Nutr Res Pract.
2017 Oct;11(5):430-434. 10.4162/nrp.2017.11.5.430.
Luteolin and fisetin suppress oxidative stress by modulating sirtuins and forkhead box O3a expression under in vitro diabetic conditions
- Affiliations
-
- 1Department of Food and Nutrition, Chonnam National University, Yongbong-ro, Buk-gu, Gwangju 61186, Korea. sosung75@jnu.ac.kr
- 2National Research Center for Dementia, Chosun University, Gwangju 61452, Korea.
- KMID: 2390136
- DOI: http://doi.org/10.4162/nrp.2017.11.5.430
Abstract
- BACKGROUND
/OBJECTIVE: Chronic hyperglycemia induces oxidative stress via accumulation of reactive oxygen species (ROS) and contributes to diabetic complications. Hyperglycemia induces mitochondrial superoxide anion production through the increased activity of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase. This study aimed to determine whether fisetin and luteolin treatments suppress the oxidative stress by modulating the expression of sirtuins (SIRTs) and forkhead box O3a (FOXO3a) under hyperglycemic conditions in human monocytes.
MATERIALS/METHODS
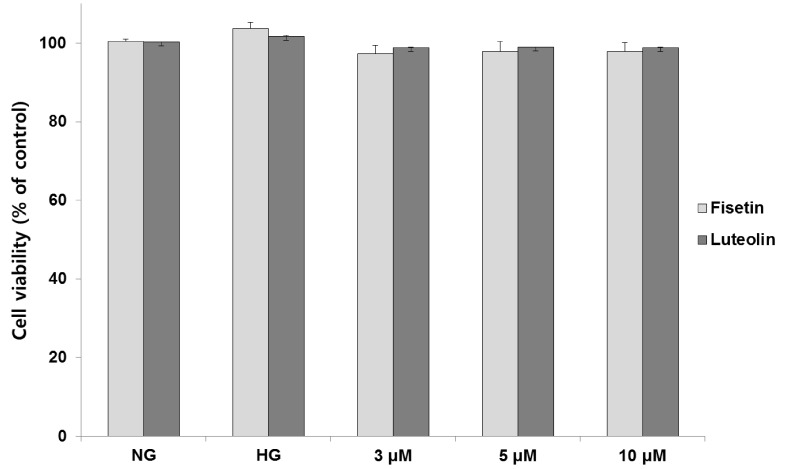
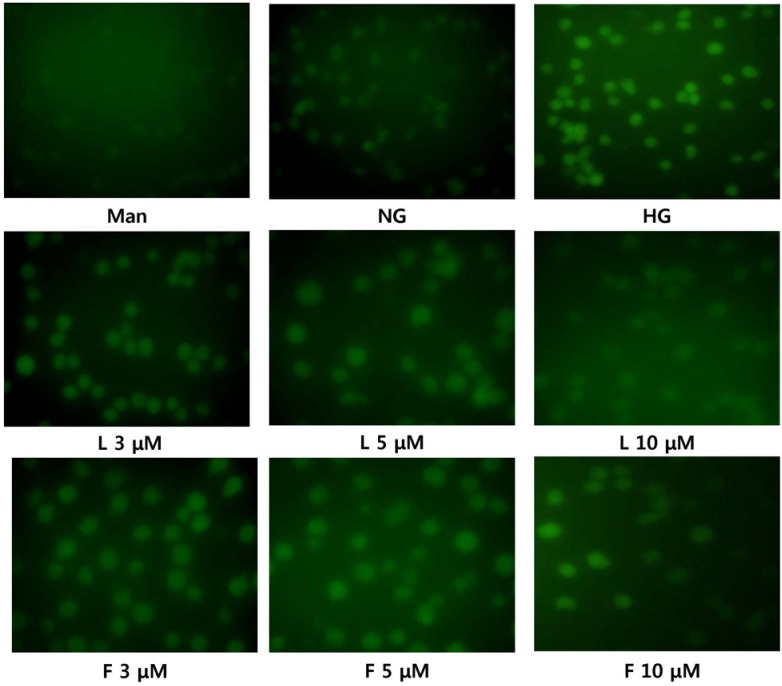
Human monocytic cells (THP-1) were cultured under osmotic control (14.5 mmol/L mannitol), normoglycemic (NG, 5.5 mmol/L glucose), or hyperglycemic (HG, 20 mmol/L glucose) conditions, in the absence or presence of fisetin and luteolin for 48 h. To determine the effect of fisetin and luteolin treatments on high glucose-induced oxidative stress, western blotting and intracellular staining were performed.
RESULTS
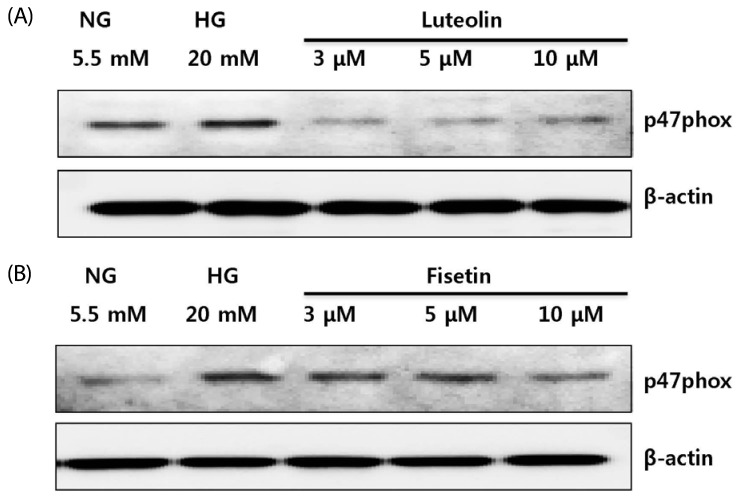
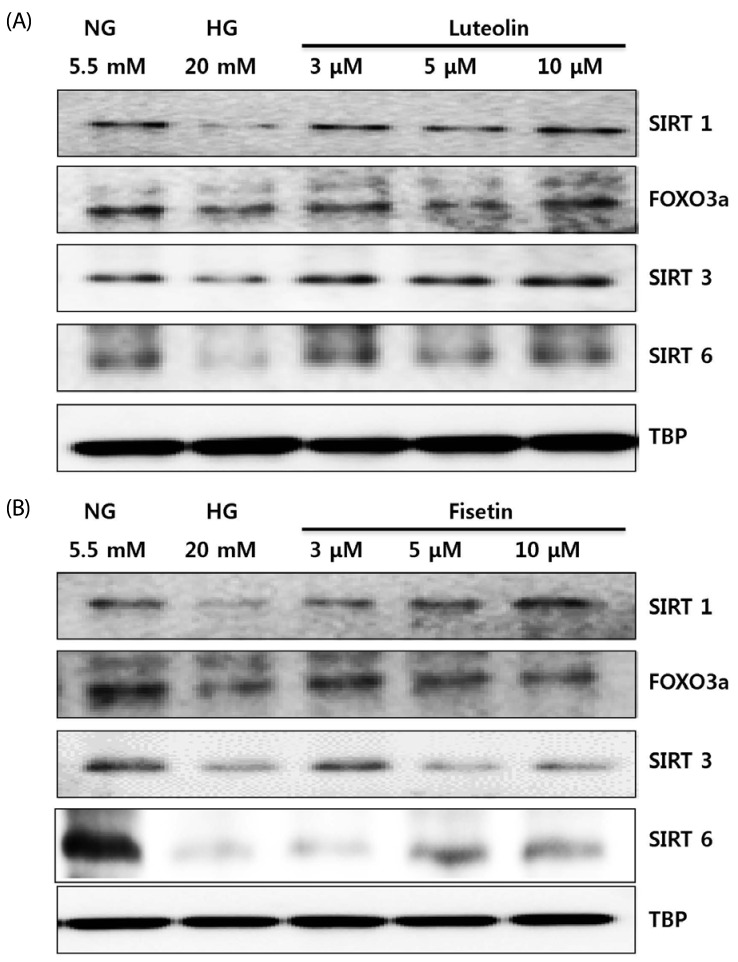
Hyperglycemic conditions increased the ROS production, as compared to normoglycemic condition. However, fisetin and luteolin treatments inhibited ROS production under hyperglycemia. To obtain further insight into ROS production in hyperglycemic conditions, evaluation of p47phox expression revealed that fisetin and luteolin treatments inhibited p47phox expression under hyperglycemic conditions. Conversely, the expression levels of SIRT1, SIRT3, SIRT6, and FOXO3a were decreased under high glucose conditions compared to normal glucose conditions, but exposure to fisetin and luteolin induced the expression of SIRT1, SIRT3, SIRT6, and FOXO3a. The above findings suggest that fisetin and luteolin inhibited high glucose-induced ROS production in monocytes through the activation of SIRTs and FOXO3a.
CONCLUSIONS
The results of our study supports current researches that state fisetin and luteolin as potential agents for the development of novel strategies for diabetes.
Keyword
MeSH Terms
Figure
Reference
-
1. Forbes JM, Cooper ME. Mechanisms of diabetic complications. Physiol Rev. 2013; 93:137–188. PMID: 23303908.
Article2. Gross JL, de Azevedo MJ, Silveiro SP, Canani LH, Caramori ML, Zelmanovitz T. Diabetic nephropathy: diagnosis, prevention, and treatment. Diabetes Care. 2005; 28:164–176. PMID: 15616252.
Article3. Brownlee M. Biochemistry and molecular cell biology of diabetic complications. Nature. 2001; 414:813–820. PMID: 11742414.
Article4. Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes. 2005; 54:1615–1625. PMID: 15919781.5. Giacco F, Brownlee M. Oxidative stress and diabetic complications. Circ Res. 2010; 107:1058–1070. PMID: 21030723.
Article6. El-Benna J, Dang PM, Gougerot-Pocidalo MA, Elbim C. Phagocyte NADPH oxidase: a multicomponent enzyme essential for host defenses. Arch Immunol Ther Exp (Warsz). 2005; 53:199–206. PMID: 15995580.7. Venugopal SK, Devaraj S, Yang T, Jialal I. Alpha-tocopherol decreases superoxide anion release in human monocytes under hyperglycemic conditions via inhibition of protein kinase C-alpha. Diabetes. 2002; 51:3049–3054. PMID: 12351446.8. Yun JM, Chien A, Jialal I, Devaraj S. Resveratrol up-regulates SIRT1 and inhibits cellular oxidative stress in the diabetic milieu: mechanistic insights. J Nutr Biochem. 2012; 23:699–705. PMID: 21813271.
Article9. Houtkooper RH, Pirinen E, Auwerx J. Sirtuins as regulators of metabolism and healthspan. Nat Rev Mol Cell Biol. 2012; 13:225–238. PMID: 22395773.
Article10. Kitada M, Koya D. SIRT1 in type 2 diabetes: mechanisms and therapeutic potential. Diabetes Metab J. 2013; 37:315–325. PMID: 24199159.
Article11. Eijkelenboom A, Burgering BM. FOXOs: signalling integrators for homeostasis maintenance. Nat Rev Mol Cell Biol. 2013; 14:83–97. PMID: 23325358.
Article12. Gross DN, van den Heuvel AP, Birnbaum MJ. The role of FoxO in the regulation of metabolism. Oncogene. 2008; 27:2320–2336. PMID: 18391974.
Article13. Kobayashi Y, Furukawa-Hibi Y, Chen C, Horio Y, Isobe K, Ikeda K, Motoyama N. SIRT1 is critical regulator of FOXO-mediated transcription in response to oxidative stress. Int J Mol Med. 2005; 16:237–243. PMID: 16012755.
Article14. Brunet A, Sweeney LB, Sturgill JF, Chua KF, Greer PL, Lin Y, Tran H, Ross SE, Mostoslavsky R, Cohen HY, Hu LS, Cheng HL, Jedrychowski MP, Gygi SP, Sinclair DA, Alt FW, Greenberg ME. Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science. 2004; 303:2011–2015. PMID: 14976264.
Article15. Rathee P, Chaudhary H, Rathee S, Rathee D, Kumar V, Kohli K. Mechanism of action of flavonoids as anti-inflammatory agents: a review. Inflamm Allergy Drug Targets. 2009; 8:229–235. PMID: 19601883.
Article16. Peterson JJ, Dwyer JT, Jacques PF, McCullough ML. Associations between flavonoids and cardiovascular disease incidence or mortality in European and US populations. Nutr Rev. 2012; 70:491–508. PMID: 22946850.
Article17. Nicolle E, Souard F, Faure P, Boumendjel A. Flavonoids as promising lead compounds in type 2 diabetes mellitus: molecules of interest and structure-activity relationship. Curr Med Chem. 2011; 18:2661–2672. PMID: 21568900.
Article18. Li J, Cheng Y, Qu W, Sun Y, Wang Z, Wang H, Tian B. Fisetin, a dietary flavonoid, induces cell cycle arrest and apoptosis through activation of p53 and inhibition of NF-kappa B pathways in bladder cancer cells. Basic Clin Pharmacol Toxicol. 2011; 108:84–93. PMID: 21054790.
Article19. Currais A, Prior M, Dargusch R, Armando A, Ehren J, Schubert D, Quehenberger O, Maher P. Modulation of p25 and inflammatory pathways by fisetin maintains cognitive function in Alzheimer's disease transgenic mice. Aging Cell. 2014; 13:379–390. PMID: 24341874.20. Kwak S, Ku SK, Bae JS. Fisetin inhibits high-glucose-induced vascular inflammation in vitro and in vivo. Inflamm Res. 2014; 63:779–787. PMID: 24923846.
Article21. Xia F, Wang C, Jin Y, Liu Q, Meng Q, Liu K, Sun H. Luteolin protects HUVECs from THF-α-induced oxidative stress and inflammation via its effects on the Nox4/ROS-NF-κB and MAPK pathways. J Atheroscler Thromb. 2014; 21:768–783. PMID: 24621786.22. Seelinger G, Merfort I, Schempp CM. Anti-oxidant, anti-inflammatory and anti-allergic activities of luteolin. Planta Med. 2008; 74:1667–1677. PMID: 18937165.
Article23. Zang Y, Igarashi K, Li Y. Anti-diabetic effects of luteolin and luteolin-7-O-glucoside on KK-A(y) mice. Biosci Biotechnol Biochem. 2016; 80:1580–1586. PMID: 27170065.24. Kim HJ, Kim SH, Yun JM. Fisetin inhibits hyperglycemia-induced proinflammatory cytokine production by epigenetic mechanisms. Evid Based Complement Alternat Med. 2012; 2012:639469. PMID: 23320034.
Article25. Kim HJ, Lee W, Yun JM. Luteolin inhibits hyperglycemia-induced proinflammatory cytokine production and its epigenetic mechanism in human monocytes. Phytother Res. 2014; 28:1383–1391. PMID: 24623679.
Article26. El-Benna J, Dang PM, Gougerot-Pocidalo MA, Marie JC, Braut-Boucher F. p47phox, the phagocyte NADPH oxidase/NOX2 organizer: structure, phosphorylation and implication in diseases. Exp Mol Med. 2009; 41:217–225. PMID: 19372727.
Article27. Liang F, Kume S, Koya D. SIRT1 and insulin resistance. Nat Rev Endocrinol. 2009; 5:367–373. PMID: 19455179.
Article28. Someya S, Yu W, Hallows WC, Xu J, Vann JM, Leeuwenburgh C, Tanokura M, Denu JM, Prolla TA. Sirt3 mediates reduction of oxidative damage and prevention of age-related hearing loss under caloric restriction. Cell. 2010; 143:802–812. PMID: 21094524.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Luteolin inhibits H2O2 -induced cellular senescence via modulation of SIRT1 and p53
- Poly(ADP-ribose) polymerase 1 contributes to oxidative stress through downregulation of sirtuin 3 during cisplatin nephrotoxicity
- The flavonoid fisetin ameliorates renal fibrosis by inhibiting SMAD3 phosphorylation, oxidative damage, and inflammation in ureteral obstructed kidney in mice
- Mitochondrial Mechanisms in Diabetic Cardiomyopathy
- Effects of S-allylcysteine on Oxidative Stress in Streptozotocin-Induced Diabetic Rats