Chonnam Med J.
2017 Sep;53(3):173-177. 10.4068/cmj.2017.53.3.173.
Therapeutic Inhibitors against Mutated BRAF and MEK for the Treatment of Metastatic Melanoma
- Affiliations
-
- 1Department of Dermatology, University of Colorado Denver Medical School, Aurora, Colorado, USA. peter.song@ucdenver.edu
- 2Department of Premedical Sciences, Chosun University School of Medicine, Gwangju, Korea.
- KMID: 2390430
- DOI: http://doi.org/10.4068/cmj.2017.53.3.173
Abstract
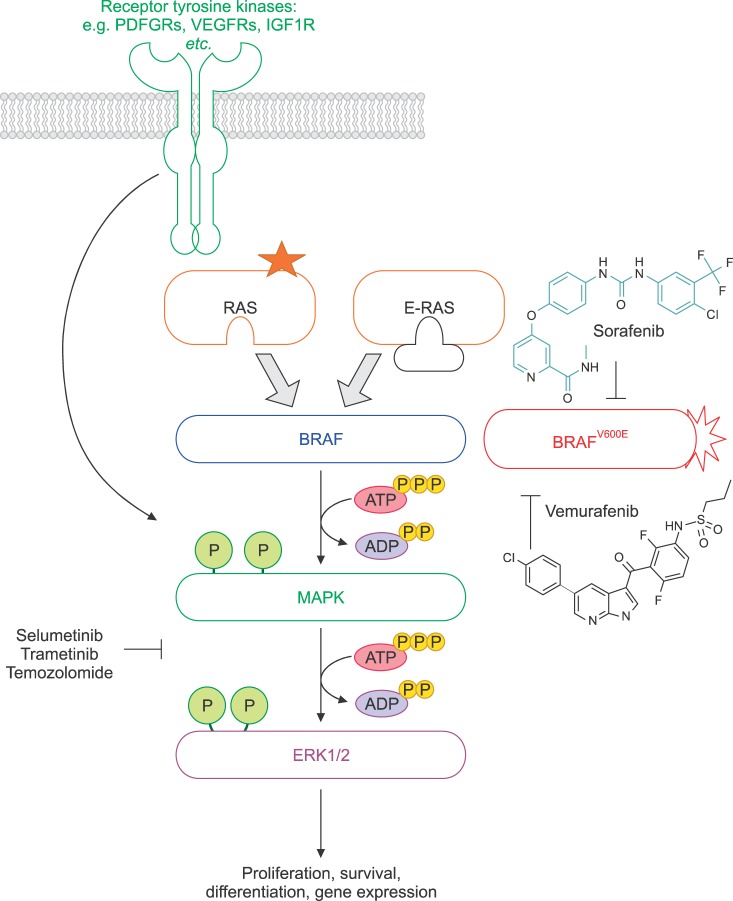
- Melanoma is one of the most aggressive cancers in the world and is responsible for the majority of skin cancer deaths. Recent advances in the field of immunotherapy using active, adoptive, and antigen-specific therapeutic approaches, have generated the expectation that these technologies have the potential to improve the treatment of advanced malignancies, including melanoma. Treatment options for metastatic melanoma patients have been dramatically improved by the FDA approval of new therapeutic agents including vemurafenib, dabrafenib, and sorafenib. These kinase inhibitors have the potential to work in tandem with MEK, PI3K/AKT, and mTOR to inhibit the activity of melanoma inducing BRAF mutations. This review summarizes the effects of the new therapeutic agents against melanoma and the underlying biology of these BRAF inhibitors.
MeSH Terms
Figure
Cited by 1 articles
-
Melanoma Cell Death Mechanisms
Lindsey Broussard, Amanda Howland, Sunhyo Ryu, Kyungsup Song, David Norris, Cheryl A. Armstrong, Peter I. Song
Chonnam Med J. 2018;54(3):135-142. doi: 10.4068/cmj.2018.54.3.135.
Reference
-
1. Ahn A, Chatterjee A, Eccles MR. The slow cycling phenotype: a growing problem for treatment resistance in melanoma. Mol Cancer Ther. 2017; 16:1002–1009. PMID: 28576947.2. Davies H, Bignell GR, Cox C, Stephens P, Edkins S, Clegg S, et al. Mutations of the BRAF gene in human cancer. Nature. 2002; 417:949–954. PMID: 12068308.3. Platz A, Egyhazi S, Ringborg U, Hansson J. Human cutaneous melanoma; a review of NRAS and BRAF mutation frequencies in relation to histogenetic subclass and body site. Mol Oncol. 2008; 1:395–405. PMID: 19383313.4. Wellbrock C, Arozarena I. The complexity of the ERK/MAP-Kinase pathway and the treatment of melanoma skin cancer. Front Cell Dev Biol. 2016; 4:33. PMID: 27200346.5. Fiskus W, Mitsiades N. B-Raf Inhibition in the Clinic: Present and Future. Annu Rev Med. 2016; 67:29–43. PMID: 26768236.6. Avruch J, Khokhlatchev A, Kyriakis JM, Luo Z, Tzivion G, Vavvas D, et al. Ras activation of the Raf kinase: tyrosine kinase recruitment of the MAP kinase cascade. Recent Prog Horm Res. 2001; 56:127–155. PMID: 11237210.7. An S, Yang Y, Ward R, Liu Y, Guo XX, Xu TR. A-Raf: A new star of the family of raf kinases. Crit Rev Biochem Mol Biol. 2015; 50:520–531. PMID: 26508523.8. Solit DB, Garraway LA, Pratilas CA, Sawai A, Getz G, Basso A, et al. BRAF mutation predicts sensitivity to MEK inhibition. Nature. 2006; 439:358–362. PMID: 16273091.9. Bollag G, Tsai J, Zhang J, Zhang C, Ibrahim P, Nolop K, et al. Vemurafenib: the first drug approved for BRAF-mutant cancer. Nat Rev Drug Discov. 2012; 11:873–886. PMID: 23060265.10. Yang H, Higgins B, Kolinsky K, Packman K, Go Z, Iyer R, et al. RG7204 (PLX4032), a selective BRAFV600E inhibitor, displays potent antitumor activity in preclinical melanoma models. Cancer Res. 2010; 70:5518–5527. PMID: 20551065.11. Halaban R, Zhang W, Bacchiocchi A, Cheng E, Parisi F, Ariyan S, et al. PLX4032, a selective BRAF(V600E) kinase inhibitor, activates the ERK pathway and enhances cell migration and proliferation of BRAF melanoma cells. Pigment Cell Melanoma Res. 2010; 23:190–200. PMID: 20149136.12. Søndergaard JN, Nazarian R, Wang Q, Guo D, Hsueh T, Mok S, et al. Differential sensitivity of melanoma cell lines with BRAFV600E mutation to the specific Raf inhibitor PLX4032. J Transl Med. 2010; 8:39. PMID: 20406486.13. Joseph EW, Pratilas CA, Poulikakos PI, Tadi M, Wang W, Taylor BS, et al. The RAF inhibitor PLX4032 inhibits ERK signaling and tumor cell proliferation in a V600E BRAF-selective manner. Proc Natl Acad Sci U S A. 2010; 107:14903–14908. PMID: 20668238.14. Pratilas CA, Solit DB. Targeting the mitogen-activated protein kinase pathway: physiological feedback and drug response. Clin Cancer Res. 2010; 16:3329–3334. PMID: 20472680.15. Sala E, Mologni L, Truffa S, Gaetano C, Bollag GE, Gambacorti-Passerini C. BRAF silencing by short hairpin RNA or chemical blockade by PLX4032 leads to different responses in melanoma and thyroid carcinoma cells. Mol Cancer Res. 2008; 6:751–759. PMID: 18458053.16. Salerno P, De Falco V, Tamburrino A, Nappi TC, Vecchio G, Schweppe RE, et al. Cytostatic activity of adenosine triphosphate-competitive kinase inhibitors in BRAF mutant thyroid carcinoma cells. J Clin Endocrinol Metab. 2010; 95:450–455. PMID: 19880792.17. Young K, Minchom A, Larkin J. BRIM-1, -2 and -3 trials: improved survival with vemurafenib in metastatic melanoma patients with a BRAF(V600E) mutation. Future Oncol. 2012; 8:499–507. PMID: 22646765.18. Paraiso KH, Fedorenko IV, Cantini LP, Munko AC, Hall M, Sondak VK, et al. Recovery of phospho-ERK activity allows melanoma cells to escape from BRAF inhibitor therapy. Br J Cancer. 2010; 102:1724–1730. PMID: 20531415.19. Basile KJ, Abel EV, Aplin AE. Adaptive upregulation of FOXD3 and resistance to PLX4032/4720-induced cell death in mutant B-RAF melanoma cells. Oncogene. 2012; 31:2471–2479. PMID: 21996740.20. Nazarian R, Shi H, Wang Q, Kong X, Koya RC, Lee H, et al. Melanomas acquire resistance to B-RAF(V600E) inhibition by RTK or N-RAS upregulation. Nature. 2010; 468:973–977. PMID: 21107323.21. Kudchadkar R, Paraiso KH, Smalley KS. Targeting mutant BRAF in melanoma: current status and future development of combination therapy strategies. Cancer J. 2012; 18:124–131. PMID: 22453012.22. Ravnan MC, Matalka MS. Vemurafenib in patients with BRAF V600E mutation-positive advanced melanoma. Clin Ther. 2012; 34:1474–1486. PMID: 22742884.23. Trino E, Mantovani C, Badellino S, Ricardi U, Filippi AR. Radiosurgery/stereotactic radiotherapy in combination with immunotherapy and targeted agents for melanoma brain metastases. Expert Rev Anticancer Ther. 2017; 17:347–356. PMID: 28277101.24. Specenier P. Ipilimumab in melanoma. Expert Rev Anticancer Ther. 2016; 16:811–826. PMID: 27403706.25. Boni A, Cogdill AP, Dang P, Udayakumar D, Njauw CN, Sloss CM, et al. Selective BRAFV600E inhibition enhances T-cell recognition of melanoma without affecting lymphocyte function. Cancer Res. 2010; 70:5213–5219. PMID: 20551059.26. Falchook GS, Long GV, Kurzrock R, Kim KB, Arkenau TH, Brown MP, et al. Dabrafenib in patients with melanoma, untreated brain metastases, and other solid tumours: a phase 1 dose-escalation trial. Lancet. 2012; 379:1893–1901. PMID: 22608338.27. Wang H, Quan H, Lou L. AKT is critically involved in the antagonism of BRAF inhibitor sorafenib against dabrafenib in colorectal cancer cells harboring both wild-type and mutant (V600E) BRAF genes. Biochem Biophys Res Commun. 2017; 489:14–20. PMID: 28536078.28. Williams CB, McMahon C, Ali SM, Abramovitz M, Williams KA, Klein J, et al. A metastatic colon adenocarcinoma harboring BRAF V600E has a durable major response to dabrafenib/trametinib and chemotherapy. Onco Targets Ther. 2015; 8:3561–3564. PMID: 26664139.29. King AJ, Arnone MR, Bleam MR, Moss KG, Yang J, Fedorowicz KE, et al. Dabrafenib; preclinical characterization, increased efficacy when combined with trametinib, while BRAF/MEK tool combination reduced skin lesions. PLoS One. 2013; 8:e67583. PMID: 23844038.30. Velho TR. Metastatic melanoma - a review of current and future drugs. Drugs Context. 2012; 2012:212242. PMID: 24432031.31. Bollag G, Hirth P, Tsai J, Zhang J, Ibrahim PN, Cho H, et al. Clinical efficacy of a RAF inhibitor needs broad target blockade in BRAF-mutant melanoma. Nature. 2010; 467:596–599. PMID: 20823850.32. Flaherty KT, Puzanov I, Kim KB, Ribas A, McArthur GA, Sosman JA, et al. Inhibition of mutated, activated BRAF in metastatic melanoma. N Engl J Med. 2010; 363:809–819. PMID: 20818844.33. Gang G, Hongkai Y, Xu Z. Sorafenib combined with radiofrequency ablation in the treatment of a patient with renal cell carcinoma plus primary hepatocellular carcinoma. J Cancer Res Ther. 2015; 11:1026.34. McDermott DF, Sosman JA, Gonzalez R, Hodi FS, Linette GP, Richards J, et al. Double-blind randomized phase II study of the combination of sorafenib and dacarbazine in patients with advanced melanoma: a report from the 11715 Study Group. J Clin Oncol. 2008; 26:2178–2185. PMID: 18445842.35. Romero AI, Chaput N, Poirier-Colame V, Rusakiewicz S, Jacquelot N, Chaba K, et al. Regulation of CD4(+)NKG2D(+) Th1 cells in patients with metastatic melanoma treated with sorafenib: role of IL-15Rα and NKG2D triggering. Cancer Res. 2014; 74:68–80. PMID: 24197135.36. Lee HJ, Wall BA, Wangari-Talbot J, Shin SS, Rosenberg S, Chan JL, et al. Glutamatergic pathway targeting in melanoma: single-agent and combinatorial therapies. Clin Cancer Res. 2011; 17:7080–7092. PMID: 21844014.37. Margolin KA, Moon J, Flaherty LE, Lao CD, Akerley WL 3rd, Othus M, et al. Randomized phase II trial of sorafenib with temsirolimus or tipifarnib in untreated metastatic melanoma (S0438). Clin Cancer Res. 2012; 18:1129–1137. PMID: 22228638.38. Kumar SK, Jett J, Marks R, Richardson R, Quevedo F, Moynihan T, et al. Phase 1 study of sorafenib in combination with bortezomib in patients with advanced malignancies. Invest New Drugs. 2013; 31:1201–1206. PMID: 23887852.39. Kirkwood JM, Bastholt L, Robert C, Sosman J, Larkin J, Hersey P, et al. Phase II, open-label, randomized trial of the MEK1/2 inhibitor selumetinib as monotherapy versus temozolomide in patients with advanced melanoma. Clin Cancer Res. 2012; 18:555–567. PMID: 22048237.40. Bowyer SE, Rao AD, Lyle M, Sandhu S, Long GV, McArthur GA, et al. Activity of trametinib in K601E and L597Q BRAF mutation-positive metastatic melanoma. Melanoma Res. 2014; 24:504–508. PMID: 24933606.41. Catalanotti F, Solit DB, Pulitzer MP, Berger MF, Scott SN, Iyriboz T, et al. Phase II trial of MEK inhibitor selumetinib (AZD6244, ARRY-142886) in patients with BRAFV600E/K-mutated melanoma. Clin Cancer Res. 2013; 19:2257–2264. PMID: 23444215.42. Johnson DB, Flaherty KT, Weber JS, Infante JR, Kim KB, Kefford RF, et al. Combined BRAF (Dabrafenib) and MEK inhibition (Trametinib) in patients with BRAFV600-mutant melanoma experiencing progression with single-agent BRAF inhibitor. J Clin Oncol. 2014; 32:3697–3704. PMID: 25287827.43. Kunz M. Oncogenes in melanoma: an update. Eur J Cell Biol. 2014; 93:1–10. PMID: 24468268.44. Nikolaou VA, Stratigos AJ, Flaherty KT, Tsao H. Melanoma: new insights and new therapies. J Invest Dermatol. 2012; 132:854–863. PMID: 22217739.45. Kulkarni A, Al-Hraishawi H, Simhadri S, Hirshfield KM, Chen S, Pine S, et al. BRAF fusion as a novel mechanism of acquired resistance to vemurafenib in BRAFV600E mutant melanoma. Clin Cancer Res. 2017; DOI: 10.1158/1078-0432.CCR-16-0758. [Epub ahead of print].46. Theodosakis N, Micevic G, Langdon CG, Ventura A, Means R, Stern DF, et al. p90RSK blockade inhibits dual BRAF and MEK inhibitor-resistant melanoma by targeting protein synthesis. J Invest Dermatol. 2017; DOI: 10.1016/j.jid.2016.12.033. [Epub ahead of print].47. Wu SH, Zhang LN, Speakman JR, Wang DH. Limits to sustained energy intake. XI. A test of the heat dissipation limitation hypothesis in lactating Brandt's voles (Lasiopodomys brandtii). J Exp Biol. 2009; 212:3455–3465. PMID: 19837887.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Differential Sensitivity of Wild-Type and BRAF-Mutated Cells to Combined BRAF and Autophagy Inhibition
- Targeting BRAF pathway in low-grade serous ovarian cancer
- Treatment Effect of Combining Lenvatinib and Vemurafenib for BRAF Mutated Anaplastic Thyroid Cancer
- BRAF/ MEK Inhibitors in Downstaging BRAFV600E Mutated Papillary Thyroid Cancer to Allow Resection: Case Report and Literature Review
- Combination of Dabrafenib and Trametinib in Patients with Metastatic BRAFV600E-Mutated Thyroid Cancer