J Periodontal Implant Sci.
2016 Jun;46(3):176-196. 10.5051/jpis.2016.46.3.176.
Evaluation of bone substitutes for treatment of peri-implant bone defects: biomechanical, histological, and immunohistochemical analyses in the rabbit tibia
- Affiliations
-
- 1Department of Oral and Maxillofacial Surgery and Implantology, Sagrado Coracao University - USC, Bauru, SP, Brazil.
- 2Department of Diagnosis and Surgery, Sao Paulo State University - UNESP School of Dentistry at Araraquara, Araraquara, SP, Brazil. molon.foar@yahoo.com.br
- 3Department of Health Sciences, Implantology Post Graduation Course, University Center of Araraquara - UNIARA Dental School, SP, Brazil.
- 4Department of Basic Sciences, Sao Paulo State University - UNESP School of Dentistry at Araçatuba, Araçatuba, SP, Brazil.
- 5Department of Surgery and Integrated Clinic, Sao Paulo State University - UNESP School of Dentistry at Araçatuba, Araçatuba, SP, Brazil.
- KMID: 2382982
- DOI: http://doi.org/10.5051/jpis.2016.46.3.176
Abstract
- PURPOSE
We sought to evaluate the effectiveness of bone substitutes in circumferential peri-implant defects created in the rabbit tibia.
METHODS
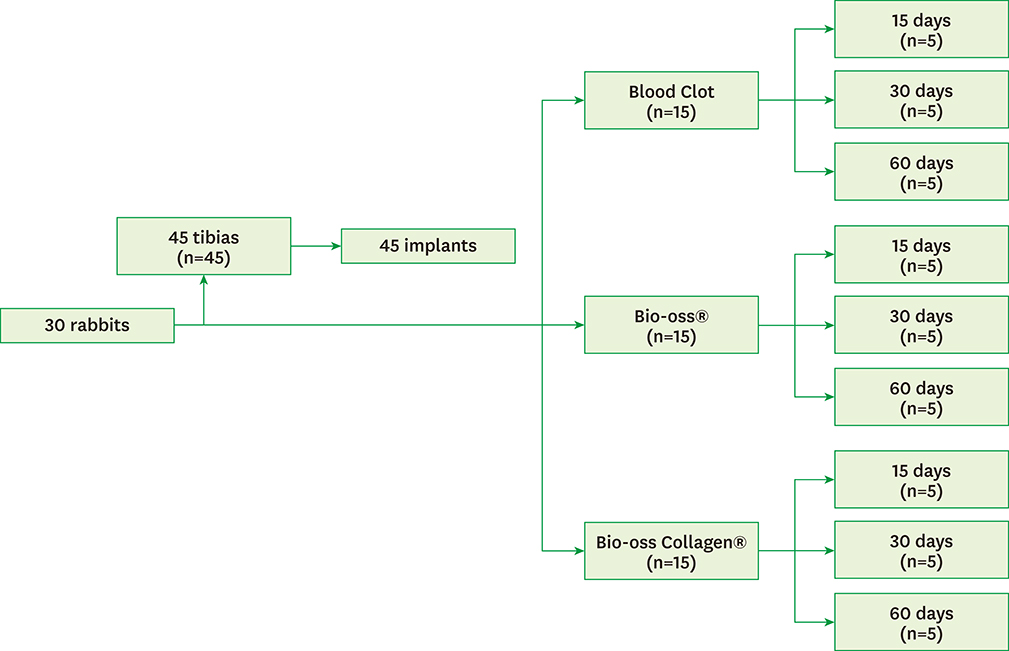
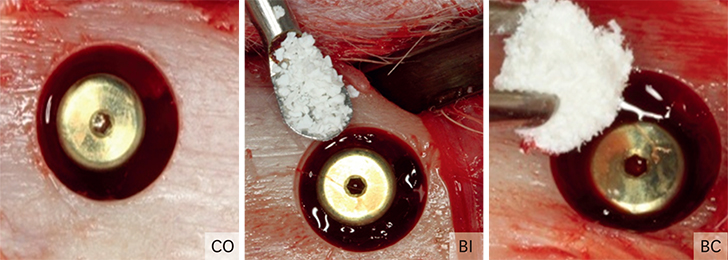
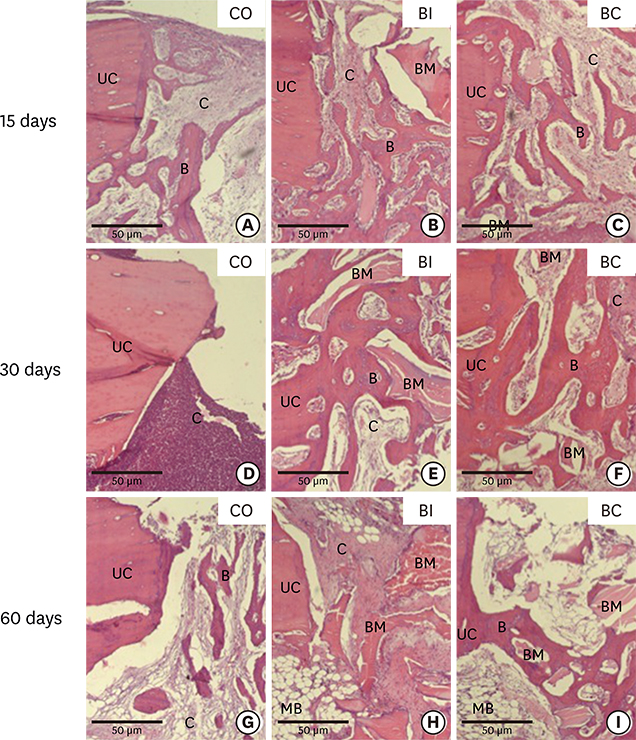
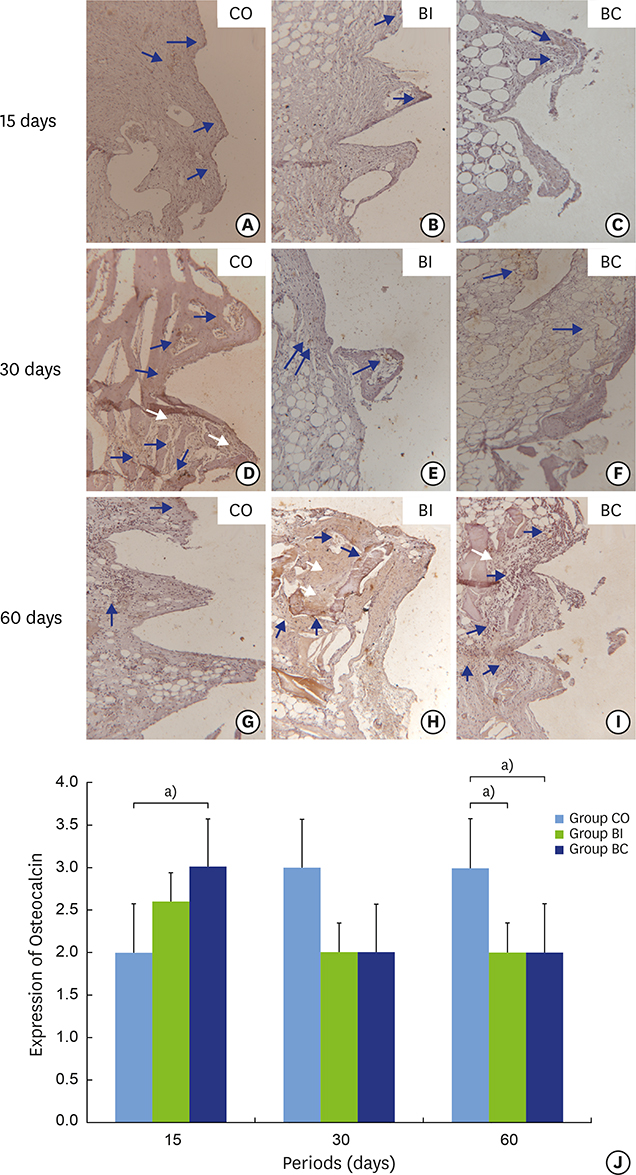
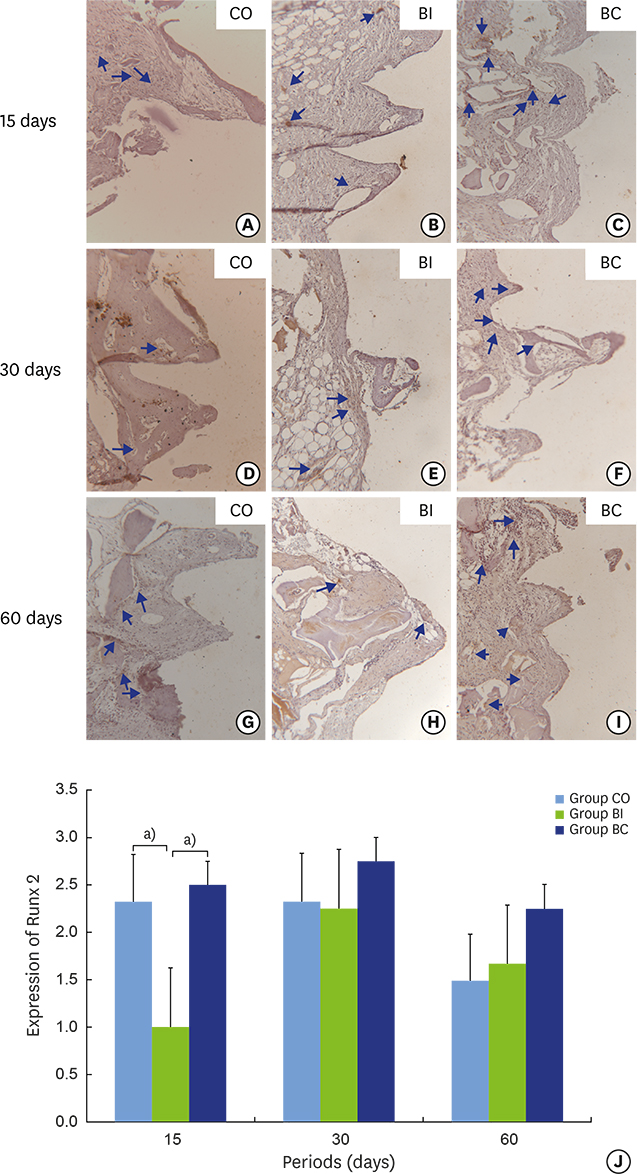
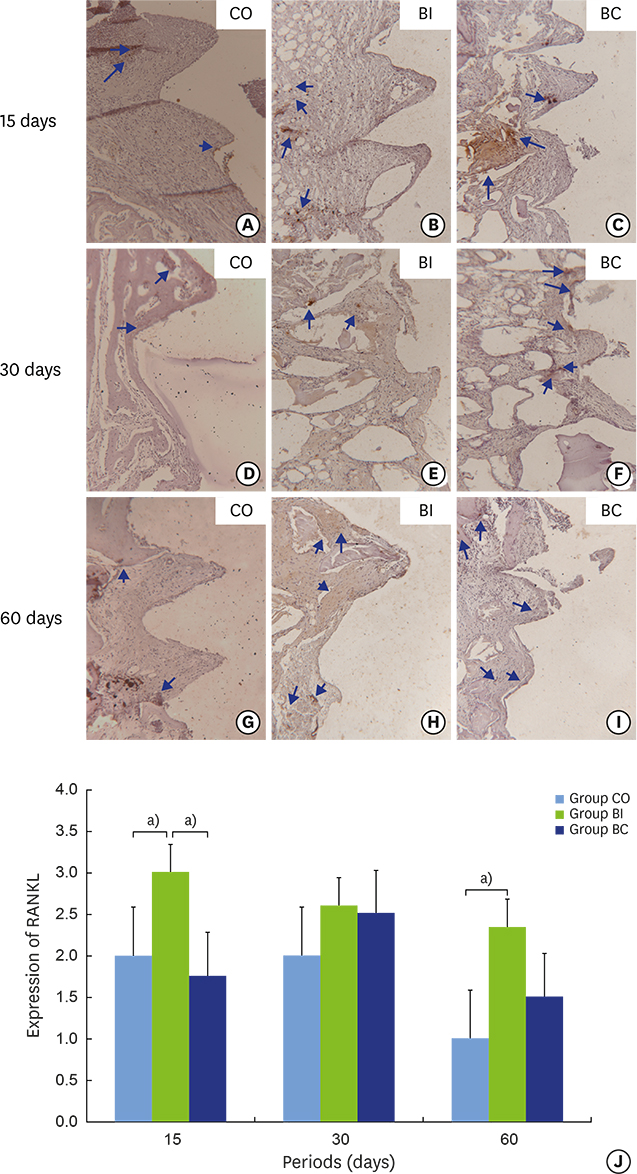
Thirty rabbits received 45 implants in their left and right tibia. A circumferential bone defect (6.1 mm in diameter/4 mm depth) was created in each rabbit tibia using a trephine bur. A dental implant (4.1 mm × 8.5 mm) was installed after the creation of the defect, providing a 2-mm gap. The bone defect gaps between the implant and the bone were randomly filled according to the following groups: blood clot (CO), particulate Bio-Oss® (BI), and Bio-Oss® Collagen (BC). Ten animals were euthanized after periods of 15, 30, and 60 days. Biomechanical analysis by means of the removal torque of the implants, as well as histologic and immunohistochemical analyses for protein expression of osteocalcin (OC), Runx2, OPG, RANKL, and TRAP were evaluated.
RESULTS
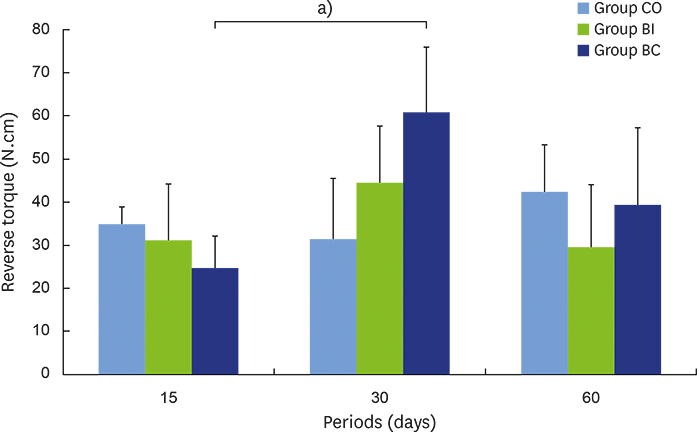
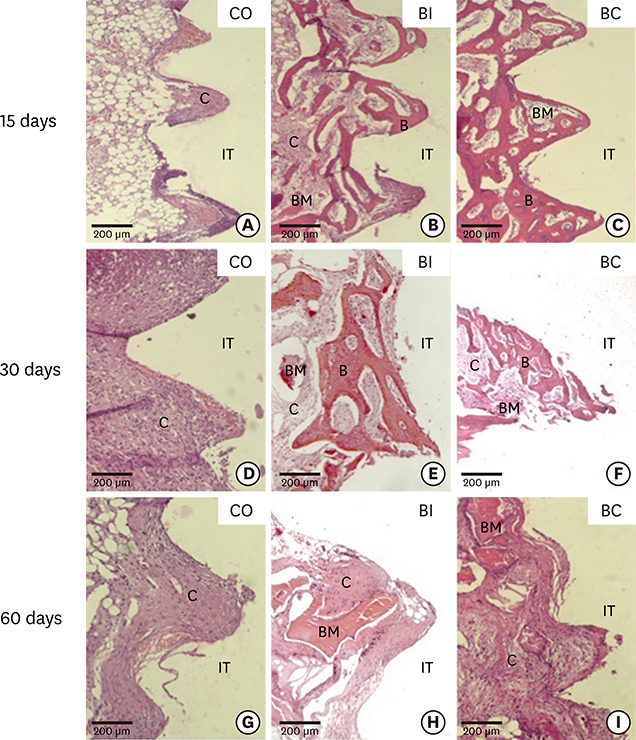
For biomechanics, BC showed a better biological response (61.00±15.28 Ncm) than CO (31.60±14.38 Ncm) at 30 days. Immunohistochemical analysis showed significantly different OC expression in CO and BC at 15 days, and also between the CO and BI groups, and between the CO and BC groups at 60 days. After 15 days, Runx2 expression was significantly different in the BI group compared to the CO and BC groups. RANKL expression was significantly different in the BI and CO groups and between the BI and BC groups at 15 days, and also between the BI and CO groups at 60 days. OPG expression was significantly higher at 60 days postoperatively in the BI group than the CO group.
CONCLUSIONS
Collectively, our data indicate that, compared to CO and BI, BC offered better bone healing, which was characterized by greater RUNX2, OC, and OPG immunolabeling, and required greater reversal torque for implant removal. Indeed, along with BI, BC presents promising biomechanical and biological properties supporting its possible use in osteoconductive grafts for filling peri-implant gaps.
MeSH Terms
Figure
Reference
-
1. Mardas N, Chadha V, Donos N. Alveolar ridge preservation with guided bone regeneration and a synthetic bone substitute or a bovine-derived xenograft: a randomized, controlled clinical trial. Clin Oral Implants Res. 2010; 21:688–698.
Article2. Brkovic BM, Prasad HS, Rohrer MD, Konandreas G, Agrogiannis G, Antunovic D, et al. Beta-tricalcium phosphate/type I collagen cones with or without a barrier membrane in human extraction socket healing: clinical, histologic, histomorphometric, and immunohistochemical evaluation. Clin Oral Investig. 2012; 16:581–590.
Article3. Shi B, Zhou Y, Wang YN, Cheng XR. Alveolar ridge preservation prior to implant placement with surgical-grade calcium sulfate and platelet-rich plasma: a pilot study in a canine model. Int J Oral Maxillofac Implants. 2007; 22:656–665.4. Schulte W, Heimke G. [The Tübinger immediate implant]. Quintessenz. 1976; 27:17–23.5. Botticelli D, Renzi A, Lindhe J, Berglundh T. Implants in fresh extraction sockets: a prospective 5-year follow-up clinical study. Clin Oral Implants Res. 2008; 19:1226–1232.
Article6. de Molon RS, de Avila ED, Cirelli JA, Mollo FA Jr, de Andrade MF, Filho LA, et al. A combined approach for the treatment of resorbed fresh sockets allowing immediate implant restoration: a 2-year follow-up. J Oral Implantol. 2015; 41:712–718.
Article7. de Molon RS, de Avila ED, de Barros-Filho LA, Ricci WA, Tetradis S, Cirelli JA, et al. Reconstruction of the alveolar buccal bone plate in compromised fresh socket after immediate implant placement followed by immediate provisionalization. J Esthet Restor Dent. 2015; 27:122–135.
Article8. Akimoto K, Becker W, Persson R, Baker DA, Rohrer MD, O'Neal RB. Evaluation of titanium implants placed into simulated extraction sockets: a study in dogs. Int J Oral Maxillofac Implants. 1999; 14:351–360.9. Botticelli D, Berglundh T, Persson LG, Lindhe J. Bone regeneration at implants with turned or rough surfaces in self-contained defects. An experimental study in the dog. J Clin Periodontol. 2005; 32:448–455.
Article10. Yoon HC, Choi JY, Jung UW, Bae EK, Choi SH, Cho KS, et al. Effects of different depths of gap on healing of surgically created coronal defects around implants in dogs: a pilot study. J Periodontol. 2008; 79:355–361.
Article11. Carmagnola D, Berglundh T, Araújo M, Albrektsson T, Lindhe J. Bone healing around implants placed in a jaw defect augmented with Bio-Oss. An experimental study in dogs. J Clin Periodontol. 2000; 27:799–805.
Article12. Araújo MG, Linder E, Lindhe J. Bio-Oss collagen in the buccal gap at immediate implants: a 6-month study in the dog. Clin Oral Implants Res. 2011; 22:1–8.
Article13. Araújo MG, Lindhe J. Ridge preservation with the use of Bio-Oss collagen: A 6-month study in the dog. Clin Oral Implants Res. 2009; 20:433–440.
Article14. Han JY, Shin SI, Herr Y, Kwon YH, Chung JH. The effects of bone grafting material and a collagen membrane in the ridge splitting technique: an experimental study in dogs. Clin Oral Implants Res. 2011; 22:1391–1398.
Article15. Carvalho AC, Queiroz TP, Okamoto R, Margonar R, Garcia IR Jr, Magro Filho O. Evaluation of bone heating, immediate bone cell viability, and wear of high-resistance drills after the creation of implant osteotomies in rabbit tibias. Int J Oral Maxillofac Implants. 2011; 26:1193–1201.16. Queiroz TP, Hochuli-Vieira E, Gabrielli MA, Cancian DC. Use of bovine bone graft and bone membrane in defects surgically created in the cranial vault of rabbits. Histologic comparative analysis. Int J Oral Maxillofac Implants. 2006; 21:29–35.17. Queiroz TP, Souza FA, Guastaldi AC, Margonar R, Garcia-Júnior IR, Hochuli-Vieira E. Commercially pure titanium implants with surfaces modified by laser beam with and without chemical deposition of apatite. Biomechanical and topographical analysis in rabbits. Clin Oral Implants Res. 2013; 24:896–903.
Article18. Sisti KE, Piattelli A, Guastaldi AC, Queiroz TP, de Rossi R. Nondecalcified histologic study of bone response to titanium implants topographically modified by laser with and without hydroxyapatite coating. Int J Periodontics Restorative Dent. 2013; 33:689–696.
Article19. Souza FA, Queiroz TP, Guastaldi AC, Garcia-Júnior IR, Magro-Filho O, Nishioka RS, et al. Comparative in vivo study of commercially pure Ti implants with surfaces modified by laser with and without silicate deposition: biomechanical and scanning electron microscopy analysis. J Biomed Mater Res B Appl Biomater. 2013; 101:76–84.20. Souza FA, Queiroz TP, Sonoda CK, Okamoto R, Margonar R, Guastaldi AC, et al. Histometric analysis and topographic characterization of cp Ti implants with surfaces modified by laser with and without silica deposition. J Biomed Mater Res B Appl Biomater. 2014; 102:1677–1688.
Article21. Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. Osteoarthritis Cartilage. 2012; 20:256–260.
Article22. de Molon RS, Morais-Camilo JA, Verzola MH, Faeda RS, Pepato MT, Marcantonio E Jr. Impact of diabetes mellitus and metabolic control on bone healing around osseointegrated implants: removal torque and histomorphometric analysis in rats. Clin Oral Implants Res. 2013; 24:831–837.
Article23. dos Santos PL, Queiroz TP, Margonar R, Gomes de Souza Carvalho AC, Okamoto R, de Souza Faloni AP, et al. Guided implant surgery: what is the influence of this new technique on bone cell viability? J Oral Maxillofac Surg. 2013; 71:505–512.
Article24. Esteves JC, Marcantonio E Jr, de Souza Faloni AP, Rocha FR, Marcantonio RA, Wilk K, et al. Dynamics of bone healing after osteotomy with piezosurgery or conventional drilling - histomorphometrical, immunohistochemical, and molecular analysis. J Transl Med. 2013; 11:221.25. Queiroz TP, Souza FA, Okamoto R, Margonar R, Pereira-Filho VA, Garcia Júnior IR, et al. Evaluation of immediate bone-cell viability and of drill wear after implant osteotomies: immunohistochemistry and scanning electron microscopy analysis. J Oral Maxillofac Surg. 2008; 66:1233–1240.
Article26. de Souza Faloni AP, Schoenmaker T, Azari A, Katchburian E, Cerri PS, de Vries TJ, et al. Jaw and long bone marrows have a different osteoclastogenic potential. Calcif Tissue Int. 2011; 88:63–74.
Article27. de Avila ED, de Molon RS, de Assis Mollo F Jr, de Barros LA, Capelozza Filho L, de Almeida Cardoso M, et al. Multidisciplinary approach for the aesthetic treatment of maxillary lateral incisors agenesis: thinking about implants? Oral Surg Oral Med Oral Pathol Oral Radiol. 2012; 114:e22–8.
Article28. de Barros LA, de Almeida Cardoso M, de Avila ED, de Molon RS, Siqueira DF, Mollo-Junior FA, et al. Six-year follow-up of maxillary anterior rehabilitation with forced orthodontic extrusion: achieving esthetic excellence with a multidisciplinary approach. Am J Orthod Dentofacial Orthop. 2013; 144:607–615.
Article29. Sisti KE, de Rossi R, Antoniolli AM, Aydos RD, Guastaldi AC, Queiroz TP, et al. Surface and biomechanical study of titanium implants modified by laser with and without hydroxyapatite coating, in rabbits. J Oral Implantol. 2012; 38:231–237.
Article30. Lioubavina-Hack N, Lang NP, Karring T. Significance of primary stability for osseointegration of dental implants. Clin Oral Implants Res. 2006; 17:244–250.
Article31. Moriya K, Maruo Y, Minagi S. Does rotational strain at screw tightening affect the attainment or maintenance of osseointegration? Clin Oral Implants Res. 2006; 17:451–458.
Article32. Wong RW, Rabie AB. Effect of bio-oss collagen and collagen matrix on bone formation. Open Biomed Eng J. 2010; 4:71–76.
Article33. Antunes AA, Oliveira Neto P, de Santis E, Caneva M, Botticelli D, Salata LA. Comparisons between Bio-Oss(®) and Straumann(®) Bone Ceramic in immediate and staged implant placement in dogs mandible bone defects. Clin Oral Implants Res. 2013; 24:135–142.
Article34. Zambon R, Mardas N, Horvath A, Petrie A, Dard M, Donos N. The effect of loading in regenerated bone in dehiscence defects following a combined approach of bone grafting and GBR. Clin Oral Implants Res. 2012; 23:591–601.
Article35. Park IP, Kim SK, Lee SJ, Lee JH. The relationship between initial implant stability quotient values and bone-to-implant contact ratio in the rabbit tibia. J Adv Prosthodont. 2011; 3:76–80.
Article36. Carlsson L, Röstlund T, Albrektsson B, Albrektsson T. Implant fixation improved by close fit. Cylindrical implant-bone interface studied in rabbits. Acta Orthop Scand. 1988; 59:272–275.
Article37. Carmagnola D, Adriaens P, Berglundh T. Healing of human extraction sockets filled with Bio-Oss. Clin Oral Implants Res. 2003; 14:137–143.38. Artzi Z, Tal H, Dayan D. Porous bovine bone mineral in healing of human extraction sockets. Part 1: histomorphometric evaluations at 9 months. J Periodontol. 2000; 71:1015–1023.
Article39. Ducy P, Schinke T, Karsenty G. The osteoblast: a sophisticated fibroblast under central surveillance. Science. 2000; 289:1501–1504.
Article40. Thorwarth M, Rupprecht S, Falk S, Felszeghy E, Wiltfang J, Schlegel KA. Expression of bone matrix proteins during de novo bone formation using a bovine collagen and platelet-rich plasma (prp)--an immunohistochemical analysis. Biomaterials. 2005; 26:2575–2584.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Evaluation of bone graft materials in the treatment of artificial bone defects around dental implant
- Guided bone regeneration in peri-implant defects using a 1:1 mixture of cancellous and cortical freeze-dried bone allograft: A randomized controlled trial
- In Vivo reaction of the Highly Porous Glass Ceramics in the Rabbit Tibia: Radiological and Histological Analysis
- Radiological , Biomechanical and Histological Analysis on the Surgical Treatment of Bone Defect in Rabbit Tibia using Glass Ceramics
- Combined surgical therapy for the treatment of combined supraand intrabony defects in peri-implantitis