Korean J Physiol Pharmacol.
2016 Jul;20(4):325-332. 10.4196/kjpp.2016.20.4.325.
Resveratrol attenuates lipopolysaccharide-induced dysfunction of blood-brain barrier in endothelial cells via AMPK activation
- Affiliations
-
- 1Department of Biomedical Engineering, College of Engineering, Peking University, Beijing 100871, China. hu15084737958@139.com
- 2Deapartment of Orthopaedics, The Third Xiangya Hospital, Central South University, Changsha 410013, China. Liu1367251@163.com
- KMID: 2371049
- DOI: http://doi.org/10.4196/kjpp.2016.20.4.325
Abstract
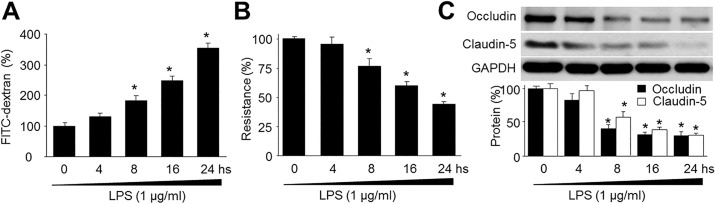
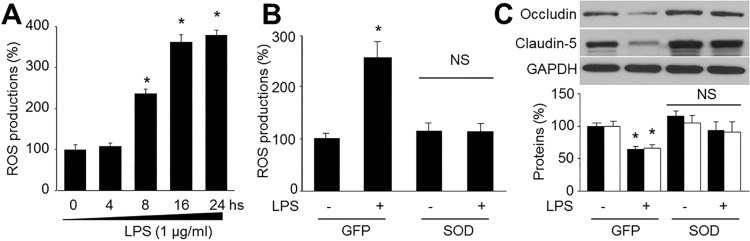
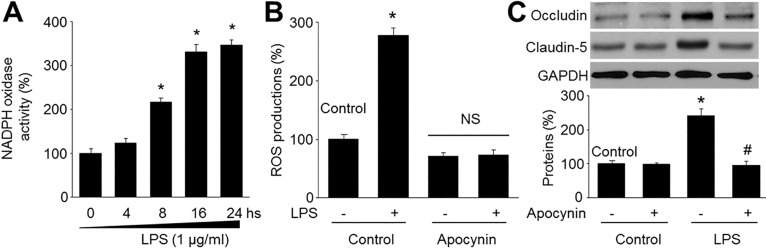
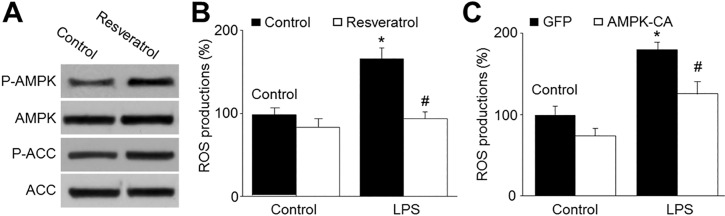
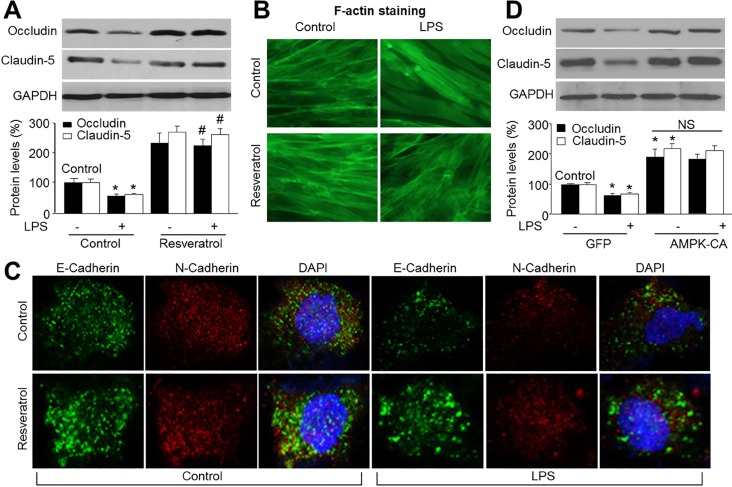
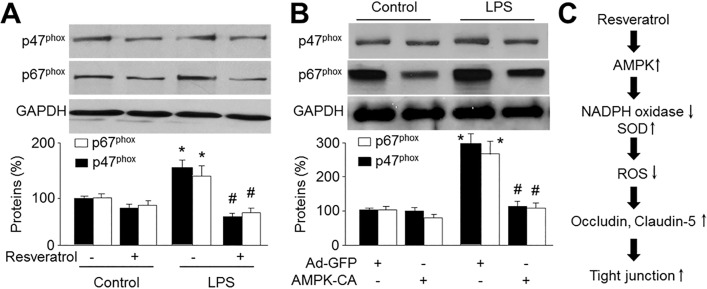
- Resveratrol, a phytoalexin, is reported to activate AMP-activated protein kinase (AMPK) in vascular cells. The blood-brain barrier (BBB), formed by specialized brain endothelial cells that are interconnected by tight junctions, strictly regulates paracellular permeability to maintain an optimal extracellular environment for brain homeostasis. The aim of this study was to elucidate the effects of resveratrol and the role of AMPK in BBB dysfunction induced by lipopolysaccharide (LPS). Exposure of human brain microvascular endothelial cells (HBMECs) to LPS (1 µg/ml) for 4 to 24 hours week dramatically increased the permeability of the BBB in parallel with lowered expression levels of occluding and claudin-5, which are essential to maintain tight junctions in HBMECs. In addition, LPS significantly increased the reactive oxygen species (ROS) productions. All effects induced by LPS in HBVMCs were reversed by adenoviral overexpression of superoxide dismutase, inhibition of NAD(P) H oxidase by apocynin or gain-function of AMPK by adenoviral overexpression of constitutively active mutant (AMPK-CA) or by resveratrol. Finally, upregulation of AMPK by either AMPK-CA or resveratrol abolished the levels of LPS-enhanced NAD(P)H oxidase subunits protein expressions. We conclude that AMPK activation by resveratrol improves the integrity of the BBB disrupted by LPS through suppressing the induction of NAD(P)H oxidase-derived ROS in HBMECs.
Keyword
MeSH Terms
-
AMP-Activated Protein Kinases*
Blood-Brain Barrier*
Brain
Claudin-5
Endothelial Cells*
Homeostasis
Humans
NADPH Oxidase
Oxidoreductases
Permeability
Reactive Oxygen Species
Superoxide Dismutase
Tight Junctions
Up-Regulation
AMP-Activated Protein Kinases
Claudin-5
NADPH Oxidase
Oxidoreductases
Reactive Oxygen Species
Superoxide Dismutase
Figure
Reference
-
1. Lee BH, Choi SH, Hwang SH, Kim HJ, Lee JH, Nah SY. Resveratrol inhibits GABAC ρ receptor-mediated ion currents expressed in xenopus oocytes. Korean J Physiol Pharmacol. 2013; 17:175–180. PMID: 23626481.2. Xu Q, Si LY. Resveratrol role in cardiovascular and metabolic health and potential mechanisms of action. Nutr Res. 2012; 32:648–658. PMID: 23084637.
Article3. Signorelli P, Ghidoni R. Resveratrol as an anticancer nutrient: molecular basis, open questions and promises. J Nutr Biochem. 2005; 16:449–466. PMID: 16043028.
Article4. Jang M, Cai L, Udeani GO, Slowing KV, Thomas CF, Beecher CW, Fong HH, Farnsworth NR, Kinghorn AD, Mehta RG, Moon RC, Pezzuto JM. Cancer chemopreventive activity of resveratrol, a natural product derived from grapes. Science. 1997; 275:218–220. PMID: 8985016.
Article5. Hamza SM, Dyck JR. Systemic and renal oxidative stress in the pathogenesis of hypertension: modulation of long-term control of arterial blood pressure by resveratrol. Front Physiol. 2014; 5:292. PMID: 25140155.
Article6. Cheng PW, Ho WY, Su YT, Lu PJ, Chen BZ, Cheng WH, Lu WH, Sun GC, Yeh TC, Hsiao M, Tseng CJ. Resveratrol decreases fructose-induced oxidative stress, mediated by NADPH oxidase via an AMPK-dependent mechanism. Br J Pharmacol. 2014; 171:2739–2750. PMID: 24547812.
Article7. Rahman MA, Kim NH, Kim SH, Oh SM, Huh SO. Antiproliferative and cytotoxic effects of resveratrol in mitochondria-mediated apoptosis in rat b103 neuroblastoma cells. Korean J Physiol Pharmacol. 2012; 16:321–326. PMID: 23118555.
Article8. Hawkins BT, Davis TP. The blood-brain barrier/neurovascular unit in health and disease. Pharmacol Rev. 2005; 57:173–185. PMID: 15914466.
Article9. Matter K, Balda MS. Signalling to and from tight junctions. Nat Rev Mol Cell Biol. 2003; 4:225–236. PMID: 12612641.
Article10. Feldman GJ, Mullin JM, Ryan MP. Occludin: structure, function and regulation. Adv Drug Deliv Rev. 2005; 57:883–917. PMID: 15820558.
Article11. Wang S, Xu J, Song P, Viollet B, Zou MH. In vivo activation of AMP-activated protein kinase attenuates diabetes-enhanced degradation of GTP cyclohydrolase I. Diabetes. 2009; 58:1893–1901. PMID: 19528375.
Article12. Wang S, Zhang C, Zhang M, Liang B, Zhu H, Lee J, Viollet B, Xia L, Zhang Y, Zou MH. Activation of AMP-activated protein kinase α2 by nicotine instigates formation of abdominal aortic aneurysms in mice in vivo. Nat Med. 2012; 18:902–910. PMID: 22561688.
Article13. Kim SJ, Lee JH, Chung HS, Song JH, Ha J, Bae H. Neuroprotective effects of AMP-activated protein kinase on scopolamine induced memory impairment. Korean J Physiol Pharmacol. 2013; 17:331–338. PMID: 23946693.
Article14. Price NL, Gomes AP, Ling AJ, Duarte FV, Martin-Montalvo A, North BJ, Agarwal B, Ye L, Ramadori G, Teodoro JS, Hubbard BP, Varela AT, Davis JG, Varamini B, Hafner A, Moaddel R, Rolo AP, Coppari R, Palmeira CM, de Cabo R, Baur JA, Sinclair DA. SIRT1 is required for AMPK activation and the beneficial effects of resveratrol on mitochondrial function. Cell Metab. 2012; 15:675–690. PMID: 22560220.
Article15. Song P, Wang S, He C, Wang S, Liang B, Viollet B, Zou MH. AMPKα2 deletion exacerbates neointima formation by upregulating Skp2 in vascular smooth muscle cells. Circ Res. 2011; 109:1230–1239. PMID: 21980125.
Article16. Wang S, Zhang M, Liang B, Xu J, Xie Z, Liu C, Viollet B, Yan D, Zou MH. AMPKalpha2 deletion causes aberrant expression and activation of NAD(P)H oxidase and consequent endothelial dysfunction in vivo: role of 26S proteasomes. Circ Res. 2010; 106:1117–1128. PMID: 20167927.17. Cardoso FL, Kittel A, Veszelka S, Palmela I, Tóth A, Brites D, Deli MA, Brito MA. Exposure to lipopolysaccharide and/or unconjugated bilirubin impair the integrity and function of brain microvascular endothelial cells. PLoS One. 2012; 7:e35919. PMID: 22586454.
Article18. Wang S, Xu J, Song P, Wu Y, Zhang J, Chul Choi H, Zou MH. Acute inhibition of guanosine triphosphate cyclohydrolase 1 uncouples endothelial nitric oxide synthase and elevates blood pressure. Hypertension. 2008; 52:484–490. PMID: 18645049.
Article19. Liu C, Wu J, Zou MH. Activation of AMP-activated protein kinase alleviates high-glucose-induced dysfunction of brain microvascular endothelial cell tight-junction dynamics. Free Radic Biol Med. 2012; 53:1213–1221. PMID: 22796592.
Article20. Liu WY, Wang ZB, Zhang LC, Wei X, Li L. Tight junction in blood-brain barrier: an overview of structure, regulation, and regulator substances. CNS Neurosci Ther. 2012; 18:609–615. PMID: 22686334.
Article21. Schreibelt G, Kooij G, Reijerkerk A, van Doorn R, Gringhuis SI, van der Pol S, Weksler BB, Romero IA, Couraud PO, Piontek J, Blasig IE, Dijkstra CD, Ronken E, de Vries HE. Reactive oxygen species alter brain endothelial tight junction dynamics via RhoA, PI3 kinase, and PKB signaling. FASEB J. 2007; 21:3666–3676. PMID: 17586731.
Article22. Song P, Zou MH. Regulation of NAD(P)H oxidases by AMPK in cardiovascular systems. Free Radic Biol Med. 2012; 52:1607–1619. PMID: 22357101.
Article23. Salminen A, Hyttinen JM, Kaarniranta K. AMP-activated protein kinase inhibits NF-κB signaling and inflammation: impact on healthspan and lifespan. J Mol Med (Berl). 2011; 89:667–676. PMID: 21431325.
Article24. Guo S, Yao Q, Ke Z, Chen H, Wu J, Liu C. Resveratrol attenuates high glucose-induced oxidative stress and cardiomyocyte apoptosis through AMPK. Mol Cell Endocrinol. 2015; 412:85–94. PMID: 26054749.
Article25. Yoshino J, Conte C, Fontana L, Mittendorfer B, Imai S, Schechtman KB, Gu C, Kunz I, Rossi Fanelli F, Patterson BW, Klein S. Resveratrol supplementation does not improve metabolic function in nonobese women with normal glucose tolerance. Cell Metab. 2012; 16:658–664. PMID: 23102619.
Article26. Penumathsa SV, Thirunavukkarasu M, Zhan L, Maulik G, Menon VP, Bagchi D, Maulik N. Resveratrol enhances GLUT-4 translocation to the caveolar lipid raft fractions through AMPK/Akt/eNOS signalling pathway in diabetic myocardium. J Cell Mol Med. 2008; 12:2350–2361. PMID: 18266981.
Article27. Gumbiner BM. Regulation of cadherin-mediated adhesion in morphogenesis. Nat Rev Mol Cell Biol. 2005; 6:622–634. PMID: 16025097.
Article28. Pasquier F, Boulogne A, Leys D, Fontaine P. Diabetes mellitus and dementia. Diabetes Metab. 2006; 32:403–414. PMID: 17110895.
Article29. Ristow M. Neurodegenerative disorders associated with diabetes mellitus. J Mol Med (Berl). 2004; 82:510–529. PMID: 15175861.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Retraction notice to: Resveratrol attenuates lipopolysaccharide-induced dysfunction of blood-brain barrier in endothelial cells via AMPK activation
- Radiation Induced Senescence-like Changes of Endothelial Cells and Blood-brain Barrier Disruption
- Resveratrol attenuates aging-induced mitochondrial dysfunction and mitochondria-mediated apoptosis in the rat heart
- Resveratrol at High Doses Acts as an Apoptotic Inducer in Endothelial Cells
- Glial and Vascular Cell Regulation of the Blood-Brain Barrier in Diabetes