Yonsei Med J.
2015 Jul;56(4):1134-1142. 10.3349/ymj.2015.56.4.1134.
Chemokine Signaling Pathway Involved in CCL2 Expression in Patients with Rheumatoid Arthritis
- Affiliations
-
- 1School of Dentistry, Tianjin Medical University, Tianjin, China. zy66@vip.sina.com
- 2Department of Nutrition and Food Hygiene, School of Public Health, Tianjin Medical University, Tianjin, China.
- 3Department of Infection and Rheumatology, General Hospital of Tianjin Medical University, Tianjin, China.
- 4Center of Joint Surgery, Tianjin Hospital, Tianjin, China.
- 5Department of Infection, Tianjin First Central Hospital, Tianjin, China.
- KMID: 2366358
- DOI: http://doi.org/10.3349/ymj.2015.56.4.1134
Abstract
- PURPOSE
Rheumatoid arthritis (RA) is an inflammatory joint disorder, the progression of which leads to the destruction of cartilage and bone. Chemokines are involved in RA pathogenesis. In this study, we investigated the chemokine signaling pathway associated with CCL2 in peripheral blood (PB) and synovial tissues (ST) of RA patients based on our previous work about chemokine signaling pathway involved in the activation of CCL2 production in collagen-induced arthritis rat ST.
MATERIALS AND METHODS
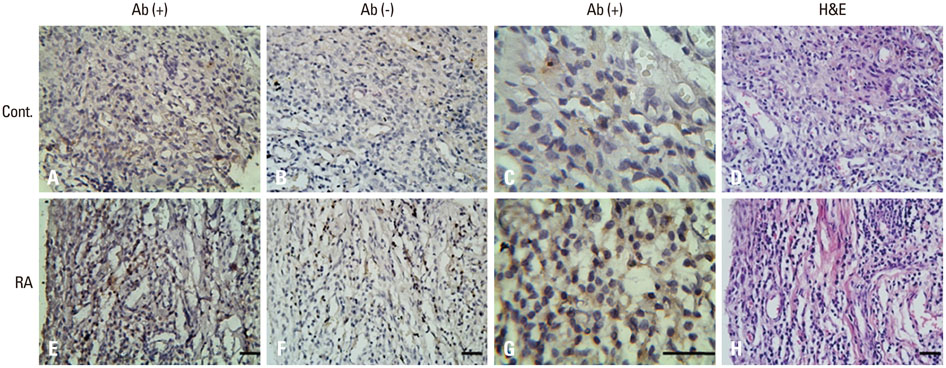
Total RNA was isolated from PB leukocytes and synovium of the knee joint in both RA patients and control populations. Real-time polymerase chain reaction was used to determine CCL4, CCR5, c-Jun, c-Fos, and CCL2 expressions. Serum level of CCL2 was assessed by enzyme-linked immunosorbent assay, and the production of CCL2 in ST was analyzed immunohistochemically.
RESULTS
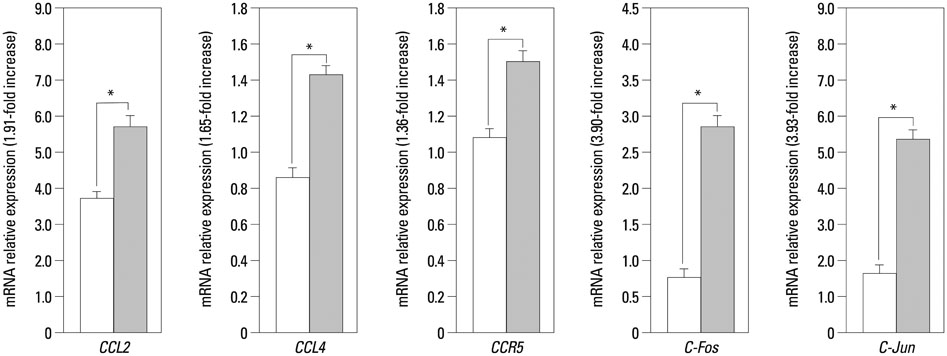
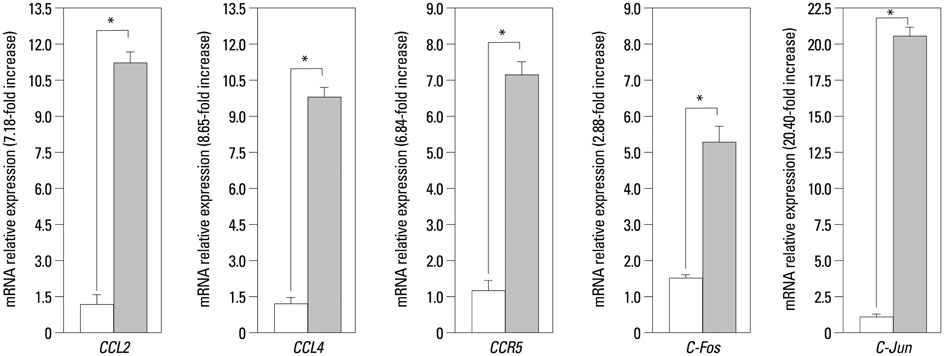
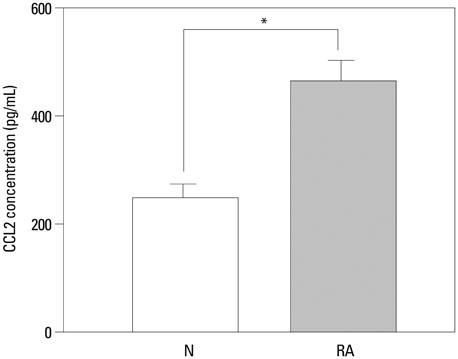
The expressions of CCL4, CCR5, c-Jun, c-Fos, and CCL2 messenger RNA in RA patients were significantly higher than those in healthy controls, both in ST and on PB leukocyte. Serum CCL2 levels were elevated in RA patients. Histological examination of rheumatoid joints revealed extensive CCL2 expression in RA ST.
CONCLUSION
CCL2, CCL4, c-Jun, c-Fos, and CCR5 may play an important role in the recruitment of PB leukocytes into the RA joints. These data provide evidence that the chemokine signaling pathway is involved in CCL2 expression in RA patient tissues, which may contribute to chronic inflammation associated with RA. Targeting this signaling pathway may provide a novel therapeutic avenue in RA.
Keyword
MeSH Terms
-
Adult
Animals
Arthritis, Rheumatoid/*blood/metabolism
Case-Control Studies
Chemokine CCL2/*blood/metabolism
Chemokines/metabolism
Enzyme-Linked Immunosorbent Assay
Female
Gene Expression
Humans
Male
Middle Aged
RNA, Messenger/genetics/metabolism
Rats
Real-Time Polymerase Chain Reaction
Signal Transduction
Synovial Membrane/*metabolism
Chemokine CCL2
Chemokines
RNA, Messenger
Figure
Reference
-
1. Firestein GS. Evolving concepts of rheumatoid arthritis. Nature. 2003; 423:356–361.
Article2. Szekanecz Z, Kim J, Koch AE. Chemokines and chemokine receptors in rheumatoid arthritis. Semin Immunol. 2003; 15:15–21.
Article3. Shahrara S, Amin MA, Woods JM, Haines GK, Koch AE. Chemokine receptor expression and in vivo signaling pathways in the joints of rats with adjuvant-induced arthritis. Arthritis Rheum. 2003; 48:3568–3583.
Article4. Dayer JM. The saga of the discovery of IL-1 and TNF and their specific inhibitors in the pathogenesis and treatment of rheumatoid arthritis. Joint Bone Spine. 2002; 69:123–132.
Article5. Akahoshi T, Wada C, Endo H, Hirota K, Hosaka S, Takagishi K, et al. Expression of monocyte chemotactic and activating factor in rheumatoid arthritis. Regulation of its production in synovial cells by interleukin-1 and tumor necrosis factor. Arthritis Rheum. 1993; 36:762–771.
Article6. Shahrara S, Proudfoot AE, Park CC, Volin MV, Haines GK, Woods JM, et al. Inhibition of monocyte chemoattractant protein-1 ameliorates rat adjuvant-induced arthritis. J Immunol. 2008; 180:3447–3456.
Article7. Schall TJ, Bacon K, Camp RD, Kaspari JW, Goeddel DV. Human macrophage inflammatory protein alpha (MIP-1 alpha) and MIP-1 beta chemokines attract distinct populations of lymphocytes. J Exp Med. 1993; 177:1821–1826.
Article8. Lundy SK, Sarkar S, Tesmer LA, Fox DA. Cells of the synovium in rheumatoid arthritis. T lymphocytes. Arthritis Res Ther. 2007; 9:202.9. Moreira AP, Cavassani KA, Massafera Tristão FS, Campanelli AP, Martinez R, Rossi MA, et al. CCR5-dependent regulatory T cell migration mediates fungal survival and severe immunosuppression. J Immunol. 2008; 180:3049–3056.
Article10. Katschke KJ Jr, Rottman JB, Ruth JH, Qin S, Wu L, LaRosa G, et al. Differential expression of chemokine receptors on peripheral blood, synovial fluid, and synovial tissue monocytes/macrophages in rheumatoid arthritis. Arthritis Rheum. 2001; 44:1022–1032.
Article11. Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 1988; 31:315–324.
Article12. Okamoto H, Cujec TP, Yamanaka H, Kamatani N. Molecular aspects of rheumatoid arthritis: role of transcription factors. FEBS J. 2008; 275:4463–4470.
Article13. Schonthaler HB, Guinea-Viniegra J, Wagner EF. Targeting inflammation by modulating the Jun/AP-1 pathway. Ann Rheum Dis. 2011; 70:Suppl 1. i109–i112.
Article14. Kok SH, Hong CY, Kuo MY, Wang CC, Hou KL, Lin YT, et al. Oncostatin M-induced CCL2 transcription in osteoblastic cells is mediated by multiple levels of STAT-1 and STAT-3 signaling: an implication for the pathogenesis of arthritis. Arthritis Rheum. 2009; 60:1451–1462.
Article15. Asahara H, Fujisawa K, Kobata T, Hasunuma T, Maeda T, Asanuma M, et al. Direct evidence of high DNA binding activity of transcription factor AP-1 in rheumatoid arthritis synovium. Arthritis Rheum. 1997; 40:912–918.
Article16. Zheng W, Li R, Pan H, He D, Xu R, Guo TB, et al. Role of osteopontin in induction of monocyte chemoattractant protein 1 and macrophage inflammatory protein 1beta through the NF-kappaB and MAPK pathways in rheumatoid arthritis. Arthritis Rheum. 2009; 60:1957–1965.
Article17. Wang N, Verna L, Hardy S, Forsayeth J, Zhu Y, Stemerman MB. Adenovirus-mediated overexpression of c-Jun and c-Fos induces intercellular adhesion molecule-1 and monocyte chemoattractant protein-1 in human endothelial cells. Arterioscler Thromb Vasc Biol. 1999; 19:2078–2084.
Article18. Kuroki Y, Shiozawa S, Yoshihara R, Hotta H. The contribution of human c-fos DNA to cultured synovial cells: a transfection study. J Rheumatol. 1993; 20:422–428.19. Gravallese EM, Darling JM, Ladd AL, Katz JN, Glimcher LH. In situ hybridization studies of stromelysin and collagenase messenger RNA expression in rheumatoid synovium. Arthritis Rheum. 1991; 34:1076–1084.
Article20. Yang-Yen HF, Chambard JC, Sun YL, Smeal T, Schmidt TJ, Drouin J, et al. Transcriptional interference between c-Jun and the glucocorticoid receptor: mutual inhibition of DNA binding due to direct protein-protein interaction. Cell. 1990; 62:1205–1215.
Article21. Villiger PM, Terkeltaub R, Lotz M. Production of monocyte chemoattractant protein-1 by inflamed synovial tissue and cultured synoviocytes. J Immunol. 1992; 149:722–727.22. Zhang L, Zhao J, Kuboyama N, Abiko Y. Low-level laser irradiation treatment reduces CCL2 expression in rat rheumatoid synovia via a chemokine signaling pathway. Lasers Med Sci. 2011; 26:707–717.
Article23. Schett G, Tohidast-Akrad M, Smolen JS, Schmid BJ, Steiner CW, Bitzan P, et al. Activation, differential localization, and regulation of the stress-activated protein kinases, extracellular signal-regulated kinase, c-JUN N-terminal kinase, and p38 mitogen-activated protein kinase, in synovial tissue and cells in rheumatoid arthritis. Arthritis Rheum. 2000; 43:2501–2512.
Article24. Angel P, Hattori K, Smeal T, Karin M. The jun proto-oncogene is positively autoregulated by its product, Jun/AP-1. Cell. 1988; 55:875–885.
Article25. Karin M. The regulation of AP-1 activity by mitogen-activated protein kinases. J Biol Chem. 1995; 270:16483–16486.
Article26. Wagner EF. Bone development and inflammatory disease is regulated by AP-1 (Fos/Jun). Ann Rheum Dis. 2010; 69:Suppl 1. i86–i88.
Article27. Lange-Carter CA, Johnson GL. Ras-dependent growth factor regulation of MEK kinase in PC12 cells. Science. 1994; 265:1458–1461.
Article28. Goebeler M, Kilian K, Gillitzer R, Kunz M, Yoshimura T, Bröcker EB, et al. The MKK6/p38 stress kinase cascade is critical for tumor necrosis factor-alpha-induced expression of monocyte-chemoattractant protein-1 in endothelial cells. Blood. 1999; 93:857–865.
Article29. Carter AB, Knudtson KL, Monick MM, Hunninghake GW. The p38 mitogen-activated protein kinase is required for NF-kappaB-dependent gene expression. The role of TATA-binding protein (TBP). J Biol Chem. 1999; 274:30858–30863.
Article30. Stein B, Baldwin AS Jr, Ballard DW, Greene WC, Angel P, Herrlich P. Cross-coupling of the NF-kappa B p65 and Fos/Jun transcription factors produces potentiated biological function. EMBO J. 1993; 12:3879–3891.
Article31. Nemoto S, DiDonato JA, Lin A. Coordinate regulation of IkappaB kinases by mitogen-activated protein kinase kinase kinase 1 and NF-kappaB-inducing kinase. Mol Cell Biol. 1998; 18:7336–7343.
Article32. Kim JM, Cho SJ, Oh YK, Jung HY, Kim YJ, Kim N. Nuclear factor-kappa B activation pathway in intestinal epithelial cells is a major regulator of chemokine gene expression and neutrophil migration induced by Bacteroides fragilis enterotoxin. Clin Exp Immunol. 2002; 130:59–66.
Article33. Han SW, Sa KH, Kim SI, Lee SI, Park YW, Lee SS, et al. CCR5 gene polymorphism is a genetic risk factor for radiographic severity of rheumatoid arthritis. Tissue Antigens. 2012; 80:416–423.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Cytokines in rheumatoid arthritis
- Multiple Signaling Molecules are Involved in Expression of CCL2 and IL-1beta in Response to FSL-1, a Toll-Like Receptor 6 Agonist, in Macrophages
- Effect of Salicylate on the Monocyte Chemoattractant Protein-1 Expression and Intracellular Reactive Oxygen Species Formation in Human Mesangial Cells
- Clinical significance of rheumatoid factor in juvenile rheumatoid arthritis
- Intracellular Signaling Pathways that Regulate Macrophage Chemokine Expression in Response to Mycobacterium abscessus