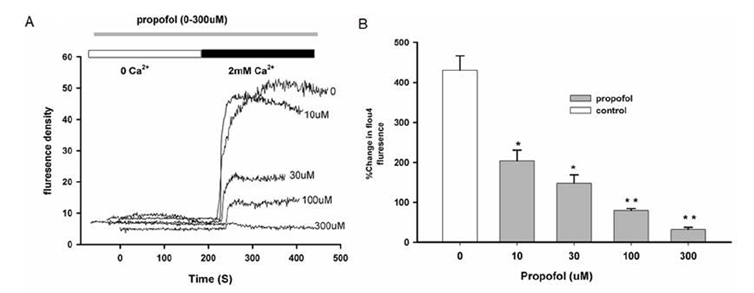
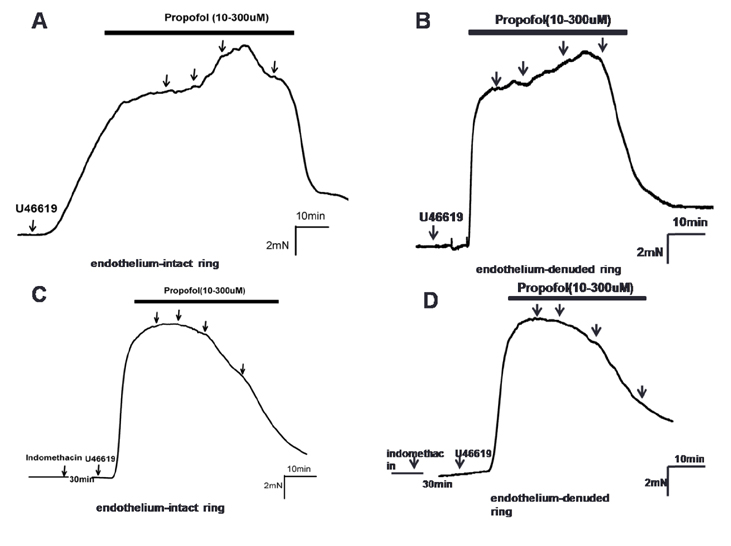
- Propofol is known to cause vasorelaxation of several systemic vascular beds. However, its effect on the pulmonary vasculature remains controversial. In the present study, we investigated the effects of propofol on human pulmonary arteries obtained from patients who had undergone surgery. Arterial rings were mounted in a Multi-Myograph system for measurement of isometric forces. U46619 was used to induce sustained contraction of the intrapulmonary arteries, and propofol was then applied (in increments from 10-300 µM). Arteries denuded of endothelium, preincubated or not with indomethacin, were used to investigate the effects of propofol on isolated arteries. Propofol exhibited a bifunctional effect on isolated human pulmonary arteries contracted by U46619, evoking constriction at low concentrations (10-100 µM) followed by secondary relaxation (at 100-300 µM). The extent of constriction induced by propofol was higher in an endothelium-denuded group than in an endothelium-intact group. Preincubation with indomethacin abolished constriction and potentiated relaxation. The maximal relaxation was greater in the endothelium-intact than the endothelium-denuded group. Propofol also suppressed CaClâ‚‚-induced constriction in the 60 mM Kâº-containing Ca²âº-free solution in a dose-dependent manner. Fluorescent imaging of Ca²âº using fluo-4 showed that a 10 min incubation with propofol (10-300 µM) inhibited the Ca²âº influx into human pulmonary arterial smooth muscle cells induced by a 60 mM Kâº-containing Ca²âº-free solution. In conclusion, propofol-induced arterial constriction appears to involve prostaglandin production by cyclooxygenase in pulmonary artery smooth muscle cells and the relaxation depends in part on endothelial function, principally on the inhibition of calcium influx through L-type voltage-operated calcium channels.
Keyword
MeSH Terms
-
15-Hydroxy-11 alpha,9 alpha-(epoxymethano)prosta-5,13-dienoic Acid
Arteries
Calcium
Calcium Channels
Constriction
Endothelium
Humans*
Indomethacin
Myocytes, Smooth Muscle
Propofol*
Prostaglandin-Endoperoxide Synthases
Pulmonary Artery*
Relaxation
Vasoconstriction*
Vasodilation
15-Hydroxy-11 alpha,9 alpha-(epoxymethano)prosta-5,13-dienoic Acid
Calcium
Calcium Channels
Indomethacin
Propofol
Prostaglandin-Endoperoxide Synthases
Figure
Reference
-
1. Sakai Y, Kawahito S, Takaishi K, Mita N, Kinoshita H, Hatakeyama N, Azma T, Nakaya Y, Kitahata H. Propofol-induced relaxation of rat aorta is altered by agingt. J Med Invest. 2014; 61:278–284.2. Klockgether-Radke AP, Schulze H, Neumann P, Hellige G. Activation of the K+ channel BK(Ca) is involved in the relaxing effect of propofol on coronary arteries. Eur J Anaesthesiol. 2004; 21:226–230.3. Liu Y, Chang H, Niu L, Xue W, Zhang X, Liang Y, Zhang M. Effects of propofol on responses of rat isolated renal arteriole to vasoactive agents. Vascul Pharmacol. 2009; 51:182–189.4. Machała W, Szebla R. Effects of propofol induction on haemodynamics. Anestezjol Intens Ter. 2008; 40:223–226.5. Funayama T, Aida S, Matsukawa T, Okada K, Kumazawa T. Systemic, but not pulmonary, hemodynamics are depressed during combined high thoraco-cervical epidural and general anesthesia in dogs. Can J Anaesth. 2003; 50:454–459.6. Tanaka H, Yamanoue T, Kuroda M, Kawamoto M, Yuge O. Propofol relaxes extrapulmonary artery but not intrapulmonary artery through nitric oxide pathway. Hiroshima J Med Sci. 2001; 50:61–64.7. Zhang G, Cui J, Chen Y, Ma J. The relaxant effect of propofol on isolated rat intrapulmonary arteries. Korean J Physiol Pharmacol. 2014; 18:377–381.8. Kondo U, Kim SO, Nakayama M, Murray PA. Pulmonary vascular effects of propofol at baseline, during elevated vasomotor tone, and in response to sympathetic alpha- and beta-adrenoreceptor activation. Anesthesiology. 2001; 94:815–823.9. Ogawa K, Tanaka S, Murray PA. Propofol potentiates phenylephrine-induced contraction via cyclooxygenase inhibition in pulmonary artery smooth muscle. Anesthesiology. 2001; 94:833–839.10. Ge Y, Cheng J, Xi W, Zheng S, Kang Y, Jiang Y. Effect of ulinastatin on thromboxane B2 and deep vein thrombosis in elderly patients after hip joint replacement. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2010; 35:1278–1281.11. Hei ZQ, Huang HQ, Luo CF, Li SR, Luo GJ. Changes of nitric oxide and endothelin, thromboxane A2 and prostaglandin in cirrhotic patients undergoing liver transplantation. World J Gastroenterol. 2006; 12:4049–4051.12. Tachtsi M, Pitoulias G, Kostoglou C, Papadimitriou D. The proinflammatory mediator's production from ischemic brain during carotid endarterectomy. Int Angiol. 2011; 30:429–433.13. Undas A, Siudak Z, Topór-Mądry R, Leśniak M, Tracz W. Simvastatin administration reduces thromboxane production in subjects taking aspirin: links between aspirin resistance and thrombin generation. Int J Cardiol. 2012; 154:59–64.14. Rich GF, Roos CM, Anderson SM, Daugherty MO, Uncles DR. Direct effects of intravenous anesthetics on pulmonary vascular resistance in the isolated rat lung. Anesth Analg. 1994; 78:961–966.15. Nakahata N, Takano H, Ohizumi Y. Thromboxane A2 receptor-mediated tonic contraction is attributed to an activation of phosphatidylcholine-specific phospholipase C in rabbit aortic smooth muscles. Life Sci. 2000; 66:PL 71-76.16. Félétou M, Huang Y, Vanhoutte PM. Endothelium-mediated control of vascular tone: COX-1 and COX-2 products. Br J Pharmacol. 2011; 164:894–912.17. Bradbury DA, Newton R, Zhu YM, Stocks J, Corbett L, Holland ED, Pang LH, Knox AJ. Effect of bradykinin, TGF-beta1, IL-1beta, and hypoxia on COX-2 expression in pulmonary artery smooth muscle cells. Am J Physiol Lung Cell Mol Physiol. 2002; 283:L717–L725.18. Bradbury DA, Newton R, Zhu YM, El-Haroun H, Corbett L, Knox AJ. Cyclooxygenase-2 induction by bradykinin in human pulmonary artery smooth muscle cells is mediated by the cyclic AMP response element through a novel autocrine loop involving endogenous prostaglandin E2, E-prostanoid 2 (EP2), and EP4 receptors. J Biol Chem. 2003; 278:49954–49964.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Requirement of Pretone by Thromboxane A2 for Hypoxic Pulmonary Vasoconstriction in Precision-cut Lung Slices of Rat
- The Relaxant Effect of Propofol on Isolated Rat Intrapulmonary Arteries
- Propofol Attenuates Prostacyclin-induced Pulmonary Vasorelaxation in U46619 Preconstricted Dog
- Disappearance of Hypoxic Pulmonary Vasoconstriction and O2-Sensitive Nonselective Cationic Current in Arterial Myocytes of Rats Under Ambient Hypoxia
- Effects of Propofol on Arginine Vasopressin-induced Contraction in Isolated Human Gastroepiploic Artery