Pediatr Gastroenterol Hepatol Nutr.
2015 Dec;18(4):230-237. 10.5223/pghn.2015.18.4.230.
Maladaptive Behavior and Gastrointestinal Disorders in Children with Autism Spectrum Disorder
- Affiliations
-
- 1Department of Child Health, Medical School, University of Indonesia, Jakarta, Indonesia.
- 2Department of Pediatrics, Universitair Ziekenhuis Brussel, Brussels, Belgium. yvan.vandenplas@uzbrussel.be
- KMID: 2383300
- DOI: http://doi.org/10.5223/pghn.2015.18.4.230
Abstract
- PURPOSE
Various gastrointestinal factors may contribute to maladaptive behavior in children with autism spectrum disorders (ASD). To determine the association between maladaptive behavior in children with ASD and gastrointestinal symptoms such as severity, intestinal microbiota, inflammation, enterocyte damage, permeability and absorption of opioid peptides.
METHODS
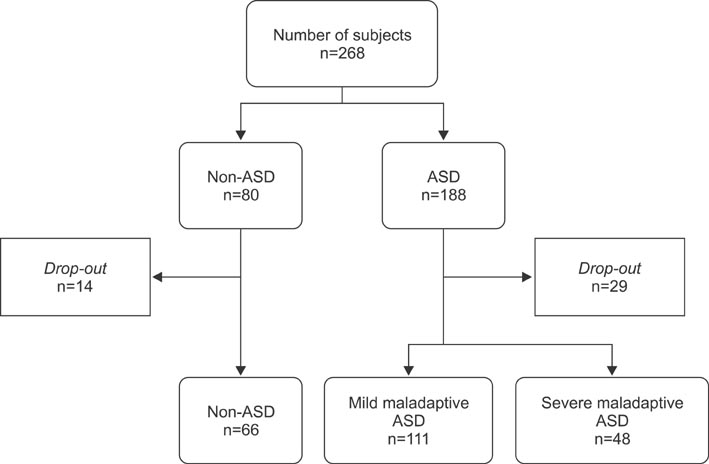
This observational cross-sectional study compared children with ASD to healthy controls, aged 2-10 years. Maladaptive behavior was classified using the Approach Withdrawal Problems Composite subtest of the Pervasive Developmental Disorder Behavior Inventory. Dependent variables were gastrointestinal symptom severity index, fecal calprotectin, urinary D-lactate, urinary lactulose/mannitol excretion, urinary intestinal fatty acids binding protein (I-FABP) and urinary opioid peptide excretion.
RESULTS
We did not find a significant difference between children with ASD with severe or mild maladaptive behavior and control subjects for gastrointestinal symptoms, fecal calprotectin, urinary D-lactate, and lactulose/mannitol ratio. Urinary opioid peptide excretion was absent in all children. Children with ASD with severe maladaptive behavior showed significantly higher urinary I-FABP levels compared to those with mild maladaptive behavior (p=0.019) and controls (p=0.015).
CONCLUSION
In our series, maladaptive behavior in ASD children was not associated with gastrointestinal symptoms, intestinal inflammation (no difference in calprotectin), microbiota (no difference in urinary D-lactate) and intestinal permeability (no difference in lactulose/manitol ratio). ASD children with severe maladaptive behavior have significantly more enterocyte damage (increased urinary I-FABP) than ASD children with mild maladaptive behavior and normal children.
Keyword
MeSH Terms
Figure
Reference
-
1. Steyaert JG, De la Marche W. What's new in autism? Eur J Pediatr. 2008; 167:1091–1101.
Article2. Autism and Developmental Disabilities Monitoring Network Surveillance Year 2008 Principal Investigators. Centers for Disease Control and Prevention. Prevalence of autism spectrum disorders--Autism and Developmental Disabilities Monitoring Network, 14 sites, United States, 2008. MMWR Surveill Summ. 2012; 61:1–19.3. Stilp RL, Gernsbacher MA, Schweigert EK, Arneson CL, Goldsmith HH. Genetic variance for autism screening items in an unselected sample of toddler-age twins. J Am Acad Child Adolesc Psychiatry. 2010; 49:267–276.
Article4. Hertz-Picciotto I, Croen LA, Hansen R, Jones CR, van de Water J, Pessah IN. The CHARGE study: an epidemiologic investigation of genetic and environmental factors contributing to autism. Environ Health Perspect. 2006; 114:1119–1125.
Article5. Currenti SA. Understanding and determining the etiology of autism. Cell Mol Neurobiol. 2010; 30:161–171.
Article6. Smith RA, Farnworth H, Wright B, Allgar V. Are there more bowel symptoms in children with autism compared to normal children and children with other developmental and neurological disorders?: A case control study. Autism. 2009; 13:343–355.
Article7. Gorrindo P, Williams KC, Lee EB, Walker LS, McGrew SG, Levitt P. Gastrointestinal dysfunction in autism: parental report, clinical evaluation, and associated factors. Autism Res. 2012; 5:101–108.
Article8. Ibrahim SH, Voigt RG, Katusic SK, Weaver AL, Barbaresi WJ. Incidence of gastrointestinal symptoms in children with autism: a population-based study. Pediatrics. 2009; 124:680–686.
Article9. Mouridsen SE, Rich B, Isager T. A longitudinal study of gastrointestinal diseases in individuals diagnosed with infantile autism as children. Child Care Health Dev. 2010; 36:437–443.
Article10. Reichelt KL, Knivsberg AM. Can the pathophysiology of autism be explained by the nature of the discovered urine peptides? Nutr Neurosci. 2003; 6:19–28.
Article11. Chaidez V, Hansen RL, Hertz-Picciotto I. Gastrointestinal problems in children with autism, developmental delays or typical development. J Autism Dev Disord. 2014; 44:1117–1127.
Article12. Wang LW, Tancredi DJ, Thomas DW. The prevalence of gastrointestinal problems in children across the United States with autism spectrum disorders from families with multiple affected members. J Dev Behav Pediatr. 2011; 32:351–360.
Article13. Kemperman RF, Muskiet FD, Boutier AI, Kema IP, Muskiet FA. Brief report: normal intestinal permeability at elevated platelet serotonin levels in a subgroup of children with pervasive developmental disorders in Curaçao (The Netherlands antilles). J Autism Dev Disord. 2008; 38:401–406.
Article14. Handen BL, Melmed RD, Hansen RL, Aman MG, Burnham DL, Bruss JB, et al. A double-blind, placebo-controlled trial of oral human immunoglobulin for gastrointestinal dysfunction in children with autistic disorder. J Autism Dev Disord. 2009; 39:796–805.
Article15. American Psychiatric Association. Diagnostic and statistical manual of mental disorders IV-TR. Washington DC: American Psychiatric Association;2000.16. Cohen IL, Sudhalter V. PDD behavior inventory. Professional manual. Florida: Psychological Assessment Resources, Inc.;2005.17. Centers for Disease Control and Prevention. National Center for Health Statistics. Clinical Growth Chart: United States [Internet]. Atlanta (GA): Centers for Disease Control and Prevention;2000. cited 2012 June 1. Available from: http://www.cdc.gov/growthcharts.18. Sjarif DR, Lestari ED, Mexitalia M, Nasar SS. Textbook of pediatric nutrition and metabolic diseases. Jakarta: Indonesian Pediatric Society;2011. p. 36–48.19. Waterlow JC. Classification and definition of protein-calorie malnutrition. Br Med J. 1972; 3:566–569.
Article20. Vreugdenhil AC, Wolters VM, Adriaanse MP, Van den Neucker AM, van Bijnen AA, Houwen R, et al. Additional value of serum I-FABP levels for evaluating celiac disease activity in children. Scand J Gastroenterol. 2011; 46:1435–1441.
Article21. Grootjans J, Thuijls G, Verdam F, Derikx JP, Lenaerts K, Buurman WA. Non-invasive assessment of barrier integrity and function of the human gut. World J Gastrointest Surg. 2010; 2:61–69.
Article22. Dettmer K, Hanna D, Whetstone P, Hansen R, Hammock BD. Autism and urinary exogenous neuropeptides: development of an on-line SPE-HPLC-tandem mass spectrometry method to test the opioid excess theory. Anal Bioanal Chem. 2007; 388:1643–1651.
Article23. Jackson PB, Boccuto L, Skinner C, Collins JS, Neri G, Gurrieri F, et al. Further evidence that the rs1858830 C variant in the promoter region of the MET gene is associated with autistic disorder. Autism Res. 2009; 2:232–236.
Article24. Campbell DB, Buie TM, Winter H, Bauman M, Sutcliffe JS, Perrin JM, et al. Distinct genetic risk based on association of MET in families with co-occurring autism and gastrointestinal conditions. Pediatrics. 2009; 123:1018–1024.
Article25. Savino F, Castagno E, Calabrese R, Viola S, Oggero R, Miniero R. High faecal calprotectin levels in healthy, exclusively breast-fed infants. Neonatology. 2010; 97:299–304.
Article26. Prell C, Nagel D, Freudenberg F, Schwarzer A, Koletzko S. Comparison of three tests for faecal calprotectin in children and young adults: a retrospective monocentric study. BMJ Open. 2014; 4:e004558.
Article27. Fernell E, Fagerberg UL, Hellström PM. No evidence for a clear link between active intestinal inflammation and autism based on analyses of faecal calprotectin and rectal nitric oxide. Acta Paediatr. 2007; 96:1076–1079.
Article28. Buie T, Campbell DB, Fuchs GJ 3rd, Furuta GT, Levy J, Vandewater J, et al. Evaluation, diagnosis, and treatment of gastrointestinal disorders in individuals with ASDs: a consensus report. Pediatrics. 2010; 125:Suppl 1. S1–S18.
Article29. de Magistris L, Familiari V, Pascotto A, Sapone A, Frolli A, Iardino P, et al. Alterations of the intestinal barrier in patients with autism spectrum disorders and in their first-degree relatives. J Pediatr Gastroenterol Nutr. 2010; 51:418–424.
Article30. Gondalia SV, Palombo EA, Knowles SR, Cox SB, Meyer D, Austin DW. Molecular characterisation of gastrointestinal microbiota of children with autism (with and without gastrointestinal dysfunction) and their neurotypical siblings. Autism Res. 2012; 5:419–427.
Article31. Boaz RT, Joseph AJ, Kang G, Bose A. Intestinal permeability in normally nourished and malnourished children with and without diarrhea. Indian Pediatr. 2013; 50:152–153.
Article32. Cass H, Gringras P, March J, McKendrick I, O'Hare AE, Owen L, et al. Absence of urinary opioid peptides in children with autism. Arch Dis Child. 2008; 93:745–750.
Article33. MacFabe DF. Enteric short-chain fatty acids: microbial messengers of metabolism, mitochondria, and mind: implications in autism spectrum disorders. Microb Ecol Health Dis. 2015; 26:28177.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Sleep Problems in Autism Spectrum Disorder
- Psychosocial Treatments for Children with Autism Spectrum Disorder
- Environmental Factors in Autism and Autistic Spectrum Disorder
- Empathy Study in Rodent Model of Autism Spectrum Disorders
- An update on the cause and treatment of sleep disturbance in children and adolescents with autism spectrum disorder