Int J Stem Cells.
2015 May;8(1):69-78. 10.15283/ijsc.2015.8.1.69.
Bone Marrow Mesenchymal Stem Cell Transplantation in a Rabbit Corneal Alkali Burn Model (A Histological and Immune Histo-chemical Study)
- Affiliations
-
- 1Department of Histology, Faculty of Medicine, Ain Shams University, Cairo, Egypt. saharhistology@gmail.com
- KMID: 2380800
- DOI: http://doi.org/10.15283/ijsc.2015.8.1.69
Abstract
- BACKGROUND
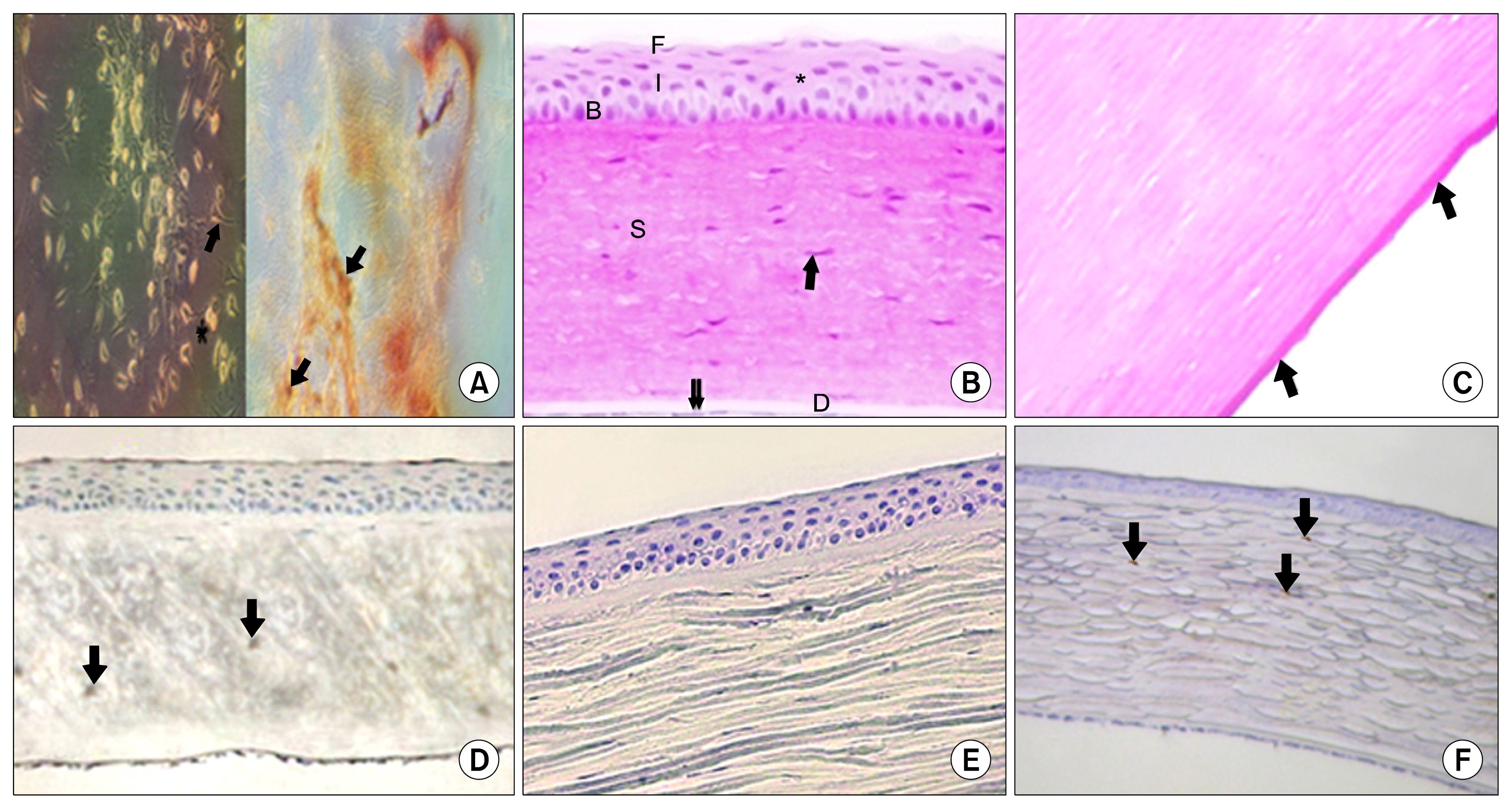
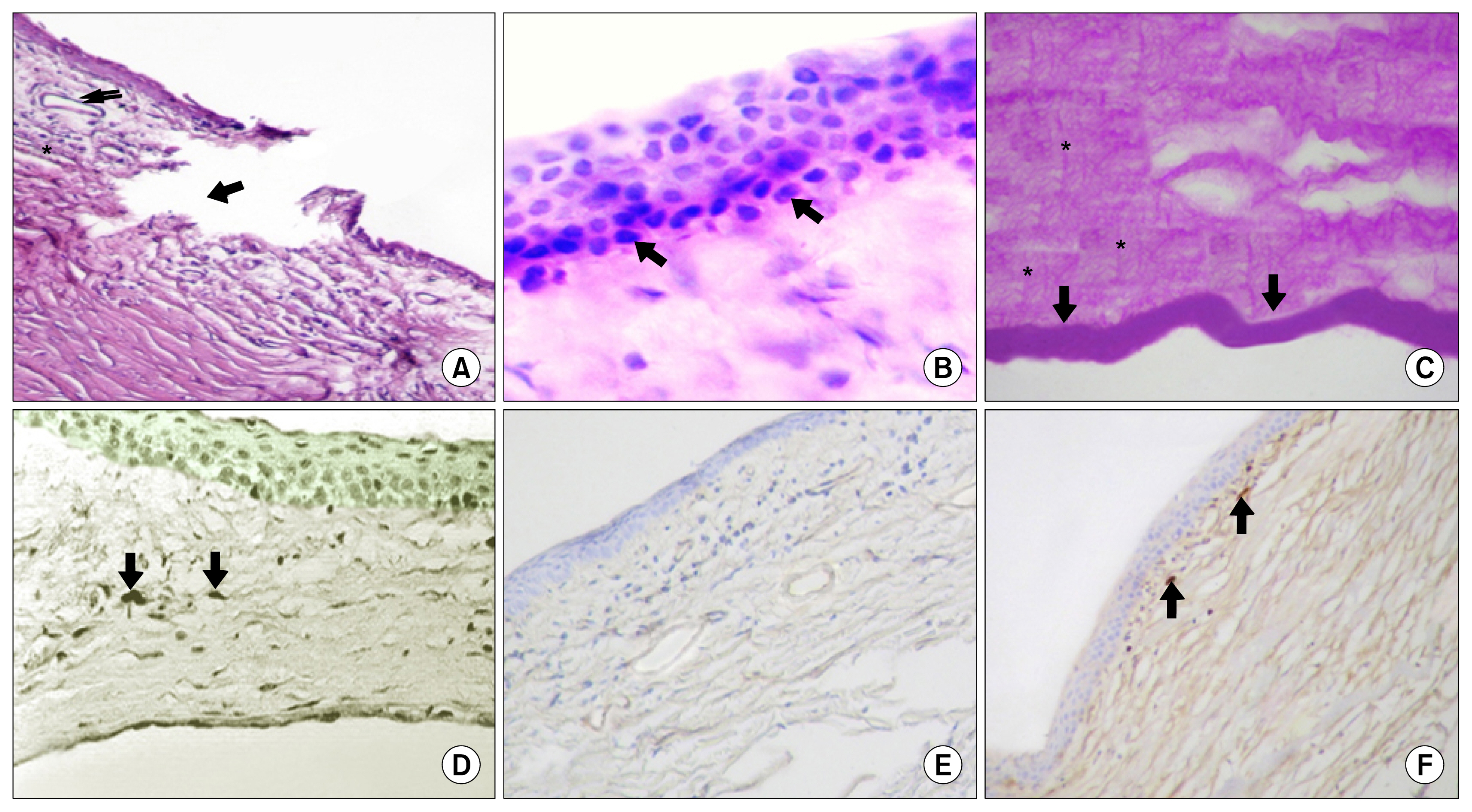
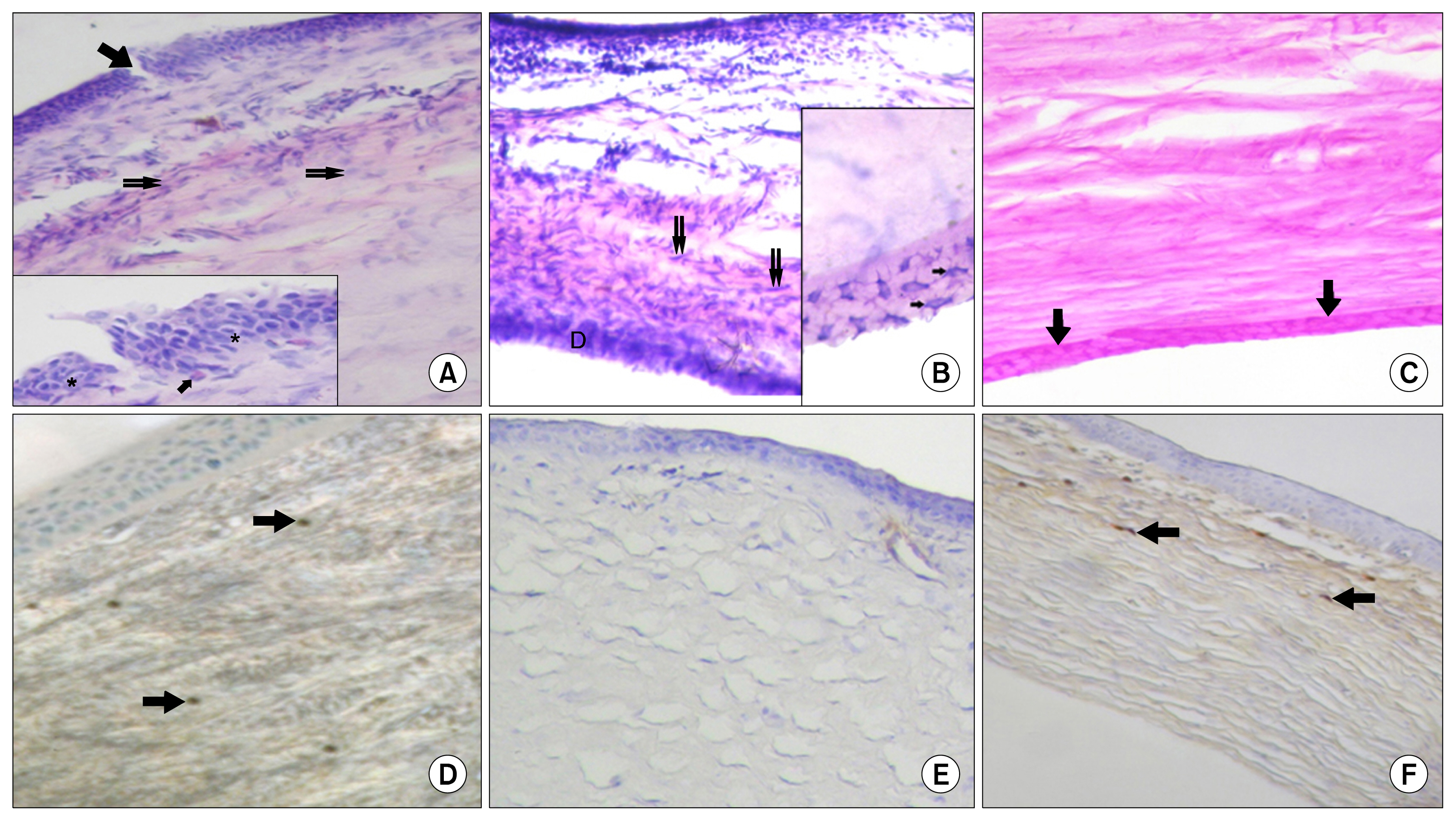
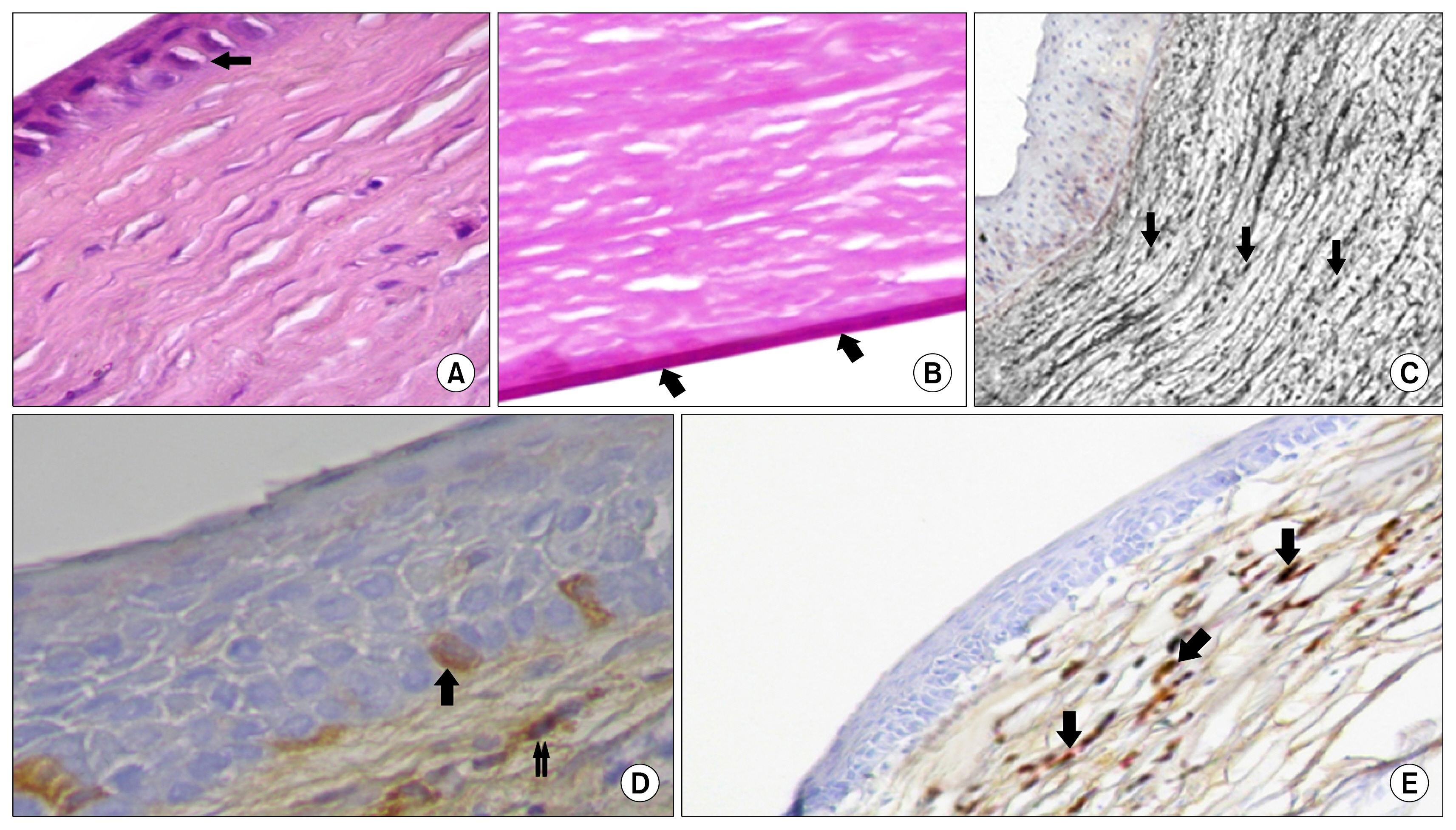
Alkali-burned corneas can seldom heal properly to restore corneal transparency. Treatment of this severe disorder of the ocular surface remains a challenge. AIM OF THE WORK: was to investigate whether systemically transplanted bone marrow mesenchymal stem cells (BM-MSCs) can promote corneal wound healing after alkali burn. MATERIAL AND METHODS: Thirty five male New Zealand rabbits were used in this study. The animals were divided into three groups. Group I; the control group was sham operated. Group II; corneal alkali burn was created. Group III; underwent corneal alkali burn then treated with BM-MSCs. All corneas were collected after fourteen and twenty eight days. Evaluation using H&E, PAS & alkaline phosphatase reaction was carried out. Immune histo-chemical staining for CD44 and vimentin was performed as well.
RESULTS
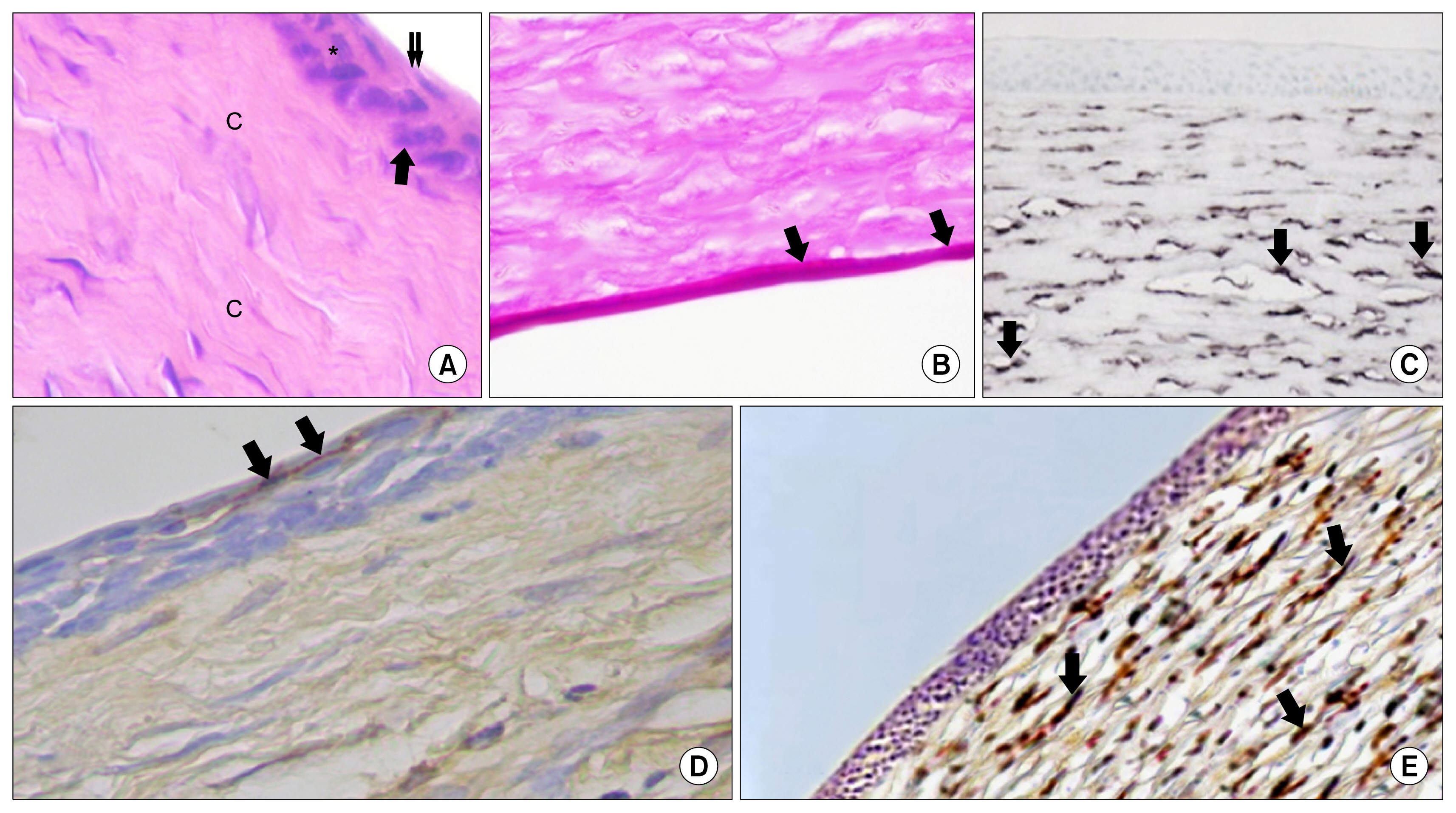
the corneal epithelium of (Group II) showed marked alterations. Vascularization, cellular infiltration and irregularity of the collagen fibers were also seen in the substantia propria. Increase in the thickness of the Descemet's membrane was noticed as well. On the other hand, at the time of 28 days, Group III rabbits showed best histological results with nearly healed corneas compared to other groups. Meanwhile, vimentin was more strongly expressed in Group III assessing the differentiating ability of BM-MSCs.
CONCLUSION
BM-MSCs could effectively promote corneal alkali burn healing.
MeSH Terms
Figure
Reference
-
References
1. Kao WW, Zhu G, Benza R, Kao CW, Ishizaki M, Wander AH. Appearance of immune cells and expression of MHC II DQ molecule by fibroblasts in alkali-burned corneas. Cornea. 1996; 15:397–408. DOI: 10.1097/00003226-199607000-00010. PMID: 8776566.
Article2. Tsai RJ, Li LM, Chen JK. Reconstruction of damaged corneas by transplantation of autologous limbal epithelial cells. N Engl J Med. 2000; 343:86–93. DOI: 10.1056/NEJM200007133430202. PMID: 10891515.
Article3. Tseng SC. Regulation and clinical implications of corneal epithelial stem cells. Mol Biol Rep. 1996; 23:47–58. DOI: 10.1007/BF00357072. PMID: 8983018.
Article4. Swift GJ, Aggarwal RK, Davis GJ, Coster DJ, Williams KA. Survival of rabbit limbal stem cell allografts. Transplantation. 1996; 62:568–574. DOI: 10.1097/00007890-199609150-00005. PMID: 8830817.5. Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science. 1999; 284:143–147. DOI: 10.1126/science.284.5411.143. PMID: 10102814.
Article6. Alhadlaq A, Mao JJ. Mesenchymal stem cells: isolation and therapeutics. Stem Cells Dev. 2004; 13:436–448. DOI: 10.1089/scd.2004.13.436. PMID: 15345137.
Article7. Campagnoli C, Roberts IA, Kumar S, Bennett PR, Bellantuono I, Fisk NM. Identification of mesenchymal stem/progenitor cells in human first-trimester fetal blood, liver, and bone marrow. Blood. 2001; 98:2396–2402. DOI: 10.1182/blood.V98.8.2396. PMID: 11588036.
Article8. Ye J, Yao K, Kim JC. Mesenchymal stem cell transplantation in a rabbit corneal alkali burn model: engraftment and involvement in wound healing. Eye (Lond). 2006; 20:482–490. DOI: 10.1038/sj.eye.6701913.
Article9. Wagoner MD. Chemical injuries of the eye: current concepts in pathophysiology and therapy. Surv Ophthalmol. 1997; 41:275–313. DOI: 10.1016/S0039-6257(96)00007-0. PMID: 9104767.
Article10. Levy SG, McCartney AC, Sawada H, Dopping-Hepenstal PJ, Alexander RA, Moss J. Descemet’s membrane in the iridocorneal-endothelial syndrome: morphology and composition. Exp Eye Res. 1995; 61:323–333. DOI: 10.1016/S0014-4835(05)80127-7. PMID: 7556496.
Article11. Leung EW, Rife L, Smith RE, Kay EP. Extracellular matrix components in retrocorneal fibrous membrane in comparison to corneal endothelium and Descemet’s membrane. Mol Vis. 2000; 6:15–23. PMID: 10731515.12. Heindl LM, Schlötzer-Schrehardt U, Cursiefen C, Bachmann BO, Hofmann-Rummelt C, Kruse FE. Myofibroblast metaplasia after descemet membrane endothelial keratoplasty. Am J Ophthalmol. 2011; 151:1019–1023.e2. DOI: 10.1016/j.ajo.2010.11.032. PMID: 21457928.
Article13. Coulson-Thomas VJ, Caterson B, Kao WW. Transplantation of human umbilical mesenchymal stem cells cures the corneal defects of mucopolysaccharidosis VII mice. Stem Cells. 2013; 31:2116–2226. DOI: 10.1002/stem.1481. PMID: 23897660. PMCID: 3812352.
Article14. Yao L, Li ZR, Su WR, Li YP, Lin ML, Zhang WX, Liu Y, Wan Q, Liang D. Role of mesenchymal stem cells on cornea wound healing induced by acute alkali burn. PLoS One. 2012; 7:e30842. DOI: 10.1371/journal.pone.0030842. PMID: 22363499. PMCID: 3281878.
Article15. Ma Y, Xu Y, Xiao Z, Yang W, Zhang C, Song E, Du Y, Li L. Reconstruction of chemically burned rat corneal surface by bone marrow-derived human mesenchymal stem cells. Stem Cells. 2006; 24:315–321. DOI: 10.1634/stemcells.2005-0046.
Article16. Hannoush EJ, Sifri ZC, Elhassan IO, Mohr AM, Alzate WD, Offin M, Livingston DH. Impact of enhanced mobilization of bone marrow derived cells to site of injury. J Trauma. 2011; 71:283–289. discussion 289–291. DOI: 10.1097/TA.0b013e318222f380. PMID: 21825928.
Article17. Smith H, Whittall C, Weksler B, Middleton J. Chemokines stimulate bidirectional migration of human mesenchymal stem cells across bone marrow endothelial cells. Stem Cells Dev. 2012; 21:476–486. DOI: 10.1089/scd.2011.0025.
Article18. Joo YO. The Anti-Inflammatory protein TSG-6 reduces inflammatory damage to the cornea following chemical injury. Am J Ophthalmo. 2012; 107:459–467.19. Zhang W, Ge W, Li C, You S, Liao L, Han Q, Deng W, Zhao RC. Effects of mesenchymal stem cells on differentiation, maturation, and function of human monocyte-derived dendritic cells. Stem Cells Dev. 2004; 13:263–271. DOI: 10.1089/154732804323099190. PMID: 15186722.
Article20. Holan V, Javorkova E. Mesenchymal stem cells, nanofiber scaffolds and ocular surface reconstruction. Stem Cell Rev. 2013; 9:609–619. DOI: 10.1007/s12015-013-9449-0. PMID: 23733704.
Article21. Pinnamaneni N, Funderburgh JL. Concise review: Stem cells in the corneal stroma. Stem Cells. 2012; 30:1059–1063. DOI: 10.1002/stem.1100. PMID: 22489057. PMCID: 3580383.
Article22. Osathanon T, Nowwarote N, Manokawinchoke J, Pavasant P. bFGF and JAGGED1 regulate alkaline phosphatase expression and mineralization in dental tissue-derived mesenchymal stem cells. J Cell Biochem. 2013; 114:2551–2561. DOI: 10.1002/jcb.24602. PMID: 23749297.
Article23. Alho AM, Underhill CB. The hyaluronate receptor is preferentially expressed on proliferating epithelial cells. J Cell Biol. 1989; 108:1557–1565. DOI: 10.1083/jcb.108.4.1557. PMID: 2466850. PMCID: 2115515.
Article24. Chen J, Li Y, Wang L, Zhang Z, Lu D, Lu M, Chopp M. Therapeutic benefit of intravenous administration of bone marrow stromal cells after cerebral ischemia in rats. Stroke. 2001; 32:1005–1011. DOI: 10.1161/01.STR.32.4.1005. PMID: 11283404.
Article25. Liu H, Zhang J, Liu CY, Hayashi Y, Kao WW. Bone marrow mesenchymal stem cells can differentiate and assume corneal keratocyte phenotype. J Cell Mol Med. 2012; 16:1114–1124. DOI: 10.1111/j.1582-4934.2011.01418.x.
Article26. Paulin-Levasseur M. Intermediate filament proteins: many unanswered questions, few unquestioned answers. Biochem Cell Biol. 1992; 70:842–848. DOI: 10.1139/o92-132. PMID: 1297350.
Article27. Risen LA, Binder PS, Nayak SK. Intermediate filaments and their organization in human corneal endothelium. Invest Ophthalmol Vis Sci. 1987; 28:1933–1938. PMID: 3316108.28. Chaurasia SS, Kaur H, de Medeiros FW, Smith SD, Wilson SE. Dynamics of the expression of intermediate filaments vimentin and desmin during myofibroblast differentiation after corneal injury. Exp Eye Res. 2009; 89:133–139. DOI: 10.1016/j.exer.2009.02.022. PMID: 19285070. PMCID: 2716066.
Article29. Ishizaki M, Zhu G, Haseba T, Shafer SS, Kao WW. Expression of collagen I, smooth muscle alpha-actin, and vimentin during the healing of alkali-burned and lacerated corneas. Invest Ophthalmol Vis Sci. 1993; 34:3320–3328. PMID: 8225867.30. Wilson SE. Analysis of the keratocyte apoptosis, keratocyte proliferation, and myofibroblast transformation responses after photorefractive keratectomy and laser in situ kerato-mileusis. Trans Am Ophthalmol Soc. 2002; 100:411–433.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical Safety and Efficacy of Autologous Bone Marrow-Derived Mesenchymal Stem Cell Transplantation in Sensorineural Hearing Loss Patients
- Effects of 0.1% Dexamethasone on Experimental corneal Epithelial Healing Following Alkali Wounds
- Effect of Solcoseryl in Corneal Alkali Burn Rat Model
- Evans Syndrome Following Unrelated Bone Marrow Transplantation
- Endothelial F-actin changes in the alkali burned rabbit cornea