Infect Chemother.
2017 Jun;49(2):117-122. 10.3947/ic.2017.49.2.117.
Humoral and Cellular Immunogenicity Induced by Avian Influenza A (H7N9) DNA Vaccine in Mice
- Affiliations
-
- 1BK21 Plus Graduate Program Biomedical Sciences, Korea University College of Medicine, Seoul, Korea. wjkim@korea.ac.kr
- 2Division of Infectious Diseases, Department of Internal Medicine, Korea University College of Medicine, Seoul, Korea.
- 3Transgovernmental Enterprise for Pandemic Influenza in Korea, Seoul, Korea.
- 4Department of Biomedical Sciences, Korea University College of Medicine, Seoul, Korea.
- 5Department of Microbiology, Korea University College of Medicine, Seoul, Korea.
- 6GeneOne Life Science, Inc., Seoul, Korea.
- KMID: 2383932
- DOI: http://doi.org/10.3947/ic.2017.49.2.117
Abstract
- BACKGROUND
In March 2013, human infection with avian influenza A (H7N9) virus emerged in China, causing serious public health concerns and raising the possibility of avian-source pandemic influenza. Thus, the development of an effective vaccine for preventing and rapidly controlling avian influenza A (H7N9) virus is needed. In this study, we evaluated the immunogenicity of a synthetic DNA vaccine against H7 HA antigens in mice.
MATERIALS AND METHODS
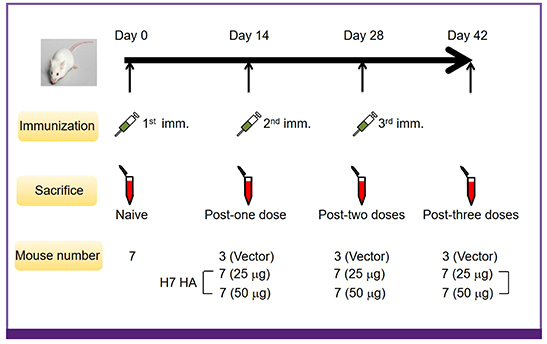
The synthetic consensus H7 HA DNA vaccine (25 or 50 µg) was administered to BALB/c mice at 0, 14, and 28 days by intramuscular injection followed by electroporation. Humoral and cellular immune responses were analyzed in a hemagglutination inhibition test and interferon-gamma enzyme-linked immunospot (ELISpot) assay, respectively.
RESULTS
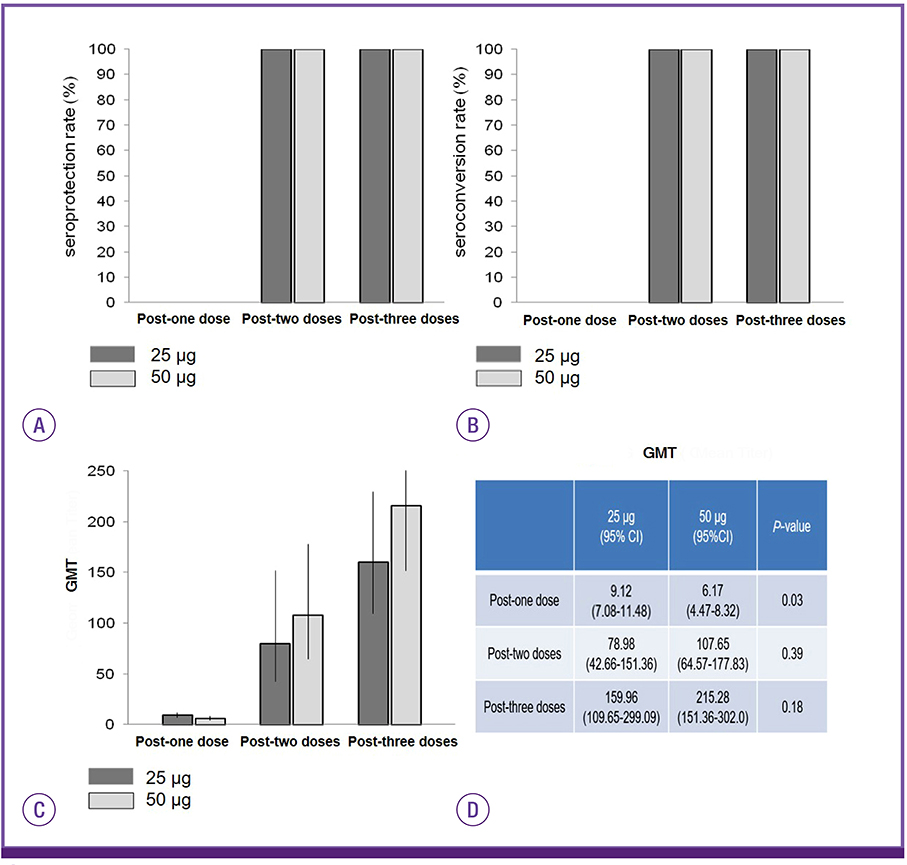
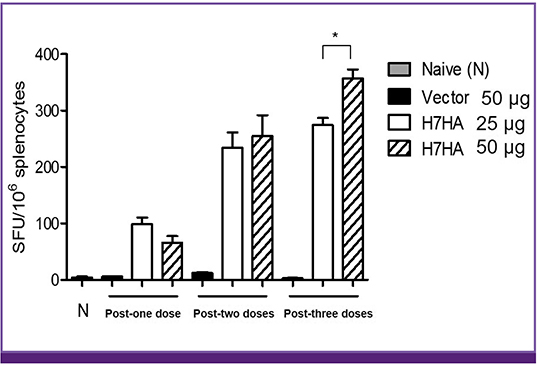
H7 HA-vaccinated mice showed 100% seroprotection and seroconversion rate against H7N9 reassortant influenza virus after both second and third immunizations. The geometric mean titer by the hemagglutination inhibition test increased with an increasing number of immunizations. However, there was no significant difference in geometric titer between the two groups injected with 25 and 50 µg of H7 HA DNA vaccine after two (79.98 vs. 107.65, P = 0.39) and three (159.96 vs. 215.28, P = 0.18) doses. In addition, the ELISpot assay revealed that administration of H7 HA DNA vaccine induced potent interferon-gamma production from mouse splenocytes.
CONCLUSIONS
This study demonstrated the humoral and cellular immunogenicity of synthetic consensus H7 HA DNA vaccine in mice. This work demonstrates the potential of the H7 HA DNA vaccine as an efficient tool for the rapid control of emerging influenza A (H7N9) virus.
MeSH Terms
Figure
Reference
-
1. Hilleman MR. Realities and enigmas of human viral influenza: pathogenesis, epidemiology and control. Vaccine. 2002; 20:3068–3087.
Article2. Simonsen L, Clarke MJ, Schonberger LB, Arden NH, Cox NJ, Fukuda K. Pandemic versus epidemic influenza mortality: a pattern of changing age distribution. J Infect Dis. 1998; 178:53–60.
Article3. Gao R, Cao B, Hu Y, Feng Z, Wang D, Hu W, Chen J, Jie Z, Qiu H, Xu K, Xu X, Lu H, Zhu W, Gao Z, Xiang N, Shen Y, He Z, Gu Y, Zhang Z, Yang Y, Zhao X, Zhou L, Li X, Zou S, Zhang Y, Li X, Yang L, Guo J, Dong J, Li Q, Dong L, Zhu Y, Bai T, Wang S, Hao P, Yang W, Zhang Y, Han J, Yu H, Li D, Gao GF, Wu G, Wang Y, Yuan Z, Shu Y. Human infection with a novel avian-origin influenza A (H7N9) virus. N Engl J Med. 2013; 368:1888–1897.
Article4. Husain M. Avian influenza A (H7N9) virus infection in humans: epidemiology, evolution, and pathogenesis. Infect Genet Evol. 2014; 28:304–312.
Article5. Kageyama T, Fujisaki S, Takashita E, Xu H, Yamada S, Uchida Y, Neumann G, Saito T, Kawaoka Y, Tashiro M. Genetic analysis of novel avian A(H7N9) influenza viruses isolated from patients in China, February to April 2013. Euro Surveill. 2013; 18:20453.
Article6. Liu D, Shi W, Shi Y, Wang D, Xiao H, Li W, Bi Y, Wu Y, Li X, Yan J, Liu W, Zhao G, Yang W, Wang Y, Ma J, Shu Y, Lei F, Gao GF. Origin and diversity of novel avian influenza A H7N9 viruses causing human infection: phylogenetic, structural, and coalescent analyses. Lancet. 2013; 381:1926–1932.
Article7. Wang C, Wang J, Su W, Gao S, Luo J, Zhang M, Xie L, Liu S, Liu X, Chen Y, Jia Y, Zhang H, Ding H, He H. Relationship between domestic and wild birds in live poultry market and a novel human H7N9 virus in China. J Infect Dis. 2014; 209:34–37.
Article8. Fries LF, Smith GE, Glenn GM. A recombinant viruslike particle influenza A (H7N9) vaccine. N Engl J Med. 2013; 369:2564–2566.
Article9. Bart SA, Hohenboken M, Della Cioppa G, Narasimhan V, Dormitzer PR, Kanesa-Thasan N. A cell culture-derived MF59-adjuvanted pandemic A/H7N9 vaccine is immunogenic in adults. Sci Transl Med. 2014; 6:234ra55.
Article10. Yan J, Villarreal DO, Racine T, Chu JS, Walters JN, Morrow MP, Khan AS, Sardesai NY, Kim JJ, Kobinger GP, Weiner DB. Protective immunity to H7N9 influenza viruses elicited by synthetic DNA vaccine. Vaccine. 2014; 32:2833–2842.
Article11. Li L, Saade F, Petrovsky N. The future of human DNA vaccines. J Biotechnol. 2012; 162:171–182.
Article12. Liu MA. DNA vaccines: an historical perspective and view to the future. Immunol Rev. 2011; 239:62–84.
Article13. Iurescia S, Fioretti D, Rinaldi M. Strategies for improving DNA vaccine performance. Methods Mol Biol. 2014; 1143:21–31.
Article14. Flingai S, Czerwonko M, Goodman J, Kudchodkar SB, Muthumani K, Weiner DB. Synthetic DNA vaccines: improved vaccine potency by electroporation and co-delivered genetic adjuvants. Front Immunol. 2013; 4:354.
Article15. Widera G, Austin M, Rabussay D, Goldbeck C, Barnett SW, Chen M, Leung L, Otten GR, Thudium K, Selby MJ, Ulmer JB. Increased DNA vaccine delivery and immunogenicity by electroporation in vivo. J Immunol. 2000; 164:4635–4640.
Article16. Shen X, Söderholm J, Lin F, Kobinger G, Bello A, Gregg DA, Broderick KE, Sardesai NY. Influenza A vaccines using linear expression cassettes delivered via electroporation afford full protection against challenge in a mouse model. Vaccine. 2012; 30:6946–6954.
Article17. Ogunremi O, Pasick J, Kobinger GP, Hannaman D, Berhane Y, Clavijo A, van Drunen Littel-van den Hurk S. A single electroporation delivery of a DNA vaccine containing the hemagglutinin gene of Asian H5N1 avian influenza virus generated a protective antibody response in chickens against a North American virus strain. Clin Vaccine Immunol. 2013; 20:491–500.
Article18. Zhao K, Shi X, Zhao Y, Wei H, Sun Q, Huang T, Zhang X, Wang Y. Preparation and immunological effectiveness of a swine influenza DNA vaccine encapsulated in chitosan nanoparticles. Vaccine. 2011; 29:8549–8556.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Zoonotic infections with avian influenza A viruses and vaccine preparedness: a game of "mix and match"
- Comparison of immunoadjuvant activities of four bursal peptides combined with H9N2 avian influenza virus vaccine
- Influenza Vaccine
- Enhancement of DNA Vaccine-induced Immune Responses by Influenza Virus NP Gene
- The significance of avian influenza virus mouse-adaptation and its application in characterizing the efficacy of new vaccines and therapeutic agents